Abstract
ABSTRACT Postgastrectomy syndromes evoked by the loss of several gastric functions after total or subtotal gastrectomy are very common. Different kinds of reconstruction have been developed to overcome the problem. Patients with preservation of a duodenal passage and jejunal interposition with or without a pouch show a better quality of life and minimal symptoms postoperatively. A jejunal interposition with or without a pouch after proximal or distal subtotal gastrictomy seems to improve the nutritional condition and quality of life. The prognostic nutritional index (PNI), Visick score, Spitzer index and the European Organization for Research and Treatment of Cancer (EORTC) questionnaire (QLQ-C30) are available to evaluate the quality of life and nutritional status after gastrectomy. .
keywords
Gastric cancer is one of the most common malignancies in China. So far surgical excision of diseased gastric and nodal components remains the primary therapy. As the detection rate of early cancer has increased, owing to improved radiography, endoscopy, and ultrasonic endoscopy, some limited procedures such as mucosal resection or segmental resection under gastroscopy or laparoscopic surgery are performed in some selected cases instead of an aggressive surgical approach of gastrectomy with extended systemic lymphadenectomy.
Clinically, most patients have an invasive disease, and a radical procedure (R0) has long been the standard surgical treatment. An extended lymphadenectomy is very important for survival of the patients. According to the tumor grade as well as the gross and histologic pathologic appearance, a radical subtotal (intestinal type) or total (diffuse type) gastrectomy with D2 lymphadenectomy will be carried out in patients with cancer of the distal stomach. Total gastrectomy with D2 lymphadenectomy is the standard procedure for patients with middle located cancer. An extensive total gastrectomy with the distal escophagus is used when mandated by a proximal cancer location. If total gastrectomy is necessary, a splenectomy is usually performed because it allows a more thorough lymph node dissection of No. 10 and No. 11 nodes. Direct spread beyond the gastric wall should be treated with en bloc extended resection to achieve negative margins of resection in order to attain a curative resection. Common examples of local tumor extension include the involvement of the body or tail of the pancreas (treated by distal pancreatectomy and splenectomy), the invasion of the transverse mesocolon (often requires transverse colectomy), and the involvement of the spleen (splenectomy) or left lobe of the liver (usually requires wedge resection).
The stomach is one of the important digestive organs. Its primary functions are to serve as a reservoir to store large quantities of recently ingested food, to initiate the digestive process and to release its contents in a controlled fashion downstream to accommodate the much smaller capacity of the duodenum. Among subjects who undergo a total or distal gastrectomy reconstructed by a Billroth I or II anastomosis, a postgastrectomy syndrome frequently develops. The postgastrectomy syndrome is a group of symptoms evoked by the loss of several gastric functions. The loss of the reservoir function reduces caloric intake, together with the consequences of the removal of the pyloric sphincter results in a rapid emptying of food to the small bowel. The lower esophageal sphincter-also being resected no longer prevents alkaline reflux into the esophagus. The removal of the parietal and chief cell mass results in a diminishing of the absorption of vitamin B12 and the digestion of proteins. Lack of hydrochloric acid also causes a weakened boundary against bacterial colonization, which further reduces the micronutrient supply. Duodenal exclusion, in the case of Roux-en-Y reconstruction, adds the disadvantages of the lost duodenal passage: disturbed gastrointestinal hormonal regulation, decreased iron and calcium absorption and reduced lipid and protein uptake. Altogether these changes lead to mal-absorption, anemia, alkaline reflux esophagitis, dumping and weight loss. Therefore, postoperative nutritional consequences of patients with gastric cancer using different reconstruction procedures will be discussed in this report.
Total Gastrictomy and Reconstruction of the Digestive Tract
Total gastrectomy is one of the common surgical procedures for gastric cancer patients, especially for those tumors located in the middle and upper 1/3 of the stomach. It may result in heartburn with reflux esophagitis, early satiety, malnutrition, and weight loss.[1] Since Schlatter performed the first successful total gastrectomy in 1897, more than 70 different gastric replacements have been described.[2] However, the best type of reconstruction after total gastrectomy remains controversial. The increased incidence of the postgastrectomy syndrome may influence the method for reconstruction after total gastrectomy. The broad spectrum of reconstruction methods can be subdivided into types that exclude a duodenal passage with and without a jejunal pouch and those with preservation of the duodenal passage with or without a jejunal pouch.
Some researchers have found that a duodenal passage with a jejunal interposition indicated that there is an association of enhanced postprandial release of hormones and regulatory peptides[3, 4, 5] but others have disagreed.[6, 7]
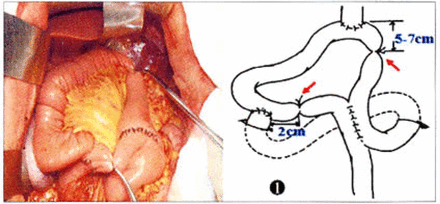
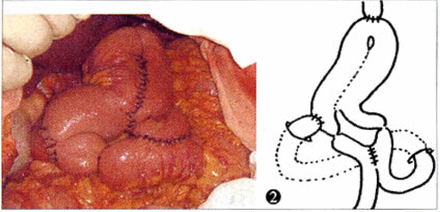
After a period of over 20 years of basic and clinical studies, a food passage and preservation of the duodenal passage using a continuous interposed jejunal reconstruction [8, 9,10] has been developed in this hospital. Laboratory studies have demonstrated that a duodenal passage with a continuous interposed jejunum enhanced absorption of glucose, iron and calcium. In comparison with the reconstruction without a duodenal passage, a continuous jejunal interposition is a reasonable reconstruction method. The construction of a jeju-nal interposition, holding food as a substitute stomach, can preserve the duodenal passage and secretion of the gastrointestinal hormones [such as secretin and chole-cystokinin-pancreozymin (CCK-PZ)], which enhance the contraction of the gallbladder, and excretion of bile and pancreatic juice resulting in better digestion of food and absorption of nutrients. The regulation of body fluids can not be restored postoperatively in a short time. Therefore, after total gastrectomy reconstruction of the alimentary tract should be performed with formation of a duodenal passage. This method simplifies the operative procedure and guarantees the blood supply for the interposed jejunum without causing ischemia at the anastomotic orifice. Using the Auto Suture surgical stapler and the Endo GIA60 linear anastomosis stapler, a continuous interposed jejunal pouch reconstruction can be carried out easily and safely. This form of gastric reconstruction is an acceptable procedure which improves the quality of life in patients undergoing total gastrectomy.
The main benefit of an original physiological reconstruction in which food passes through the duodenum, is to enhance digestion and absorption. A dumping syndrome is rare after this reconstruction because the pouch allows adequate storage function resulting in gradual passage of food into the duodenum. The storage and discharge functions of the pouch are almost equivalent to those of a normal functioning stomach. Among all the patients receiving this reconstruction, 96% are able to work at a job or to do household jobs.[11]
As a gastric substitute, the pouch acts as a physiological gastric reservoir for food storage and can allow patients to adapt immediately to their physical condition, recover and gain body weight in a short time.[12] After long-term follow-up, in comparison with those without a duodenal passage, the duodenal passage-preserving procedure shows a significant advantage in the patient’s quality of life as well as in nutritional metabolism. Animal experiments [3] indicate that the gastrointestinal dynamics and body weight recovery in mice 6 months postoperatively with a jejunal interposition are much better than those without a jejunal interposition.
Six months postoperatively, Tono et al.[4] found that the half-emptying time was 6.9 min in a jejunal-interposed (JI) group and 46 min in a jejunal-pouch interposed (JPI) group. The reservoir function influenced the recovery of the patients' body weight and production of the serum total protein in the early postoperative period. There were no dumping symptoms in the JPI group.
Hoksch et al.[5] investigated the different jejunal pouch lengths and found that the food transit time, one year postoperatively, was significantly prolonged in patients with a 15-cm pouch in comparison to those with a 7-cm pouch and to those without a pouch reconstruction (P=0.005, P=0.012). Patients with a pouch reconstruction tend to have better eating habits. An interposition of a large 15-cm jejunal pouch can lead to a prolonged food transit time with better patient nutritional status. However, an extended pouch length, results in a prolonged half-emptying time (longer than 60 min) and causes postprandial symptoms such as reflux, vomiting, and discomfort. Therefore, the ideal pouch length is around 12-15 cm.
A clinical study conducted by Mochiki et al.[12] demonstrated that an interposed jejunum with a pouch showed marked motor pattern disturbances compared to normal jejunum during fasted and fed states. These motor abnormalities may be responsible for insufficient food intake seen in the J-P group.
Other authors [13] showed no benefits, with regard to their postoperative quality of life, from reconstructing the duodenal food passage by a jejunal interposition with a pouch when compared to the widely used and technically less demanding Roux-en-Y reconstruction with a pouch. Li et al.[14] found that jejunal pouch reconstruction without a duodenal passage was superior soon after operation to a simple jejunal interposition relative to gain of body weight, volume of food intake and frequency of food intake.
The lack of standardization and the variable outcomes reported by these clinical trials fails to support any single operation as the preferred method of reconstruction following total gastrectomy. Furthermore, the selection of an optimal technical procedure may not reflect the operation that will enable an optimal quality of life for the individual patient.
Subtotal Gastrectomy and Reconstruction of the Digestive Tract
The Roux-en-Y and Billroth II techniques are commonly used to reconstruct the stomach after distal gastrectomy. The problems associated with these techniques are decreased food volume, digestive absorption failure with resultant malnutrition, dumping, reflux esophagitis and residual gastritis. The most common post-operative problem is reflux esophagitis after proximal subtotal gastrectomy. Some authors try to use an interpositioned, jejunal pouch reconstruction to overcome these problems.
Hoshikawa et al.[15] compared a groups of female beagle dogs. One group underwent proximal gastrectomy with a jejunal pouch interposition (JP) and the other group esophagogastrostomy (EG). The time course of the development of electrophysiological changes based on electromyograms was compared between the 2 groups. The number of action potentials per unit time, the mean amplitude, and the length of the resting period in the preprandial state were significantly different. All parameters tended to be normalized sooner after surgery in the JP group. The same researchers[16] performed proximal gastrectomy in 44 patients with tumors in the upper third of their stomach; 21 had reconstruction using a jejunal pouch interposition between the esophagus and the remnant stomach (JP), while 23 had reconstruction by esophagogastrostomy (EG). The postoperative course of patients were reviewed in terms of symptoms, weight maintenance, nutritional status, blood chemistry values, endoscopic findings, and radiographic appearance after a barium meal. Concentrations of gastrointestinal hormones were measured in response to a test meal. The JP procedure permitted increased dietary volume and those patients experienced fewer severe postoperative symptoms than the EG group. After operation, all the patients in both groups showed hypergastrinemia and all the patients in the JP group developed hypersecretinemia. With proximal gastrectomy, the JP procedure improved patient’s post-operative quality of life.
Tomita et al.[17] developed a novel operative technique distal gastrectomy in which the vagal nerve and pyloric sphincter were preserved. This reconstruction was performed by interposition of a 5 cm jejunal J pouch with a 3 cm jejunal conduit as a function preserving technique to prevent postgastrectomy disorders. During the excision with lymph nodes, the hepatic and celiac branches of the vagal nerve bifurcating from the anterior and posterior trunks were preserved. The antrum was severed 1.5 cm from the pyloric sphincter, preserving the arteria supraduodenalis. The substitute stomach was created as a 5 cm jejunal pouch with a 3 cm jejunal conduit for orthodromic peristaltic movement using an automatic suture instrument to complete a side-to-side anastomosis of the folded jejunum. The anal side of the gastric remnant was manually anastomosed with a jejunal J pouch, and anastomosis of the pyloric antrum with the jejunal conduit was manually completed by stratum anastomosis. Post-operatively, the procedure alleviated gastrointestinal symptoms such as appetite loss, epigastric fullness, reflux esophagitis, early dumping syndrome, body weight loss, endoscopic reflux esophagitis, endoscopic gastritis in the remnant stomach, postprandial stasis of the substitute stomach, and postgastrectomy cholecystolithiasis better than those patients who underwent conventional distal gastrectomy. The results suggested that the proposed technique is a function-preserving gastric operation appropriate for preventing postgastrectomy disorders.
Ikeda et al.[18] performed a modified interposition double-jejunal pouch after distal gastrectomy. This method uses a triangulating stapling technique with wide end-to-side anastomosis between the residual stomach and the pouch. In all patients, the anastomosis site was without leakage or stenosis, and there were no episodes of severe reflux esophagitis, residual gastritis, or dumping syndrome. The mean pooling rate was 44.2%, and emptying half-time was 73.0 min. After 2 years the patients' body weight was 91.3% of the preoperative weight, the food volume was 89.2% of normal intake, and meal frequency was 3 per day.
Proximal gastrectomy has been reappraised as the surgical procedure of choice for upper third gastric cancer. Although some concerns remain about the oncological adequacy of proximal gastrectomy, the data published so far are promising. Retrospective studies comparing proximal to total gastrectomy showed no differentces in the long-term survival between the 2 groups.[19, 20] On the other side, the major concern after proximal gastrectomy is the risk of reflux esophagitis as a result of direct anastomosis between the esophagus and the remnant stomach.[21] To avoid this complication, gastric substitutes with sufficient capacity for food have been made by interposition of a jejunal pouch which has been reported not only to provide a reservoir function and nutritional adventages but also to prevent reflux esophagitis.[22, 23] A recently published randomized study, comparing a proximal gastrectomy with a jejunal pouch interposition (PGJP) with a total gastrectomy with Roux-en-Y esophagojejunostomy (TGRY),[24] showed that more favorable results were obtained after PGJP in all aspects of postgastrectomy symptoms: the overall rate of postgastrectomy symptoms was significantly lower in the PGJP group than in the TGRY group (32% vs. 73%, P<0.012). The rates of reflux esophagitis after PGJP and TGRY were 19% and 4%, respectively. Patients in the PGJP group had the adventage of a larger food intake and subsequently better maintenance of body weight throughout the follow-up period.
In order to obtain a satisfactory nutritional condition and quality of life, a gastric reconstruction following gastrectomy should aim to have (1) a near-normal dietary volume, (2) similar rates of digestion and movement of food into the duodenum, and (3) no esophageal reflux. The ideal reconstruction should function well and the operative procedure should be simple. Further prospective randomized comparisons of standardized operations designed to achieve a good quality of life will have to be performed in order to determine if there is, in fact, a superior approach to restoring intestinal continuity.
Nutritional and Quality of Life Assessment of Patients after Gastrectomy
The prognostic nutritional index (PNI) devised by Onodera et al.[25] is used to investigate the nutritional condition of patients. It is calculated by the formula for percentage of 10x(ABL)-0.005x(L), where ABL is the serum albumin level (g/dL) and L is the absolute lymphocyte count in the peripheral blood. The PNI ratio (PNIR) is calculated as the percentage by the PNI at each time/pre-illness PNI. The results are compared between the 2 groups.
The Visick score[26] and Spitzer index[27] are the most commonly used methods to assess the patient’s quality of life. The patients are instructed to make their own choice of score points in the Visik or Spitzer questionnaires. With the Spitzer index five aspects of daily life (activity, daily living, health, support from other people/family, and outlook such as future perspective of the patient’s life) are recorded. Visick scoring consists of five score levels.
1=No complaints whatsoever; patient goes to work or follows a hobby.
2=Patient is well; precise interrogation reveals light symptoms such as dumping, fullness, occasional anorexia; patient goes to work or follows a hobby.
3=Patient needs doctor’s help regularly; he or she cannot work; patient is mobile; symptoms can be treated.
4=Patient has troublesome symptoms and cannot leave home; patient needs constant doctor’s help; body weight problems.
5=Patient is in bed; repetitive admissions to hospital; recurrent disease; re-operation.
The European Organization for Research and Treatment of Cancer (EORTC) questionnaire (QLQ-C30) [28] can be used (see Appendix-2) to evaluate the quality of life after gastrectomy. This questionnaire has been validated and proved to be specific for the LQ of patients with cancer. The QLQ-C30 is composed of both multi-item scales and single-item measures. They include 5 functional scales, 3 symptom scales, a general health status, and 6 single items. Each of the multi-item scales includes a different set of items (i.e., no item occurs in more than one scale). All of the scales and single-item measures have scores from 0 to 100. Thus a high score for a functional scale represents a high/ healthy level of functioning, a high score for the global health status represents a high LQ, but a high score for a symptom scale item represents a high degree of symptomatology or number of problems. The evaluation of the LQ is accomplished preoperative and usually after re-established postoperative periods: 14 days and 3, 6, and 12 months. Filling in the questionnaire takes the patients about 10 to 15 min.
Agreement on the parameters to be used for measure quality of life, as well as the index to assess it, should be a future objective. Further prospective randomized comparisons of standardized operations de signed to achieve a good quality of life will have to be performed in order to determine if there is, in fact, a superior approach to restoring intestinal continuity.
Appendix 1. Diagnostic criteria for dumping syndrome (Japan Society of Gastrointestinal Surgery)
Systemic symptoms Do you have cold sweats?
Is your heart pounding?
Do you have dizziness?
Do you have numbness and/or syncope?
Do you become red in the face?
Do you become pale in the face?
Does your body become hot?
Do you feel general malaise and weakness?
Do you find it impossible to resist sleepiness?
Do you have headaches or dull headaches?
Do you feel chest pressure?
Abdominal symptoms Is your stomach growling?
Do you have abdominal pain (except when drinking milk)?
Do you have diarrhea (except when drinking milk)?
Do you have nausea?
Do you vomit?
Do you have abdominal distension?
Do you have abdominal discomfort?
Early dumping syndrome is diagnosed when the patient has one or more of the systemic symptoms.
Pseudo-dumping syndrome is diagnosed when the patient has any of the abdominal symptoms.
Appendix 2. The QLQ-C30 and food consumption patterns Questions from the QLQ-C30
I. Functional scales
A. Physical functioning
1. Do you have any trouble doing strenuous activities, such as carrying a heavy shopping bag or a Suitcase?
2. Do you have any trouble taking a long walk?
3. Do you have any trouble taking a short walk outside the house?
4. Do you need help eating, dressing, washing yourself, or using the toilet?
B. Role functioning
1. Are you limited in any way when doing either your work or household jobs?
2. Are you completely unable to work at a job or to do household jobs?
C. Emotional functioning
1. Do you feel tense?
2. Do you worry?
3. Do you feel irritable?
4. Do you feel depressed?
D. Cognitive functioning
1. Have you had difficulty concentrating on something, such as reading the newspaper or watching television?
2. Have you had difficulty remembering things?
E. Social functioning
1. Has your physical condition or medical treatment interfered with your family life?
2. Has your physical condition or medical treatment interfered with your social life?
II. General health status and quality of life
1. How would you rate your overall physical condition during the past week?
2. How would you rate your overall quality of life during the past week?
III. Symptom scales/items
1. Are you short of breath?
2. Have you had pain?
3. Do you need to rest?
4. Do you have trouble sleeping?
5. Have you felt weak?
6. Have you lacked an appetite?
7. Have you felt nauseated?.
8. Have you vomited?
9. Have you had constipation?
10. Have you had diarrhea?
11. Are you tired?
12. Does pain interfere with your daily activities?
13. Has your physical condition or medical treatment caused you financial difficulties?
Questions about Food Consumption Patterns
1. How many meals do you take daily?
2. How extensive are the changes in your food consumption pattern?
All of the scales and single-item measures have scores from 0 to 100. Thus a high score for a functional scale represents a high/healthy level of functioning, a high score for the general health status represents a high QOL, but a high score for a symptom scale item represents a high degree of symptomatology or number of problems.
Picture and illustration of total gastrectomy with continuous jejunal interposition.
Picture and illustration of total gastrectomy with continuous jejunal pouch interposition.
- Received April 20, 2006.
- Accepted May 15, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer