Abstract
OBJECTIVE In this study, RNA interference was used to evaluate the effects of HMGB1 expression on cell cycle and proliferation of the human cervical cancer cell line HeLa.
METHODS We had previously constructed and screened effective eukaryotic expression vectors carrying PGCsi3.0-1/HMGB1 siRNA and PGCsi3.0-3/HMGB1 siRNA, then the vectors were transfected into HeLa cells. The expression of HMGB1 before and after transfection in HeLa cells were detected by RT-PCR and Western blot. The cell viability and proliferating activity was tested by Trypan blue dye test and MTT, and the cell cycle was determined by flow cytometry.
RESULTS The introduction of PGCsi3.0-1/HMGB1 siRNA and PGCsi3.0-3/HMGB1 siRNA inhibited the expression of HMGB1 mRNA and protein efficiently and specifically, there was a significant difference between the siRNA groups and the control groups (P < 0.05). The proliferation speed of PGCsi3.0-1 group and PGC si3.0-3 group were obviously slower than those of PGCsi3.0-Neg group and non-transfected group. Flow cytometry showed that the content of DNA in G2 phase in PGCsi3.0-1 group and PGCsi3.0-3 group were obviously more than those in PGCsi3.0-Neg group and non-transfected group, but the content in S phase was less (P < 0.01). The progression of cell cycle was arrested from G2 to S phase.
CONCLUSION PGCsi3.0-1/HMGB1 siRNA and PGCsi3.0-3/HMGB1 siRNA could specially suppress the expression of HMGB1 gene, inhibit the proliferation speed of HeLa cells effectively, and arrest the progression of cell cycle from G2 to S phase. RNAi provides a new approach to the bio-therapy of cervical cancer.
keywords
Introduction
HMGB1, a recently identified nuclear protein, has being founded to be closely associated with tumorigenesis, tumor neoangiogenesis as well as infiltration, metastasis, proliferation, apoptosis and drug resistance of tumor cells[1]. But its mechanism of action in cervical cancer is not well known, with the molecular mechanism becoming a focal point in present research of tumorigenesis and development[2,3]. Therefore, it is important to further study the function of HMGB1. We used RNA interference to investigate the effects of HMGB1on cell cycle and proliferation of human cervical cancer cell line HeLa.
Materials and Methods
Materials
Eukaryotic expression vectors carrying PGCsi3.0-1/HMGB1 siRNA and PGC si3.0-3/HMGB1 siRNA were obtained from Genechem (Shanghai). Lipofectamine 2000 was purchased from Invitrogen (USA) and RT-PCR kit from Takara (Japan). Monoclonal antihuman HMGB1 antibody was obtained from R&D systems (Germany).
Cell culture and transfection assay
Cells in this experiment were divided into 4 groups namely PGCsi3.0-1 group (cells transfected with PGCsi3.0-1/HMGB1 siRNA), PGCsi3.0-3 group (cells transfected with PGCsi3.0-3/HMGB1 siRNA), PGCsi3.0-Neg group (cells transfected with ineffective sequence siRNA), and the non-transfection group. All cells were cultivated in RPMI1640 medium supplemented with 10% fetal bovine serum and 5% antibiotics, and incubated at 37°C in a humidified atmosphere of 5% CO2, 24 h before transfection, cells were seeded at 2 × 105 per well of 6-well plate. The amounts of siRNA and transfection reagent were given according to the procedures provided by the manufacturer.
RT-PCR
Reverse transcription was performed by using total RNA from HeLa cells of the 4 groups and Reverse Transcription kit. The primers for HMGB1 were 5’-AAT AGG AAA AGG ATA TTG CT-3’(upstream) and 5’-GCG CTA GAA CCA ACT TAT GA-3’(downstream). The mixture of RNA and primers were loaded in the PCR amplifier with predenaturate setting at 95°C for 5 min, 94°C for 45 s, annealing at 50°C for 1 min and extension at 72°C for 1 min. The procedure were carried out 40 cycles, followed by a final extension at 72°C for 10 min. The PCR product then underwent 1.5% of agarose gel electrophotometer and quantitatively analyzed with Lab Works 4.5 analysis software.
Western blot
The same quantities of cells were collected from the 4 groups, and the protein of cells was extracted at the 24th h, 48th h, 72th h after transfection. The content of protein in the supernatant liquid was detected by the BCA method. Equal weight of protein was used for SDS-PAGE electrophoresis and transferred to PVDF membrane. The PVDF membrane was incubated overnight at 4°C with monoclonal anti-human HMGB1 (1:300), and then was incubated for 2h with the secondary antibody (1:5000). Finally after staining and fixing, the film was analyzed by the Image Analysis System.
Immunocytochemistry method
Cells in the logarithmic phase of the 4 groups were harvested to be inoculated on slides which were placed in 24-well plates. Till 90% confluence, the cells were paraformaldehyde fixed. After antigen retrieval and blockade with 1% serum, the primary antibody (1:100) was applied dropwise to the slides, then secondary antibody was added dropwise. Finally, after staining, counterstained and mounting, the slides were analyzed with a light microscope.
Trypan blue dye test used for observing cell viability
4 groups cells were seeded in 96-well plates (5 × 103/well) respectively, and then cells were counted at the 24th h, 48th h, 72th h, 96th h after transfection. There were 6 ambi-wells for each group, cells were counted 3 times, and the growth curve was drawn.
MTT used for observing cell proliferation
The cell suspension of the 4 groups was inoculated into 96-well plates at 1000 cells per well with 8 ambi-wells and kept for 24 h, 48 h, 72 h, 96 h after transfection, daily sampling was taken, and incubated with 20 μl MTT for 4 h. After centrifugation, 150 μl of DMSO was added to the precipitate and the absorbance(A) of the enzyme was measured at the 570-band. Cell growth rates (average absorbance A of each transfected group/non-transfected group) were calculated.
Flow cytometry for cell cycle
Cells of the 4 groups at the 24thh, 48thh, 72thh after transfection were fixed respectively with 75% alcohol overnight and washed 3 times with PBS, then mixed with RNase A and incubated at 37°C for 30 min. The mixture was stained with PI for 30 min. The number of cells was adjusted to 1 × 106/ml followed by flow cytometry analysis by using 200-mesh nylon nets in the procedure.
Statistical analysis
Statistical analysis was performed using SPSS13.0 statistical software. Data were presented as mean ± SEM. Statistical evaluation of the results was made by two-tailed Student’s test, F test and linear correlation. Significance was determined with 95% confidence.
Results
Transfection efficiency
After transfection at the 24th h, 48th h, 72th h, HeLa cells were observed with a fluorescence inverted microscope, and 5 visual fields were selected for observation and the number of cells calculated by visualizing green fluorescence. The transfection efficiency at different time point after transfection was computed respectively, the results were 15%, 75%, 80%, with the strongest brightness at the 48th h (Fig. 1).
HeLa cells at different time point after transfection. A, 24th h; B, 48 th h; C, 72nd h.
Analysis of the expression of HMGB1mRNA by RT-PCR
Fig. 2 shows the results of RT-PCR in the 3 groups. The expression of mRNA had no significant difference in the 3 groups which were transfected for 24 hours (P = 0.17). But at the 48th h, 72th h after transfection, the expression of mRNA in PGCsi3.0-1 group and PGCsi3.0-3 group was both significantly reduced compared to when that was in PGCsi3.0-Neg group (P = 0.039, P = 0.008), and the highest inhibition ratio of HMGB1 expression in PGCsi3.0-1 group and PGCsi3.0-3 group were both at the 48th h. The inhibition ratio in PGCsi3.0-1 group was higher than that in PGCsi3.0-3 group (P < 0.05).
The expression of HMGB1mRNA in each group cells at different time.
Western blotting analysis of the expression of HMGB1
Western blotting analysis indicated that the expression of HMGB1 protein in the 4 groups had no significant differences at the 24th h after transfection, but at the 48th h, 72th h after transfection, the expression of HMGB1 protein in PGCsi3.0-1 group and PGCsi3.0-3 group were both significantly reduced compared with those in PGCsi3.0-Neg group and non-transfection (P < 0.01). There was no significant difference between PGCsi3.0-Neg group and non-transfection (P > 0.05) (Table 1).
Data analysis of Western blot and immunocytochemistry.
Immunocytochemical method analysis of the expression of HMGB1
The positive stain was mostly located in the nucleus, along with a little cytoplasmic stain (special HMGB1 stained in brown). The rates of positive staining in PGCsi3.0-1 group and PGCsi3.0-3 group (21.60%, 26.00%) were both lower compared with non-transfection (94.00%) (P < 0.05). There was no significant difference (P > 0.05) in the expression of HMGB1 in the PGCsi3.0-Neg group and non-transfection group (Table 1).
Analysis of inhibition of cell growth
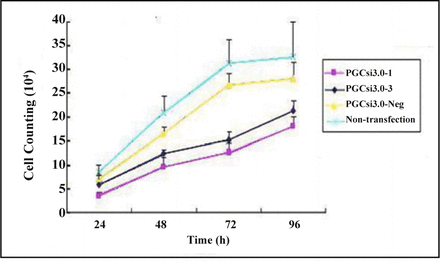
The trypan blue dye test showed that there were no significant differences in the number of cells in the 4 groups (P > 0.05) at the 24th h after transfection, but the number in the PGCsi3.0-1 group and PGCsi3.0-3 group were both less than those in PGCsi3.0-Neg group and non-transfection group at the 48th h, 72th h, 96th h after transfection (P < 0.05). There was no significant difference between PGCsi3.0-1 group and PGCsi3.0-3 group, as well as PGCsi3.0-Neg group and non-transfection at different time point (P > 0.05) (Fig. 3).
Growth curves of cells in different groups.
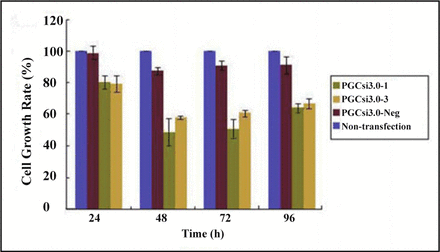
The colorimetric assay was used to get the absorbance (A) of MTT samples at different time point after transfection. Cell growth rates of the 4 groups were calculated, with the rates being the ordinate and time the abscissa (Fig. 4). The results showed that the cell growth of PGCsi3.0-1 group was significantly inhibited by RNAi compared with PGCsi3.0-Neg group at the 24thh, 48thh, 72thh, 96thh after transfection (P < 0.05, P < 0.01, P < 0.01, P < 0.05). The cell growth rates of PGCsi3.0-3 group at the 48th h, 72th h, 96th h were much lower than those of PGCsi3.0-Neg group (P < 0.01, P < 0.01, P < 0.05). The growth inhibition rate of PGCsi3.0-1 group was higher than that of PGCsi3.0-3 group, but the difference was not significant (P > 0.05).
The rate of HeLa cell growth after HMGB1 siRNA transfection at different time point.
The effect of RNAi on cell cycle
Flow cytometry analysis indicated that the cells in PGCsi3.0-1 group and PGCsi3.0-3 group exhibited arrested cell growth at stage G2. The content of DNA at stage G2 in PGCsi3.0-1 group and PGCsi3.0-3 group were both much more than those in PGCsi3.0-Neg group and non-transfection at the 24th h, 48th h,72th h after transfection (P < 0.05), but the content of DNA at stage S were less than those in PGCsi3.0-Neg group and non-transfection (P < 0.01). At the 24th h, 48th h, 72th h after transfection, there were no significant difference between PGCsi3.0-1 group and PGCsi3.0-3 group at stage G2 and S (P > 0.05), and the difference between PGCsi3.0-Neg group and non-transfection was also insignificant (P > 0.05) (Table 2).
DNA content at the different stage of cell cycle in 4 groups cells.
Discussion
Cervical cancer is one of the most common gynecologic malignancies, whose incidence and mortality is second only to breast cancer[4]. The generation of cervical cancer is a complex process which multi-steps and multigenes are involved in[5]. In recent years, an increasing number of oncogenes related to the incidence, invasion, and metastasis of cervical cancer have been discovered, such as P53, bcl-2, survin, HPV E6 and E7[6,7]. HMGB1 was discovered as a multifunctional cytokine involved in the growth, invasion, metastasis, clinical stage and prognosis of pancreatic cancer[8], liver cancer, gastric cancer, melanoma, colon cancer[9], breast cancer, bile duct cancer, and prostate cancer. Our early experiments demonstrated that HMGB1 protein in cervical squamous cell carcinoma (CSCC) especially in metastatic tissues was a strongly positive expression. Therefore, HMGB1can become a new target for biological therapy for cervical cancer[10,11].
As a technology for blocking gene expression efficiently and specifically, RNAi forms a new antitumor therapy which has been applied to a variety of human tumors such as breast cancer, ovarian cancer, prostate cancer, pancreatic cancer, sarcoma, etc.[12] Many researches[13-17] have shown that HMGB1siRNA could significantly inhibit the expression of HMGB1 as well as the proliferation of cancer cells in vitro. The cells were arrested in G1 phase and the percentage of apoptotic cells was increased. In the previous experiments we had confirmed that HMGB1 was highly expressed in CSCC. In the FIGO stage I, II period of CSCC the positive expression rates were 53.9%, 79.5%, and the difference was statistically significant (P < 0.01). The positive expression rate was 55.7% in CSCC tissues whose diameter was < 4 cm, while the rate was 72.6% with those having a diameter ≥ 4 cm. It was demonstrated that high expression of HMGB1 accelerated the CSCC cells proliferate. In our study, we found that the expression of HMGB1 was reduced effectively by the HMGB1 eukaryotic expression vectors being transfected into HeLa cells, cell growth and proliferation were also inhibited, and the cell cycle was arrested in the G2 phase. Possible mechanisms are as follows: Firstly, with the mRNA-competitive effects of the translation process, HMGB1siRNA could inhibit the expression of HMGB1 gene, thereby affecting cell proliferation-related signal transduction pathways, such as downstream MARK, JNK, etc., leading to slowed cell growth[18-20]. Secondly, numerous studies confirmed that the regulation of cells was a strict, orderly, and complex network activity, and any imbalance in the link in cyclin could lead to tumor formation. Cyclin D1 played a role in the conversion from G0/G1 to S phase, while cyclin A was the key to the midterm cell division and the conversion from G2 to S phase[21,22]. It is inferred that HMGB1siRNA might reduce the expression of cyclin A, thereby blocking the transition process from G2/M phase to S phase, and leading to the cell division and proliferation inhibition. Thirdly, HMGB1 might inhibit the activity of the tumor suppressor gene P53, thus preventing cells from repairing DNA damage and activating the signal transduction pathway Ras/ERK excessively[15].
In this experiment, we used the vector-mediated RNA interference method and found in RNAi the common shortcomings. Firstly, the functional efficiency of Dice enzyme may be different in different cell lines, leading to the same siRNA, but different interference efficiency. While it was reported that the transfection efficiencies of HMGB1siRNA plasmid were both more than 90% in pancreatic cancer and liver cancer, in this experiment, it was found to be slightly lower at 70%-80%. The cell proliferation and cell cycle had no significant changes at the 24th h after transfection, and the cell proliferation inhibition at the 48th h was higher than that at the 72th h. The results demonstrated that the interference efficiency of HMGB1siRNA in HeLa declined gradually with time for transfection. The possible reason was that the transient transfection efficiency decreased with time compared with stable transfection[23]. The proliferation of cells decreased due to the gradual increase in cell load, and the transfected cells were easily removed by the body. But the non-transfected cells proliferated rapidly, resulting in the weakened inhibition of siRNA acting on the target gene. Secondly, the gene-specific sequence of different target genes in the same cell was different, and the interference efficiency was also different[24]. The sites on the secondary structure of target mRNA could be identified and combined by siRNA, and those sites affected the interference efficiency of siRNA. Software was used to analyze the functional sites of HMGB1siRNA and 4 siRNA sequences were designed. In preliminary experiments, we had identified that the sequences HMGB1 siRNA-1and HMGB1 siRNA-3 had a certain interference effects. Our results showed that these two sequences had different effects on the proliferation rate and cell cycle of HeLa cells, the sequence HMGB1 siRNA-1 was a more specific interference sequence of HMGB1mRNA.
In summary, this study showed that HMGB1 was a contributing factor in the occurrence and development of cervical cancer, and it was involved directly or indirectly in the regulation of cell proliferation and in the cell cycle of cervical cancer. The application of RNAi technology in this research could reduce the expression of HMGB1 gene in cervical cancer cell line HeLa, thereby affecting the growth characteristics of cervical cancer cells. Thus, as a novel means of cancer gene therapy, RNAi technology provides a new approach to cervical cancer gene therapy.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received March 31, 2010.
- Accepted April 18, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer