Abstract
OBJECTIVE To explore the effects of cisplatin chemotherapy via the vein and the abdominal cavity on the functions of endothelial cells of mice with non-small cell lung cancer.
METHODS 75 mice were divided equally into a untreated group, a group treated with chemotherapy (cisplatin: 2 mg/kg, 0.5 mL) via an intravenous method and a group treated with chemotherapy (cisplatin: 2 mg/kg, 0.5 mL) via an intraperitoneal method; changes in the morphology and ultrastructure of vein endothelial cells were observed.
RESULTS The injury of the intima of the intravenous chemotherapy group was similar to that of the intraperitoneal chemotherapy group.
CONCLUSION The injury of vein endothelial cells is not significantly related to the choice of intravenous injection or intraperitoneal injection.
keywords
Introduction
Venous thromboembolism (VTE) is a common complication in the natural course of disease or in the treatment of a tumor, and chemotherapy is an independent risk factor for the occurrence of VTE among tumor patients. The incidence rate of VTE among patients with a malignant tumor is 4 times that of the general population, increasing to 6 times that of the general population among patients undergoing chemotherapy. Together with the development of combined treatment technology for tumors, such as interventional therapy, use of non-intravenous chemotherapy has also expanded rapidly[1]. Non-intravenous chemotherapy has received great attention for the clinical treatment of tumors because it can act directly on the tumor-feeding artery and, in combination with embolism treatment, improve the overall clinical effect. In theory, non-intravenous chemotherapy can prevent the chemotherapy drugs from acting directly on the veins, therefore reducing the injuries of corresponding vascular endothelial cells[2]. However, no reports are available indicating whether, in practice, non-intravenous chemotherapy does reduce vein injury.
Materials and Methods
Materials
Mice models of non-small cell lung cancer were prepared, and cisplatin chemotherapy carried out on the tumor-bearing mice. Electron microscopy was used to observe the morphology of vascular endothelial cells, and the von Willebrand factor (vWF) immunohistochemistry method was used to follow changes in the injury of vascular endothelial cells and vWF staining of the vein intima of mice with Lewis lung cancer in the natural course of disease and of those undergoing intravenous and non-intravenous chemotherapy. In a preliminary investigation, this paper explores the relationship between the injury of vein endothelial cells and the natural course of disease and combined cisplatin chemotherapy and assesses whether or not non-intravenous chemotherapy has advantages over intravenous chemotherapy, resulting in less injury to vascular endothelial cells.
Methods
The experimental animals comprised 75 male C57BL6 mice weighing 20–25 g. They were randomly divided into an untreated group, a group treated with cisplatin via an intravenous method and a group treated with cisplatin intraperitoneally, with 25 mice in each group. After preparing the models of Lewis lung cancer, and immediately after completion of cisplatin chemotherapy treatment, which was considered the starting point, the mice were randomly divided into a 3-day group, 7-day group, 14-day group, 21-day group and 28-day group, with 5 mice for each time point. Experiments were carried out in complete accordance with the requirements of animal ethics standards.
In the group receiving intravenous injection of cisplatin 4 days after implantation of Lewis tumor cells, the tumor in the abdomen grew to a size of approximately 0.5 cm3. Then 2 mg/kg (0.5 mL) cisplatin was injected into the mice through the caudal vein once each day for 2 consecutive days. The group undergoing intraperitoneal injection of cisplatin was injected with the same dosage of cisplatin at the same time points and with the same frequency.
At the observation time points, pathomorphological observation and a vWF immunohistochemical study were carried out on the mice.
The MRAS-2000 Image Analysis System of BUAA Center was used to examine the tissue sections. An examination index was obtained-defined as the ratio of the percentage of the staining granule area to the area of the vascular intima and media. Examination was carried out by the lumen analysis module predesigned by the image analysis software.
Statistical analysis
SPSS13.0 Statistical Software was used for the statistical processing. The covariance analysis method was used, and P < 0.05 was considered a significant difference.
Results
Immunohistochemical observation
vWF immunohistochemical observation was carried out on these mice. The microscope was preset at a high magnification level of SP × 400, and the quantity and description of granules stained for vWF were as follows: rare (stained granules ≤ 3); micro-quantity (4–9 stained granules); small quantity (10–15 stained granules); medium quantity (16–24 stained granules); large quantity (≥ 25 stained granules).
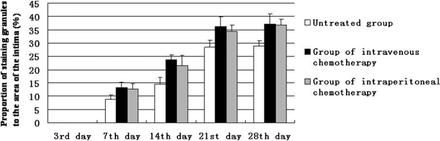
Over time, the quantity of granules staining brown at the endothelial layer and the subendothelial layer of the femoral vein in the three groups of mice showed a trend towards a steady increase, but the differences were not significant during the early period (3rd and 7th day). This indicates that injury to the endothelial cells of the femoral vein increases gradually, but is not significant during the early period.
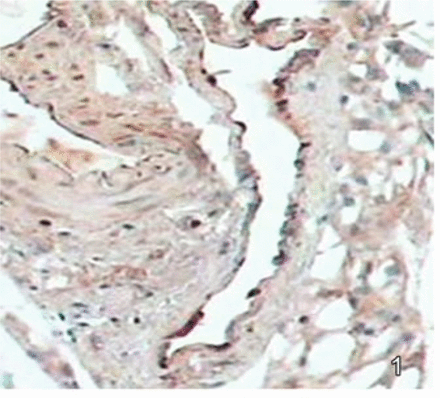
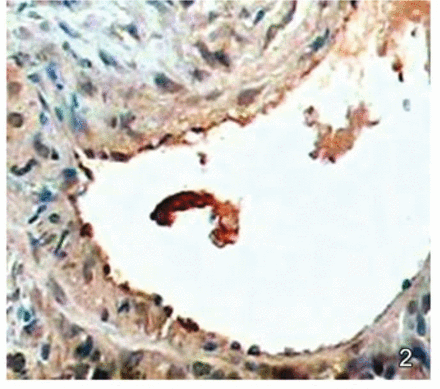
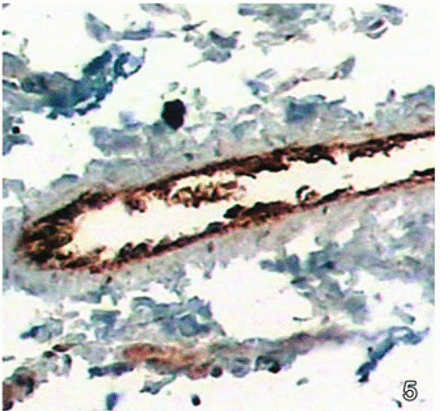
Visual analysis with a microscope at different time points of the endothelial and subendothelial layers of the femoral vein showed no significant differences between the three groups in the quantity of granules staining brown on the 3rd and 21st days; but differences were seen in the amount of staining on the 7th and 14th days (Figs.1 and 2). The differences were especially significant on the 14th day, when the quantity of granules staining brown at the endothelial and subendothelial layers of the femoral vein in the untreated group was significantly lower than that of the groups receiving intravenous chemotherapy or intraperitoneal chemotherapy. Thus the three groups had a similar degree of injury of vein endothelial cells during the first period (3rd day) but by the 7th and 14th days, the level of injury to vein endothelial cells of mice in the groups receiving intravenous chemotherapy or intraperitoneal chemotherapy was higher than that of the untreated group (Figs.1–3). By the 21st day, mice in all the three groups had a high level of injury to the vein endothelial cells, and no significant differences could be seen by visual inspection with a microscope (Figs. 4 and 5). At the last observation (on the 28th day), all the groups had roughly the same extent of damage, which was similar to the damage occurring naturally in the late period of the disease.
On the seventh day after cisplatin chemotherapy, a micro-quantity of brown granules could be seen staining the intima of the femoral vein of mice in the untreated group.
On the 14th day after cisplatin chemotherapy, a small quantity of brown granules could be seen staining the intima of the femoral vein of mice in the untreated group.
On the 14th day after cisplatin chemotherapy, a medium quantity of brown granules could be seen staining the intima of the femoral vein of mice in the intravenous chemotherapy group.
On the 21st day after cisplatin chemotherapy, a large quantity of brown granules could be seen staining the intima of the femoral vein of mice in the intravenous chemotherapy group.
On the 21st day after cisplatin chemotherapy, a large quantity of brown granules could be seen staining the intima of the femoral vein of mice in the untreated group.
The MRAS-2000 Image Analysis System was used to examine the tissue sections. The results of the examination and statistical analysis are shown in Fig.6 and Table 1.
Expression of granules stained for von Willebrand factor at the intima of the femoral vein.
Expression of granules stained for von Willebrand factor at the intima of the femoral vein.
Electron microscopy observation
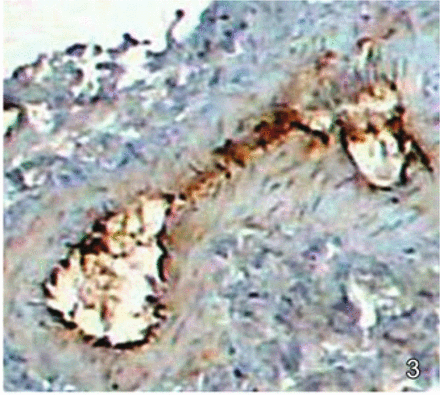
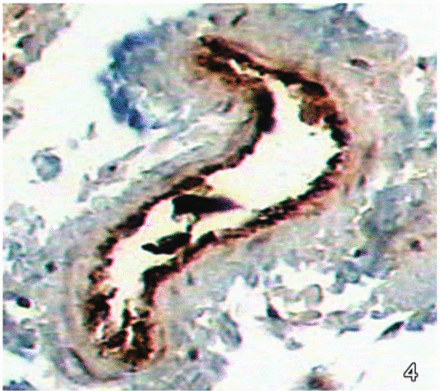
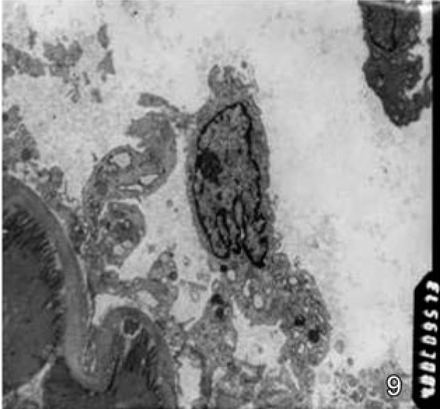
Results of electron microscopy of the endothelial layer of the femoral vein of mice in the untreated, intravenous chemotherapy and intraperitoneal chemotherapy groups showed a change from a continuous to a discontinuous state and the morphology of the endothelial cells changed from regular to irregular. Changes were the greatest on the 21st day, and this is generally consistent with the results of immunohistochemistry. Among the three groups, on the 14th, 21st and 28th days, a greater number of enlarged Weibel-Palade bodies were found in the endothelial cells of the femoral vein of mice in the intravenous chemotherapy and intraperitoneal chemotherapy groups, and the trend towards separation of endothelial cells from the intima was more apparent in these two groups. This indicates that injury to the endothelial cells was more serious in these two groups than in the untreated group (Figs. 7–9).
On the third day after chemotherapy, the endothelial cells of the femoral vein of mice in the untreated group were continuously arrayed and tightly adhered to the basal membrane, the proportion of karyotypes was normal, the organelle morphosis was normal, and only a small quantity of synthesized granules was visible.
On the 14th day after cisplatin chemotherapy, the endothelial cells of the femoral vein of mice in the intravenous chemotherapy group had closely adhered to the basal membrane, but the cell surface was rough, the cells were in an activated state, the cells were hypertrophied, the cytoplasm was filled with various types of synthesized granules, and enlarged Weibel-Palade bodies could be seen in the cytoplasm.
On the 21st day after cisplatin chemotherapy, the endothelial cells of the femoral vein of mice in the intravenous chemotherapy group were not continuously arrayed, large quantities of endothelial cells were detached and separated from the basal membrane, the endothelial cells were in an activated state, the cell surface was rough, the cytoplasm was filled with synthesized granules, the proportion of karyotypes became unbalanced, and there was a significant increase in heterochromatins.
The MRAS-2000 Image Analysis System was used to examine the tissue sections. The results of examination and statistical analysis are shown in Table 2.
Expression of granules stained for von Willebrand factor at the intima of the femoral vein (electron microscope).
Discussion
Vascular endothelial cells have an antithrombotic function. The negative charges on the endothelial surface, and active substances released by endothelial cells, prevent adhesion and agglutination of platelets and facilitate fibrinolysis, which inhibits blood coagulation. When vascular endothelial cells are damaged, the amount of the active substances released decreases, leading to a favorable condition for the thrombus formation. Injury to vascular walls by chemotherapy drugs is one of the major reasons for vein thrombosis[3].
Endothelial cells are well-differentiated monolayer tubular cells that adhere to the surface of blood vessels of the body’s circulatory system. They provide a major protective screen between blood flow and the vascular wall, and can also synthesize and secrete many kinds of peptides and proteins which directly participate in pathophysiological processes[4]. Endothelial cells secrete TM-selectin, TF-selectin, E-selectin, plasminogen activator, antithrombin III, platelet activation factor and adherence factor to maintain an equilibrium between coagulation and anticoagulation in the body[5]. Because endothelial cells come into direct contact with the circulating blood, they are the cells first affected by pathophysiological changes in the blood.
Research indicates that activation of vein endothelial cells is the key step leading to vein thrombosis in tumor patients. When vein endothelial cells are activated, they synthesize and secrete several cytokines associated with blood coagulation. These cytokines regulate the coagulation activity and cellular adhesive capacity of the blood system, which ultimately facilitates vein thrombosis[6, 7].
This experiment used two methods for studying the relationship between combined chemotherapy for non-small-cell lung cancer and the injury of vein endothelial cells: observation of the morphology of endothelial cells by electron microscopy and the vWF immunohistochemistry count[8]. The results indicate that when endothelial cells are activated, their morphology and function changes. These changes are clearly apparent in the following ways: the surface of the endothelial cells becomes rough, expression of many kinds of molecule on the surface of the membrane increases, hypertrophy of cells occurs, the cytoplasm is filled with synthesized granules, enlarged Weibel-Palade bodies can be seen in the cytoplasm, the proportion of karyotypes becomes unbalanced, there is a significant increase in heterochromatins, the endothelial cells are not continuously arrayed, the gaps between cells widen, and some endothelial cells become detached and separated from the basal membrane[9,10].
This result is consistent with the changes in endothelial cells observed in previous studies on non-tumor diseases. Although the injury to endothelial cells seen by electron microscopy is similar, the mechanisms at work are different in these two cases. In non-tumor diseases, the vascular endothelial cells are injured by inflammatory factors, autologous immune complexes and hemorheologic changes[11]. In the case of tumors, the vascular endothelial cells are injured by cytokines directly or indirectly released by malignant tumor cells. Whether there are other means of injuring vascular endothelial cells in the case of tumors and what the working mechanisms for these might be requires further study.
After endothelial cells have been activated, vWF in the Weibel-Palade bodies in the cytoplasm is released into the blood plasma. vWF is an adhesion molecule, whose function is to activate platelets and induce them to adhere to the vascular walls[12]. Therefore, the quantity of vWF released reflects the degree of activation of endothelial cells and is a sensitive molecular marker of injury to endothelial cells.
Our experiment used immunohistochemistry to observe adhesion of vWF to the vascular walls and image analysis software to calculate the quantity of vWF adhering. As previously mentioned, the quantity of vWF released reflects the degree of activation of endothelial cells. Therefore, it is possible to prove by calculating the quantity of vWF released that malignant tumor cells damage and activate endothelial cells, and to show that the longer the course of the disease, the more serious is the injury to the endothelial cells[13]. Our experiment, which by electron microscopy and immunohistochemistry observed the morphology of vascular endothelial cells proved that non-small cell lung cancer does cause injury to the endothelial cells of the veins of mice and that the degree of damage increases with time.
Chemotherapy is an independent risk factor for the occurrence of VTE among tumor patients. According to statistics, the incidence rate of VTE among patients with a malignant tumor is about 10.9%[14], which is 4 times greater than that of the general population, and for patients with malignant tumor undergoing chemotherapy is even higher, at about 6 times that of the general population[15]. Similarly, the recurrence rate of VTE among patients with a malignant tumor is twice that of the general population, and 4 times that of the general population for patients undergoing chemotherapy[16].
Our experiment showed, using electron microscopy and immunohistochemistry, that the degree of injury to endothelial cells of mice in the groups receiving intravenous chemotherapy or intraperitoneal chemotherapy is greater than that of mice in the untreated group. Many publications have reported that injury to endothelial cells is the chief step for the formation of VTE[17]. Our observation of injury to endothelial cells in this experiment further illustrates the correlation between chemotherapy and the occurrence of VTE. During this experiment, injury to the endothelial cells in all the three groups increased with time, which is consistent with the observation of a higher incidence rate of VTE during the advanced stages of cancer as shown in a previous study[18]. However, as far as we know, no studies have been published indicating whether the VTE incidence rate after cisplatin chemotherapy can be reduced by changing the method of administration.
At the same time points, mice in the two groups receiving cisplatin chemotherapy had more granules stained for vWF than those in the untreated group, and electron microscopy demonstrated that injury to the endothelial cells was more serious, and the Weibel-Palade bodies were thicker and more abundant in the former two groups[19]. These results suggest that the rise in the level of vWF in Lewis mice may be due to activation of the vascular endothelial cells by the malignant tumor cells, and the increased level of vWF after cisplatin chemotherapy may be due to a synergistic effect between activation of the vascular endothelial cells by the malignant tumor cells and by cisplatin. In vitro experiments conducted by Togna[20] and clinical analysis by Licciardell[21] indicated that chemotherapy may raise the level of vWF in the blood plasma of tumor patients, which, in turn, facilitates the adhesion and concentration of platelets. Our study further suggests that this effect might be explained by the injury to the endothelial cells by cisplatin. However, the experimental data show that there are no significant differences between intravenous and intraperitoneal injection of cisplatin in the degree of injury to endothelial cells of the animals[20]. This shows that after cisplatin chemotherapy of the tumor-bearing animals, the chemotherapy drug has a direct and local action but this is not the sole factor.
The group receiving non-intravenous injection of cisplatin had less injury to the vascular endothelial cells during the early period, but the difference is not statistically significant. As the disease progresses, and the concentration of the drug in the blood reaches a certain level, no significant differences in injury to the vein endothelial; cells are found between the groups. This indicates that, although non-intravenous chemotherapy can, in theory, avoid direct injury to local veins, its contribution to prevention of damage of the vein system of the whole body is limited. This is because injury to the endothelial cells by the chemotherapy drug is accompanied by blood circulation, and the proportion of chronic damage is significantly greater than that of acute injury. The precise mechanism of injury to the veins needs to be verified by further study.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received January 12, 2011.
- Accepted May 10, 2011.
- Copyright © 2011 by Tianjin Medical University Cancer Institute & Hospital and Springer