Abstract
OBJECTIVE To detect Mycoplasma hyorhinis in ovarian cancer tissues and the relationship between mycoplasma infection and ovarian cancer.
METHODS All specimens obtained from 109 cases with ovarian cancer were fixed in freshly prepared 10% neutral buffered formalin, embedded in paraffin, and cut into 4-μm sections for insitu hybridization (ISH) and then detected with immunohistochemistry (IHC). The expressions of 16S rRNA and P37 protein from mycoplasma hyorhinis were detected respectively using ISH and IHC. SPSS 13.0 software was employed to analyze the relationship between the results of the study and clinical pathological materials.
RESULTS The expression rate of mycoplasma hyorhinis 16S rRNA gene and P37 protein was 20.2% (22/109) and 43.1% (47/109 cases) in ovarian cancer tissues, respectively, but it was 0 (0/30 cases) in the normal ovarian tissues. The difference in mycoplasma infection ratio between ovarian cancer tissues and normal tissues was extremely significant (P < 0.001). Anyhow, we didn’t found any association between the mycoplasma infection and clinical pathological characters.
CONCLUSION There was a mycoplasma infection in ovarian cancer tissues, which may play a role in oncogenesis of ovarian cancer.
keywords
Introduction
M. hyorhinis was originally isolated from the nasal cavity of a pig. Under certain conditions, M. hyorhinis causes polyserositis and arthritis in swines[1]. Previous studies have shown that there is a possible association between mycoplasma infection and tumorigenesis[2,3]. To further validate that the infection of M. hyorhinis exists in the tumor tissue and to investigate a possible link between M. hyorhinis infection and tumorigenesis, M. hyorhinis 16S rRNA gene and P37 protein expression were detected in ovarian cancer using ISH and IHC, respectively. The association between the expressions of the genes detected and the clinical staging and prognosis of the patients were also discussed.
Materials and Methods
Ovarian carcinoma specimens and reagents
All the samples were taken from the patients treated in the Surgical Department of Beijing Cancer Hospital between 1997 and 2005. In the study, the specimens processed to paraffin blocks were cut into 4 μM sections. The patients’age ranged from 19 to 84 (with a median of 56 years). The stage of ovarian cancer was determined according to FIGO. Sixteen cases were classified as stage I, 10 cases as stage II, 57 as stage III, and 26 as stage IV based on pathological examination on the 109 ovarian tumor specimens. The histologic types of the tumors were classified according to WHO. Seventy-nine cases were diagnosed as serous papillary cystadenocarcinoma, 7 cases as endometrioid carcinoma, 8 cases as moderately and poorly differentiated adenocarcinoma, 2 cases as mucinous cystadenocarcinoma, 13 cases as other histologic types. The non-tumor ovarian specimens used as controls were taken from 30 cases who received surgery of total abdominal hysterectomy with bilateral salpingo-oophorectomy (TAH-BSO).
PD4, a mouse monoclonal antibody against p37 of M. hyorhinis, was generated and characterized in the laboratory[4]. Digoxigenin RNA Labeling Kit (SP6/T7) and DIG Nucleic Acid Detection Kit were purchased from Roche. EnVision was purchased from Dako. Diaminobenzidine (DAB) Staining Kit was from ZhongShan Biotechnology Company.
ISH
Digoxigenin-Labeled RNA probes were conducted following the procedures. M. hyorhinis 16S rRNA specific sequence (153-449 bp) was selected as probes and its specificity was analyzed by BLAST. The plasmids pGEM-16SrRNA, which were confirmed by sequencing, were linearized by NcoI or SalI digestion for about 2 h. After phenol/chloroform extraction, 1/10 volume of 3 mol/L NaAc and 2 volume of ethanol was added to the plasmids for 5 min, at room temperature (RT), followed by centrifugating at 12,000 rpm for 5 min, and additionally washing with 70% ethanol. Then the plasmids were dried and dissolved in 15 μL diethyl pyrocarbonate (DEPC) water, and the quality and quantity of the plasmids were tested by 1% agarose gel electrophoresis. A transcription kit (Roche) was used to transcribe digoxigenin-labeled RNA probes in both the sense and antisense direction. RNA probes were obtained by in vitro transcription with linearized DNA as a template in Sp6 or T7, and the RNA polymerase was catalyzed and then tested by 1% agarose gel electrophoresis. The probes were stored at -80°C.
ISH was conducted following the procedures. Briefly, the sections were deparaffinized with xylene, and sequentially rehydrated in a graded alcohol series. After being neutralized with 0.2 mol/L HCL for 30 min at RT, the slides were treated with 100 mmol/L proteinase K for 15 min at 37°C. The slides were then prehybridized at 42°C for 2 h and then incubated with hybridization solution containing freshly antisense digoxigenin-labeled RNA probes at 44°C overnight in a humidity chamber. After slides for 1 h with blocking solution, hybridized DIG probes were detected using sheep anti-DIG-alkaline phosphatase antibody (1:5000 dilution), at 37°C for 40 min. The slides were then washed twice with buffer 1 and then with buffer 2. The slides with NBT/BCIP solution were incubated in the dark. Sense probes were used as negative control. The blue-violet staining in the cytoplasm and the cytoplasmic membrane was evaluated. The results were evaluated by 2 investigators separately. The sign of (-) which means negative stain was observed in cells, and (+) which means positive stain was observed.
IHC
Tissues embedded in paraffin were cut into 4-μM slices with microtome. Sections were baked at 50°C-60°C overnight, and deparaffinized with xylene, followed by sequentially rehydrated in a graded alcohol series. The endogenous peroxidase activity in the tissues was blocked by immersing the sections in 3% H2O2 for 10 min. After the slides were blocked with mixture of 1% bovine serum albumin and 5% goat serum for 20 min, the slices were then incubated with PD4 (4 μg/mL) overnight, at 4°C in a humidity chamber. Unspecialized mouse IgG was used as a negative control. EnVision was used as a secondary antibody. For each step, the slides were washed twice for 5 min with phosphate-buffered saline. The slices were stained with DAB for 3-5 min and counterstained with hematoxylin. The stain in the cytoplasm and the cytoplasmic membrane was observed. The results were evaluated by 2 investigators separately. The sign of (-) which means no stained cells was observed, and the sign of (+) which means less than 50% of lightly positive stained cells were observed, and the sign of (++) which indicates over 50% of stained cells or strong stained cells were observed.
Statistical analysis
The data were analyzed using the χ2 test. A value of P < 0.05 was considered statistically significant. All statistical analyses were performed with SPSS 13.0 software.
Results
The results of probe labelling
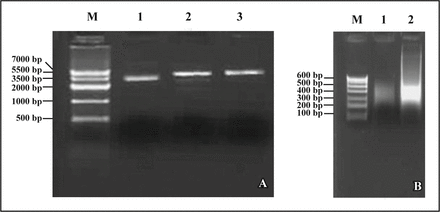
For prepared the DIG-labeled RNA probes, the recombinant plasmids were linearized by NcoI or SalI digestion. After being tested by 1% agarose gel electrophoresis (Fig. 1A), the recombinant plasmids were extracted by phenol/chloroform and finally resuspended in DEPC. Using linearized DNA templates, both sense- and antisense-oriented RNA probes were synthesized for in vitro transcription and then tested by 1% agarose gel electrophoresis (Fig. 1B).
Probe labeling. (A) 1, No digested; 2, Enzyme digested by NcoI; 3, Enzyme digestion by SalI; (B) Identification of probe by 1% agarose gel electrophoresis. 1, sense probe; 2, antisense probe.
ISH
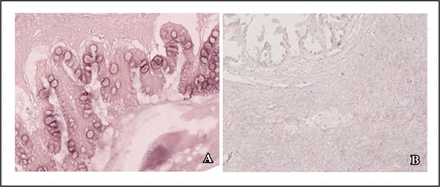
In the 109 specimens of ovarian cancer, 22 had mycoplasma infection coexisted. The positive rate was 20.2%. 16S rRNA gene expression mainly localized in the cytoplasm of tumor cells (Fig. 2).
Expression of M. hyorhinis 16S rRNA in ovarian cancer. A, Positive result; B, Negative control (BCIP-NBT staining, × 20).
Analysis revealed that there was no significant association between 16S rRNA gene expression and clinical pathologic parameters (e.g. age, menstrual history, grade of differentiation, clinical stage, histologic types, obstetrical history, degree of lymph node involvement, adjuvant therapy and prognosis) (P > 0.05).
IHC
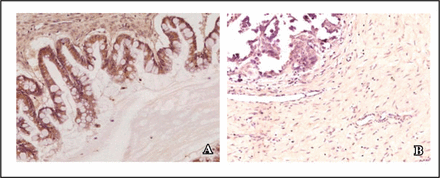
In the 109 specimens of ovarian cancer, 47 had mycoplasma infection. The positive rate was 43.1%. p37 expression was generally found in the cytoplasm of tumor cells, and was not found in the negative controls (Fig. 3).
Expression of P37 protein in ovarian cancer. A, Positive result; B, Negative control (DAB staining, × 20).
The statistical analysis for the result of IHC and the data of clinical pathology revealed that there was no significant association between IHC and the clinical pathology (P > 0.05).
comparing the correlation between ISH result and IHC result
In the 49 specimens of ovarian cancer, 47 were IHC positive; 48 were ISH and IHC negative; 39 were IHC positive with ISH negative; 14 were ISH positive with IHC negative (Table 1).
Comparison of the results of ISH and IHC in 109 specimens of ovarian cancer.
The correlation between the 2 methods (ISH and IHC) was compared with the independent fourfold table χ2 test. The results showed that the difference between the 2 was not significant (χ2 = 0.513, P = 0.474, r = -0.069). Comparison of the 2 results, difference between the 2 positive rates was significant (χ2 = 10.868, P < 0.001). The positive rate of IHC in ovarian cancer was much higher than that of ISH.
Discussion
The relationship between mycoplasma infection and tumorigenesis has generally been reported in foreign countries although the underlying mechanism remains elusive[5-8]. Mycoplasma infection has been detected in different types of tumor and some kinds of mycoplasma infection have been found to induce malignant transformation of mammalian cells[9,10]; to increase the expression of oncogenes; and to change the biological behaviors of tumor cells[11-13]. All these results suggest a possible link between mycoplasma infection and tumorigenesis. Previous work demonstrated the high rate of M. hyorhinis infection in cancer tissue by isolation and culture of mycoplasma, PCR and IHC, which suggests a possible association between mycoplasma infection and tumorigenesis[2,3].
p37 protein is the major immunogen and membrane lipoprotein of M. hyorhinis[11,12]. Exogenous p37 protein has been found to alter gene expression, morphology of cancer cells and to stimulate tumor metastasis in mice[14,15]. PD4, a specific monoclonal antibody directed against p37, inhibits the growth of MGC803 cells in nude mice by inducing apoptosis[16]. PD4 also inhibits the proliferation of transformed cells[17]. These results suggest that there are correlations between p37 and tumorigenesis.
Traditional detection of the mycoplasma under microscope was very difficult because of their small size. At present, there are many methods to be used, such as PCR, nested-PCR, PCR-ELISA, DAPI or Hoescht staining. Chan et al.[18] reported that mycoplasmas was detected in 59.3% of the malignant ovarian cancer specimens using sensitive PCR-ELISA. In this study, the expression of 16S rRNA gene and p37 in M. hyorhinis infection were examined respectively by ISH and by IHC using PD4 antibody in 109 ovarian carcinoma specimens. The results of ISH and IHC were matched to find the concordance rate and the significance of M. hyorhinis infection in ovarian cancer. Statistical analysis revealed that there were no correlation between the 2 methods and between the 2 results and clinicopathologic characteristics, Consistency of IHC and ISH is affected by the technological intrinsic factors and other factors, such as tissue fixation, degradation of cell membrane proteins and mRNA after being paraffin-embedded and long storage, variable sensitivity and specificity of antibodies and probes, differences in scoring criteria with interobserver variability in interpretation of results[19].
The present studies have demonstrated the presence of mycoplasma in ovarian cancer tissues through 2 independent methods: ISH and IHC. The studies have also analyzed the association between the results of the detections and the clinicopathologic data. There are a lot of work to do about the relationship between mycoplasma infection and tumor and the molecular mechanisms because of the complex type of mycoplasma. Once it is proved that mycoplasma infection is an important factor in tumorigenesis, it may significantly improve the way of the prevention and treatment of ovarian cancer.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Footnotes
This work was supported by a grant from the Nature Science Foundation of China (No.30130190).
- Received September 30, 2010.
- Accepted October 15, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer