Abstract
OBJECTIVE To investigate the antiproliferative and apoptogenic activities of peptide extracted from the Chinese toad (Bufo bufo gargarizans) skin (TSP) and its effects on hepatocarcinoma cell line.
METHODS MTT assay was used to detect the effects of TSP (50 μg/mL and 5 μg/mL) on the proliferation and viability of Hepatocarcinoma cell line (HepG2) and liver cell line (L-02); Flow cytometry was used in DNA content analysis to determine the cell distribution in different phases of cell cycle; Annexin V-FITC/PI stained fluorescence-activated cell sorter (FACS) and transmission electron microscope (TEM) were used to detect the apoptosis of the treated cells.
RESULTS TSP could not suppress the proliferation and viability of normal liver L-02 cells, but strongly inhibited the proliferation and viability of HepG2 cells; TSP (50 μg/mL) primarily arrested the HepG2 cells at G1 phase of the cell cycle; TSP (50 μg/mL) induced apoptosis in HepG2 cells and enhanced the effects of 5-Fu.
CONCLUSION TSP has potent antineoplastic activity against human hepatocarcinoma cells with little toxicity to normal liver cells and can enhance the effects of 5-Fu.
keywords
Introduction
Cancer is a complex multifactorial genetic disease. Transformed cells are controlled by certain genes which regulate proliferation of normal cells. Nowadays, chemotherapy is one of the most important modalities for cancer treatment. However, the effects of the chemo-drugs on most solid tumors are disappointing.
Drug discovery against cancer is ventured throughout the world especially from the natural products. The skin of the amphibian skin fulfils a wide range of functions necessary for the amphibian’s survival. It is evident that amphibians, such as frogs and toads possess various bioactive substances in their skin[1]. Earlier investigation in the Indians revealed that Indian toad (Bufo melanostictus) skin extract was pharmacologically potent, including immunomodulatory and antineoplastic activities in Ehrlich ascites carcinoma bearing mice[2,3]. Up to now, a number of cationic peptides extracted from the skin of various amphibians have been isolated and reported that some of their derivatives show anti-tumour activity with little toxicity against non-malignant cells either by triggering apoptosis[4–6] or by forming ion channels/pores[7]. Furthermore, some peptides were found to be cytotoxic against Multidrug Resistance (MDR) cancer cells. The discovered peptides in the amphibian has encouraged further study and exploitation for novel amphibian biochemicals.
In traditional Chinese medicine, toad skins are usually treated in whole as drugs to treat tumours, and its anticancer activities particularly interest many cancer researchers. Bufalin is one of the prominent components of bufadienolides which was isolated from “Chan Su” a Chinese traditional medicine. Its pharmacological activities, as an anticancer agent, have been reported. A number of researches focusing on the anticancer activities of Chinese toad have been in progress around the world to analyze the biogenic amines, bufodienolides, alkaloids and steroids isolated from the skin secretions, but few published evidences are available on its skin extract. The present communication is an approach to study the anticancereffects of Chinese toad skin extract on hepatocarcinoma cells.
Materials and Methods
Preparation and collection of toad skin extract (named as ‘TSP’)
Chinese toads were collected from the local farmlands during October and November. The skins of the caught adult toads were prepared for use. The skins overlying the abdomen, thorax, back and legs of the toads were surgically removed, and then the animasl were subsequently sacrificed. The skins were homogenized in a chilled blender, in which 4 vol (based on initial skin weight) of 0.14 M NaCl (pH 6.0), 5 mM EDTA and 0.2% Triton X-100 were added just prior to homogenization. The foamy gray homogenate was centrifuged at 18,000 g for 20 min, and the clear supernatant was heated in water at 50°C for 10 min, centrifuged at 8,000 g for 15 min, and then the obtained precipitate was discarded. The supernatant was concentrated and desalinated in an ultrafiltrate centrifuge tube for ultrafiltration, and finally was frozen and dried in lyophilizer. Crude fraction was collected and further purified using C18 reversed-phase HPLC. After that, peptide molecular mass was analyzed through a MALDI-TOF mass spectrometer.
Human cell cultures
HepG2, a kind of human cancer cells, and L-02, a kind of normal liver cells were respectively cultured in RPMI1640 and DMEM medium which was supplemented with 10% fetal bovine serum (heat inactivated), penicillin (100 units/mL) and streptomycin (100 mg/mL). Cell lines were maintained in a humidified incubator with 5% CO2 at 37°C and passaged twice a week started at an initial density of 1×106 cells/mL.
MTT assay
The MTT cell proliferation assay was performed according to the method described by Kawada et al.[8] One hundred and fifty μL of cancer cells (HepG2) suspension in RPMI 1640 supplemented with 10% fetal calf serum containing 1×105 cells was seeded to each well of a 96-well microtiter plate. The cells were cultured in the presence and absence of TSP (5 μg/mL, 50 μg/mL), 5-Fu (25 μg/mL) or TSP+5-Fu, respectively. The plate was incubated in the humidified incubator containing 5% CO2, at 37°C in for 48 h. Then, 20 μL of MTT (5 mg/mL) was added to each well and following that the plate was incubated again in the humidified incubator with 5% CO2 at 37°C in a for 4 h. Next, 150 μL of DMSO was added to each well to dissolve the formazan crystal that formed. The optical density (OD) was recorded at 490 nm. Growth inhibition rate (%) was calculated as follows: (1-OD of treated sample/OD of control sample) × 100%.
Cell cycle analysis
For analyzing distribution of the cells in different phases of cell cycle, control cells and TSP-treated cancer cells (1×106 cells per group) were washed with PBS 24 h after TSP treated (5 μg/mL, 50 μg/mL), fixed in 70% ethanol, stained with 10 μL of propidium iodide(100 μg/mL)and then treated with 10 μL of RNaseA (5 μg/mL). The DNA content of the cells was subsequently analyzed using flow cytometry (Becton Dickinson, USA) with standard protocols. The distribution of DNA content was expressed using the percentage of the cells in each phase of G1, S, and G2.
Annexin V-FITC/PI stained fluorescence-activated cell sorter (FACS) analysis
The cells were harvested by trypsinization, washed twice with cold PBS, and centrifuged at 1,000 rpm for 5 min. The supernatant was discarded and the pellet was resuspended in binding buffer at a density of 1.0×105–1.0×l06 cells per mL. Then, 100 μL of the sample solution was transferred to a 5 mL culture tube, and incubated with 5 μL of FITC-conjugated Annexin V and 5 μL of PI at room temperature in the dark for 15 min. Following that, 400 μL of binding buffer was added to each sample. The samples were subsequently analyzed using FACS and Cell Quest Research Software.
Transmission electron microscope (TEM)
The cells were fixed in the mixture of 0.1 M phosphate buffer with 2.5% glutaraldehyde and 4% paraformaldehyde. The samples were then fixed in 1% osmium tetroxide, dehydrated in graded acetones, and flat-embedded in Epon plastic 812 in a cross-sectional orientation. Then 85-nm sections which were obtained after stained with 0.25% lead citrate and 5% uranyl acetate in 50% acetone were observed and photographed under a JEOL 100CX transmission electron microscope (Los Angeles, CA, USA).
Statistical analysis
SPSS 13.0 was used for statistical analysis. Results based on four replicates per experimental group. For all the measurements, one-way ANOVA followed by Student’s Newman Keuls (SNK) test was used to assess the statistical significance of difference between control and TSP-treated, A P-value < 0.05 was considered statistically significant.
Results
Purification and molecular characterization of the peptides
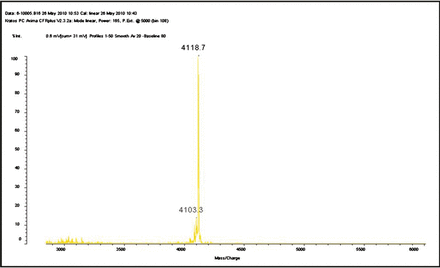
The crude fraction was divided into 6 peaks by C-18 HPLC column. The peak indicating anticancer activity was collected and the molecular mass was analyzed through MALDI-TOF mass spectrometer (Fig.1).
TSP molecular mass analyzed through MALDI–TOF mass spectrometer.
Effects of TSP, 5-Fu, combination of TSP and 5-Fu on the proliferation of HepG2 and L-02 cell lines
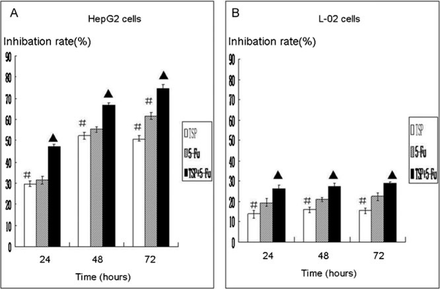
TSP produced dose-dependent inhibition effects on the growth of HepG2 cells compared to untreated control cells (P < 0.05). The inhibition rate of the cells treated with TSP (5 μg/mL) combined with 5-Fu (25 μg/mL) was significantly greater (P < 0.05) than that of the cells treated with TSP (5 μg/mL) or 5-Fu (25 μg/mL) alone. The effect of TSP on L-02 cell proliferation was mild. (Table 1, Fig.2).
Inhibition ratio of the cells treated with TSP, 5-Fu, combination of TSP and 5-Fu on the proliferation of HepG2 and L-02 cell lines
Cell growth inhibition (MTT assay) at 24 h, 48 h and 72 h in (a) HepG2 and (b) L-02 cells treated with TSP(50 μg/mL), 5-Fu (25 μg/mL), 5-Fu (25 μg/mL)+ TSP (5 μg/mL). Values represent the mean±SDAsterisk (▲) sign indicates statistically significant values compared to (#)(P < 0.05)
Cell cycle analysis
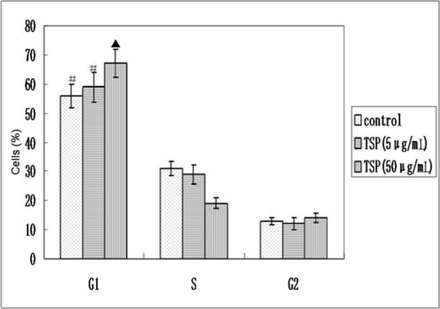
The distribution of the cells in different phases of cell cycle generated by flow cytometry revealed that HepG2 cells treated with TSP (50 μg/mL) or TSP (5 μg/mL) for 24 h exhibited a distribution of 67.3%, 18.4%, 14.3% and 58.0%, 29.2% and 12.8% of the HepG2 cells in the G1, S and G2 phases of the cell cycle, respectively. On the other hand, the HepG2 cells which were untreated showed a distribution of 56.1%, 30.9% and 13.0% of cells in G1, S and G2 phases of the cell cycle, respectively (Fig.3). The HepG2 cells treated with TSP (50 μg/mL) showed arrest in the G1 phase of the cell cycle and a decreased number of the cells in the S phase.
Effect of TSP on the cell distribution in different phases of the cell cycle. The mean percentage of the cells in G1, S and G2 phases of cell cycle are presented. Error bars represent SD. Asterisk (▲) sign indicates statistically significant values compared to (#) (P < 0.05).
Annexin V-FITC/PI stained fluorescence-activated cell sorter analysis
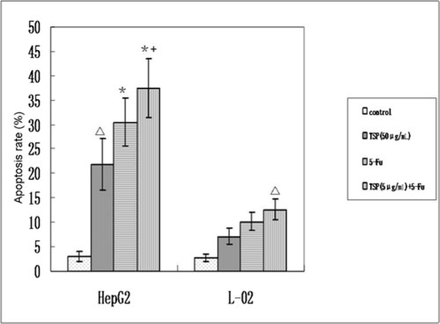
Annexin V-FITC and PI stained cells were analyzed using FACS to determine the percentage of survived or apoptotic cells after treated with TSP (50 μg/mL), 5-Fu (25 μg/mL) or TSP (5 μg/mL) + 5-Fu for 24 h. TSP (50 μg/mL) treatment induced apoptosis of the L-02 cells for 7.14%, and of HepG2 cells for 21.86%. The apoptosis rate of the hepatocarcinoma cell lines treated with TSP +5-Fu was higher than that treated with 5-Fu alone (Fig.4).
TSP (50 μg/mL), 5-Fu (25 μg/mL) and TSP (5 μg/mL) + 5-Fu induced apoptosis of HepG2 and L-02 cells. Values represent the mean±SD. Asterisk (*) sign indicates statistically significant values compared to (△) (P < 0.05); Plus (+) sign indicates statistically significant values compared to Asterisk (*) (P< 0.05).
Transmission electron microscope (TEM)
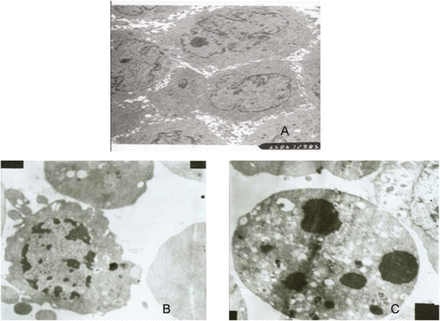
Hepatocarcinoma cell lines treated with TSP (50 μg/mL) showed changes in its morphology(Fig.5).
(A) Vehicle treated control HepG2 cells. (B) Cells nuclei present polymorphic or deformed shape with high plication in nucleus membranes, chromatin condensation, and nucleus fragments in various size. (C) The cell apoptosis body. (5000×)
Discussion
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors worldwide. As sufficient doses of chemotherapy drugs are limited due to poor liver function of HCC patients and as they have low sensitivity to anti-cancer agents, chemotherapy plays little role in the overall survival of the HCC patients. Novel agents which have greater targeting on HCC but lower toxicity for normal liver cells, are seen as a direction of enormous potential. Up to now, a number of agents extracted from the skin of various amphibians have been analyzed and found to have antitumor activity with little toxicity against non-malignant cells[9–11].
It is now well established that the reduced capacity of tumor cells undergoing cell death through apoptosis, plays a key role both in the pathogenesis of cancer and in therapeutic failure[12–16]. In the present study, the peptide extracted from the Chinese toad (Bufo bufo gargarizans) skin (TSP) showed strong inhibition to the proliferation and viability of the HepG2 cells. As shown in the results, TSP was a moderate inhibitor suppressing the proliferation of the L-02 cells in culture, and it could enhance the effects of 5-Fu. Analyses through flow cytometry and transmission electron microscope (TEM) showed that TSP could induce apoptosis of HepG2 cells. The antiproliferative effect of TSP can be attributed to the ability of TSP to hold the cell cycle at the G1 phase, and as a result, prevent further cell proliferation and initiate apoptotic signaling. The fact that TSP has two opposite effects on tumor cells and on normal cells is intriguing. The exact mechanism remains to be clarified. Bufalin, which is one of Chinese toad (B. b. gargarizans) skin extract preparation, may induce apoptosis in human cancer cells by up or down-regulating some genes expression[17–21]. It has been reported that some peptides isolated from amphibian skin show antitumor activity with little toxicity against nonmalignant cells, either by triggering apoptotic process[4,5,22,23] or by forming ion channels/pores[6]. In this study, The results from flow cytometric analysis, TEM and the outcome of the HepG2 cells arrested at G1, all demonstrate the apoptogenic nature of TSP, whereas the inhibition rate of cell growth, decreased absorbance of TSP-treated HepG2 show the antiproliferation and cytotoxic activity of TSP.
In recent years, combination therapy in cancer treatment or in cancer prevention has been widely recognized as an effective approach to maximize treatment effects[24,25]. The data from our study demonstrate that TSP acts synergistically with 5-Fu to inhibit the growth and induce apoptosis of HepG2 cells, suggesting that this strategy could be an effective way to destroy hepatocarcinoma cells. However, the mechanism of such enhancement remains unclear and the research on the characteristics of TSP is in progress.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received April 21, 2011.
- Accepted June 27, 2011.
- Copyright © 2011 by Tianjin Medical University Cancer Institute & Hospital and Springer