keywords
Cytophagic histiocytic panniculitis (CHP) was first described by Winkelmann and Bowie in 1980.[1] It is a rare group of diverse illnesses involving benign and malignant proliferation of macrophages in various organs and tissues. It presents with subcutaneous panniculitis with or without a hemophagocytic syndrome (HPS). It occurs predominantly in women (male: female ratio 1:1.3) between the years of 5—61 (average, 33.5). The major clinical features are recurrent fever, multiple panniculitic lesions, anemia, leukopenia and coagulation abnormalities. In the later phase, liver dysfuction, serosal effusion, mucosal ulceration and hemorrhage may occur. Histological findings show activated histiocyte infiltration of the fat tissue. Cytologically the benign-looking histiocytes containing cell fragments (bean-bag cells) are very typical.[2] CHP has a broad spectrum from mild to severe. Benign CHP is selflimiting and sensitive to treatment, but up to now there is no effective therapy for malignant CHP. We report here a case of progressive and fatal cytophagic histiocytic panniculitis in a young woman who had encephalodastic changes immediately prior to her death.
Case Report
A 21-year-old girl was first admitted to the No.2 Affiliated Hospital of China Medical University on March 23, 2005 having had a subcutaneous nodule in her right axilla for 10 days and a high fever for 7 days. Her medical history was significant for feeling a painful firm peanut-like subcutaneous nodule in her right axilla. The size of the nodule gradually enlarged over 10 days. No additional nodules on her extremities or trunk were noted but there was a progressive swelling of the right breast without pain. The continuous 7-day fever which occurred prior to entering the hospital reached 39.5 °C. There was no accompanying tremor. Past medical and family history were unremarkable.
On physical examination, the patient was febrile with a temperature of 39.5 °C. There was no evidence of ulceration and no lymphadenopathy. The subcutaneous nodule in her right axilla was characterized as follows: 6x8 cm in size, hard, painful, with a unclear rim, Received March 13, 2006; accepted May not movable and no redness or edema on the surface. There was obvious swelling of her right breast and right upper arm. A respiratory and cardiovascular examination was normal. There was moderate hep-atosplenomegaly.
Laboratory investigations showed pancytopenia, WBC: 1.9xl09/L [normal range (NR): 3.6~9.7xl09/L], HbG: 118 g/L (NR: 118-160 g/L), PLT: 90xl09/L (NR:150~400xl09/L); abnormal hepatic fimctions, elevated alkaline phosphatase (ALP) 138 U/L (NR: 45-132 U/L), elevated alanine transferase (ALT) 1454 U/L (NR: 5-40 U/L), elevated aspartate transaminase (AST) 1581 U/L (NR: 8-40 U), low total protein 52.3 g/L (NR: 60-83 g/L), decreased albumin 28.5 g/L (NR: 35-55 g/L). Total bilirubin, direct bilirubin and indirect bilirubin were in the normal range. There was decreased fibrinogen, 1.786 g/L (NR: 2-4 g/L) and negative disseminated intravascular coagulation (DIC) results. Immunoglobulin levels of IgA, IgG, and IgM were within the normal range. Viral hepatitis (A-E), Epstein-Barr virus, cytomegalovirus (CMV), human immunodeficiency virus (HIV) and two blood cultures were all negative showing no infective agents. Tests for antinuclear antibody (ANA) and rheumatoid factor gave negative results. Serum Fe was 21.6|xmol/L (NR: 10-25 (xmol/L) and total iron binding capacity 74.1 pjnol/L (NR: 54-77 pmol/L). Ferritin was markedly elevated to more than 1,500 ng/ml (NR: 11-336 ng/ml), the Coombs' and Ham’s tests were also negative. Examination of CA19-9, CEA, CA15-3, AFP, CA125 (prompting tumor existence) were in the normal range.
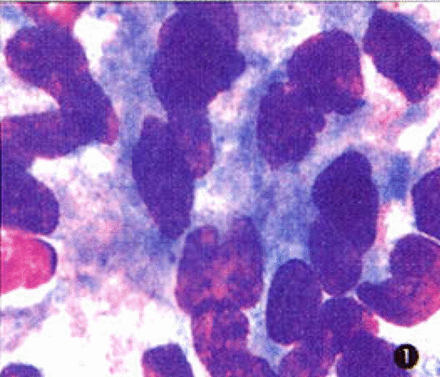
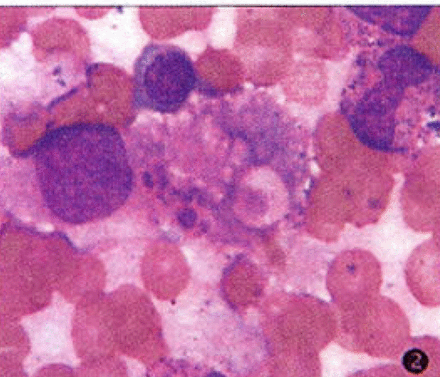
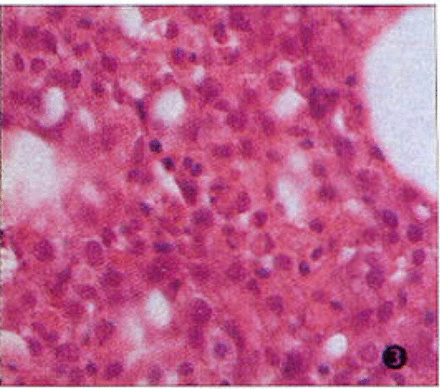
The chest radiograph and ECG were normal. Ultrasound showed multiple subcutaneous nodules in the right axilla and right subclavian area, the biggest one having a size of 2.4x3 cm. There was diffused swelling of the right breast, wide subcutaneous edema in both the right arm and chest. Abdominal ultrasound confirmed moderate hepatosplenomegaly. Bone marrow biopsy showed a hypercellular marrow with histiocyte hyperplasia, some histiocytes and occasionally phagocytosed white blood cells. Red cells and cell particles, described as hemophagocytosis were also present, but there was no evidence of infiltration by lymphoma cells Fig.1,2. A biopsy of the nodule in the right axilla showed benign subcutaneous panniculitis. Striking histopathologic changes were found in the subcutaneous fatty tissue characterized by inflammatory infiltration of typical lymphocytes and histiocytes and classic 'bean-bag' cells (i.e histiocytic cells containing nuclear fragments) Fig.3. The histiocytes and lymphocytes appeared benign and had normal nuclei without heterochromatin, and normal nuclear-cytoplasmic ratios. Immunohistochemical studies showed the following CD3(++), CD68(+++), CD74(++), CD20(-), CD30 (-), kappa(-), lambda(-).
Many histiocytes infiltrated in the bone marrow, WG * 1000.
Histiocytes infiltrated in the bone marrow with phagocytosed white blood cells, red cells and cell particles, described as hemophagocytosis, WG x 1000.
Subcutaneous histiocytic cells containing nuclear fragments to form ' bean-bag' cells, H&E x 400
According to the clinical and histologic findings, a diagnosis of cytophagic histiocytic panniculitis (CHP) was made. The patient was treated with multi-agent chemotherapy 'CHOP' (cyclophosphamide CTX, 600 mg, d 1, d 3; vincristine VCR, 2 mg; dexamethasone 10 mgx 5 d; doxurubicin 30 mgx 3 d).The patient showed marked improvement, with disappearance of fever, loss of swelling of her right breast and regression of hepatosplenomegaly. Both liver function tests and blood count values became normal. Bone marrow aspirations were repeated after chemotherapy revealing no signs of hemophagocytosis or malignancy. She was discharged on the 36th day of hospitalization, but a month later, another nodule was found in her left inguinal area so she was admitted again to our hospital. This time, there was no fever and the laboratory examinations were almost normal. A second cycle of chemotherapy ('CHOP', same dose) was used. The nodule disappeared after chemotherapy and the patient was discharged again. Sixteen days after the second dischargement, she developed a fever, another 3x5 cm nodule was found in the right axilla and there was swelling of her right breast. Laboratory examinations and bone marrow aspirations presented as normal. 'CHOP' (the CTX was increased to 1 g, d 1, d 3) was given for the third time, but this time she resisted the chemotherapy so the symptoms failed to disappear. The patient complained of a headache and was somnolent. Computed tomography (CT) of her head and cranial magnetic resonance imaging (MRI) were performed, but no significant findings were revealed. CSF examination showed: pressure was 1.9 kPa, trasparent, protein was 0.6 g/L, glucose was 2.7 mmol/L, chloride was 126 mmol/L, cell count was 2xl06/L. The disease developed rapidly, she convulsed, showed obnubilation, and soon died on June 11, 2005.
Discussion
Cytophagic histiocytic panniculitis is the term commonly used to refer to a lobular panniculitis with an inflammatory infiltrate of small, benign, mature lymphocytes and benign histiocytes.[1]
The cause of CHP is unknown. It is thought that CHP is the product of immune dysrégulation caused by cytokines released by CD4-positive T helper lymphocytes (benign or malignant).[3] Many cytokines, including phagocytosis initiating factor, tumour necrosis factor and interleukin have been implicated. The immune disorder may be associated with many factors, including infections (EB viral etc)[4] connective tissue disorders,[5] nodal and cutaneous lymphoma etc. In ornease we reported neither infections (bacterial and viral) nor connective tissue disorders[6]
The diagnosis of CHP is mostly based on the pathological analysis. Histologically, CHP is characterized by a lobular panniculitis with an infiltrate of small, benign, mature lymphocytes and benign histiocytes. Classically, evidence of cytophagocytosis may be seen in the skin and organs of the reticuloendothelial system (liver, spleen and bone marrow), which are not seen in other causes of lobular panniculitis.
CHP may run a benign chronic course, but rapid aggressive disease has also been reported. Many cases of CHP are now being classified as a natural disease progression of subcutaneous panniculitic T-cell lymphoma (SPTCL),[7] and a rapid aggressive CHP is considered as a lower malignant SPTCL. In some reported cases of CD4-positive SPTCL, initial histology has been consistent with CHP.[8] SPTCL is regarded to be a tumor of activated cytotoxic T cells. In 2005, SPTCL was classified as T cell and NK cell lymphoma of the skin by the WHO and EORTC, and was limited to a/ß type T-cell originating lymphoma. The related articles suggested that this kind of tumor developed from the precursor of plasma cell-like dendritic cells. It also was termed as an early plasma cell-like dendritic cell leukemia/lymphoma.[9] CHP and SPTCL share many clinical and immunologic features. The infiltrating T cell is typical and mature in CHP while atypical and multimorphic in SPTCL. But in some cases, the histological appearance of CHP may be indistinguishable from SPTCL. T-cell receptor gene rearrangements have revealed evidence of monoclonal rearrangements in most cases of SPTCL and in a few cases of CHP[10] The significance of these findings is unclear. Latent EBV infection (demonstrated by in situ hybridization methods) may be found in CHP and the various types of subcutaneous lymphomas.[11] The usefulness of this investigation in distinguishing CHP from SPTCL is unclear and the exact relationship between CHP and SPTCL is still vague at the present time.
Treatment of CHP with systemic corticosteroids and immunosuppressive agents (such as cyclophosphamide or azathioprine), chemotherapy (mainly CHOP-based regimens) and cyclosporin have all been used with variable success[12] More recently, high-dose regimens (such as ifosphamide, carboplatin and etoposide or 'ICE') with autologous peripheral stem cell transplantation (APBSCT) have been used for salvage treatment of CHP with mixed success[13] Cyclosporin may be the initial treatment of choice in the cases of the haemophagocytic syndrome in CHP or SPTCL, given its comparatively safer side effect profile compared with chemotherapy. If cyclosporin fails or if disease progression occurs, chemotherapy (CHOP or a CHOP-based regimen) appears to be the best treatment option[14] High-dose chemotherapy followed by APBSCT is considered to be one of the most beneficial therapies for patients with aggressive CHP.
The prognosis of CHP is variable. The patients with benign CHP often respond effectively to chemotherapy and show a quite long survival while malignant CHP is generally resistant to treatment with corticosteroids or chemotherapy, and has a high mortality rate of greater than 70%. Rapid deterioration and death usually relate to the development of the haemophagocytic syndrome as the patient we reported.
There is a noteworthy point in our case. From the existing literature, patients with CHP mostly die from hepatic dysfunction or disseminated intravascular coagulation (DIC). However the finding of our patient who died from encephaloclastic changes has not yet been reported in the literature. Both the clinical features and pathological analysis showed that the diagnosis of cytophagic histiocytic panniculitis was correct in our case. This case was malignant CHP according to the rapidly deteriorating course and maybe progressed to early SPTCL. We inferred that the encephaloclastic changes occurred because of tumor cell infiltration after excluding cranial hemorrage or thrombosis by cranial MRI and CT. There are case reports of peripheral neural system infiltration by tumor cells of SPTCL which supported our supposition.[15] It is a rare sign of both the clinic features and the diagnosis of CHP. We trust that future developments in the diagnosis and treatment for patients diagnosed with CHP and SPTCL will lead to improved survival.
- Received March 13, 2006.
- Accepted May 8, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer