Abstract
OBJECTIVE To summarize the clinical characteristics and treatment effects of Burkitt lymphoma (BL) and Burkitt-like lymphoma (BLL), and to explore the best possible optimal regimens and the treatment-related complications.
METHODS Clinical data of 13 BL and BLL patients, who were pathologically diagnosed and treated in the Beij ing Cancer Hospital from August 1996 to October 2008, were retrospectively analyzed. All patients received the therapeutic regimen with chemotherapy as the first-line treatment. The treatment effect and adverse reactions were evaluated.
RESULTS Of the 13 patients, 12 were men and 1 was woman, with a median age of 15 years (range, 11-62). Three of the patients were in stage I, 2 in stage II, 2 in stage III, and 6 in stage IV. Advanced cases were 8 (stage III and IV), accounting for 61.5% of the patients. Bone marrow involvement was found in 2 cases (15.4%) and central nervous system invasion in 4 (30.8%) at the beginning of the treatment. The common involved sites included the superficial lymph nodes (61.5%), abdominal organs (53.8%), and abdominal and retro-peritoneal lymph nodes (38.5%). B symptoms were observed in 7 patients (53.8%). Serum lactate dehydrogenase (LDH) level was increased in 8 of the 10 patients who underwent the serum LDH determination, while serum uric acid level was increased in 1 of the 10. Pathological diagnosis showed that 11 of the cases were BL and 2 BLL. Of the 13 patients, 11 (84.6%) achieved a complete remission (CR) or CR/unconfirmed (CRu), and 1 (7.7%) a partial remission (PR), with a total responsive rate of 92.3%. At a median follow-up of 8 months (range 5-35), 6 patients were found dead at the end of the follow-up. One of the 13 patients was lost to the follow-up. The 1-year overall survival, progression-free survival, and disease-free survival rates were 56.98%, 32.31% and 39.77%, respectively. The grade-III or IV myelosuppression was found in 9 patients during the chemotherapy (69.2%), and tumor lysis syndrome and grade-IV total gastrointestinal mucositis were seen in 1.
CONCLUSION Intensive short-course chemotherapy is recommended as the optimal first-line treatment for BL and BLL, and actively preventive treatments for chemotherapy-related adverse reactions are essential in the treatment course.
keywords
Introduction
Burkitt lymphoma (BL) and Burkitt-like lymphoma (BLL) are highly invasive B-cell non-Hodgkin’s lymphomas (NHL), with a rapid progression of the disease and a high death rate. BL and BLL is usually found in children and adolescents, accounting for about 35%-40% of the incidence rate of children NHL[1], but rare in adults. It has been reported in west Europe and in the United States that the disease approximately covers a proportion of 1%-2% of adult lymphoma cases[2]. For the past few years, the prognosis of the children with BL or with BLL has been significantly improved, and the 2-year disease free survival rate of BL or BLL patients in advanced stage has attained 75%-89%, however, the prognosis of the adult patients hasn’t been preferable[2]. In our study, the data of clinical features, therapeutic effects, and adverse reactions collected from 13 patients with BL or BLL treated in our hospital were retrospectively analyzed.
Materials and Methods
Source of cases
The 13 BL or BLL patients selected in this study were pathologically diagnosed and treated in Beijing Cancer Hospital during a period from August 1996 to October 2008.
Clinical data
The general data of the 13 patients were as follows: the age of the patients when diagnosed ranged from 11 to 62 years, with the median age of 15, and 6 of them were at the age below 14 (46.2%), and the other 7 over 14 years (53.8%). Of the 13 patients, 12 were male and 1 was female. The score of the 11 cases based on ECOG scoring ranged from 0 to 1 (84.6 %), and that of the remaining 2 was 2 (15.4%).
Pathologic diagnostic criteria
The patients were diagnosed as BL or BLL based on the morphologic and immunohistochemical examinations according to 2001 WHO classification of the lymphoid neoplasms. Typically pathological changes in the patients included diffuse infiltration of medium-sized B-cell (12 μm) with the round or oval nucleus and without cleaves or folds. The cell nucleus in division status and “starry sky” phenomenon were commonly seen. In the immunohistochemical assay, the B-cell marker (CD20, CD79a), CD10, and Bcl-6 were positive, and the index of nuclear proliferation was high (Ki-67)[3].
Staging
The Ann Arbor staging was used for classifying NHL. An overall check for staging was conducted in the patients, including the hematologic tests, ESR, LDH, β2-micro-globulin (β2-MG), x-ray on chest, ultrasonic inspection, CT, bone marrow aspiration, and laboratory test of cerebrospinal fluid.
Diagnostic criteria of central nervous system (CNS) invasion
The diagnostic criteria of the CNS invasion indicated that tumor cells of any amount presented in cerebrospinal fluid, or the space-occupying lesion presented in CNS. The diagnostic criteria of the bone marrow involvement was that the abnormal immature lymphocytes were over 5% in each of the bone marrow slide.
Treatment
Chemotherapy was used as the first-line therapeutic regimen in all the cases. The CHOP regimen or a regimen similar to CHOP, i.e. the CHOP like regimen was employed during a period from 1996 to 2004, and the ProMACE/CytaBOM protocol was used from 2005 to 2006. With the support of autologous peripheral blood stem-cell transplantation, the high-dose chemotherapy was administered to the patients who had achieved CR for consolidation. From 2007 to 2008, the Hyper-CVAD protocol was used for the patients, but the chemotherapeutic CHOPE regimen was still used in only 2 of the patients owing to their lack of money for medical expenses and old age. During the treatment, a preventive intrathecal chemotherapy was conducted. At the beginning of the treatment, other preventive treatments were given, such as hydration, alkaline alkalization, diuresis, reducing the uric acid, and applying prephase chemotherapy, etc., to prevent the tumor lysis syndrome (TLS).
Evaluation of therapeutic effect and adverse reactions
The therapeutic effect on lymphoma was evaluated based on the Cheson criteria[4,5], including the complete remission (CR), unconfirmed CR (CR/unconfirmed, CRu), partial remission (PR), and progression of disease (PD). The adverse reactions were evaluated based on the related WHO standards.
The overall survival (OS) time was calculated from the date when the diagnosis was firstly made to the day of death owing to any reason or to the date of the last follow-up. The disease-free survival (DFS) time indicated the interval time from the day when CR was confirmed to the day of tumor relapse, or to the day of death or the last follow-up. The progression-free survival (PFS) time was computed from the date of diagnosis to the day of first discovery of the tumor progression (TP), or to the day of dead of tumor or the last follow-up. The survival time of the patients lost to the follow-up was calculated based on the date of the last follow-up.
Statistical analysis
The SPSS 13.0 was used for statistical analysis and the Kaplan-Meier curve for computing the survival rate of the patients.
Results
Clinical manifestations and the results of laboratory examinations (Table 1)
Clinical features of the 13 patients with BL and BLL.
Chief complaints
Abdominal discomfort and neoplasm presented in 5 of the 13 cases, swelling superficial lymph nodes (SFLN) in 5, and the space-occupying lesions in the mediastinum and lung were accidentally found by coughing in 2, and intra-spinal space-occupying lesion identified by pain in the lower limbs in 1.
Involvement sites
The sites of the involvement were, based on the sequence of the occurrence frequency, the SFLN, including the nodes in sub-mandibular part, neck, supraclavicular part, armpit and iliac region, in 8 cases (61.5%), celiac organs including the intestine, kidney, adrenal gland, liver, spleen, pancreas, and stomach, etc., in 7 (53.8%), celiac and retroperitoneal lymph node in 5 (38.5%), mediastinal lymph node in 5 (38.5%), pleura and pericardium in 4 (30.8%), pelvic nodes in 3 (23.1%), oral pharynx, nasopharynx, paranasal sinus and maxillofacial region in 3 (23.1%), osseous involvement in 2 (15.4%), invasions to breast, lung, testis and skin in 4 cases. Bone marrow involvement occurred in 4 (30.8%), and CNS invasion in 5 (38.5%).
B symptoms
B symptoms were found in 7 cases (53.8%), among which fever was in 5 (38.5%), night sweat in 3 (23.1%), and body weight loss in 1 (7.7%).
Ann Arbor staging
In the 13 cases, 3 were at stage-I, 2 at stage-II, 2 at stage-III, and 6 at stage-IV, and among the 6 cases, bone marrow invasion was seen in 2, and CNS invasion in 4 at the beginning of the treatment.
LDH, uric acid, ESR and β2-MG determinations
Determination of serum LDH was conducted in 10 (range of normal values: 110-240 IU/L), with a median of 306.5 IU/L. Among the 10 patients, increased level of LDH occurred in 8, including 5 cases at the level around 240-500 IU/L (50%), 2 around 500-1000 IU/L (20%), and 1 at over 1000 IU/L. The maximum of LDH in the 10 cases was 1844 IU/L (10%). Ten of the 13 patients received the serum uric acid test, and the test result from 1 of them was higher than normal. The ESR test was conducted in 9 of the patients, and the result higher than normal was seen in 4, among which 2 attained more than 50 mm/h. Of the 7 patients received serum β2-MG test, and the test result higher than normal was in 6.
IPI scoring
The IPI score of 0-1 was achieved in 5 of the patients, the score of 2 in 2, the score of 3 in 4, and the score of 4-5 score in 2.
Pathologic types
The final diagnosis was made in all 13 cases after the pathological histomorphology and immunohistochemical examinations. Based on 2001 WHO classification of the lymphoid neoplasms, 11 cases were diagnosed as BL, and the other 2 were diagnosed as BLL. All the B-cell markers were positive. The result of CD10 detection was positive in 9 cases, and that of Bcl-6 detection was positive in 4 of the 7 cases, and the result was positive in 1 of the 5 with Bcl-2 detection. The result of TdT test was negative in 9 cases. The high expression rate attained > 90% in 8 of the 9 cases receiving Ki-67 detection and 80% in the other 1 case. One of the 2 cases received EBV detection (EBER), the outcome was positive. The c-Myc translocation, i.e. t (8; 14) was seen in 1 case in chromosome examination.
Treatment effect and survivals
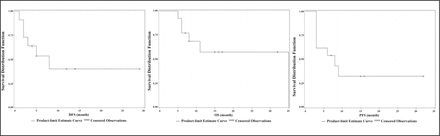
The deadline of the follow-up was April, 2009 for all cases in our group, with the median follow-up time of 8 months (range: 5-35 months). One case was lost to the follow-up 17 months after the follow-up started. Chemotherapy was the first-line in treatment for all cases. CR reached in 7 cases (53.8%), CRu in 4 (30.8%), and PR in 1 (7.7%), PD in 1 (7.7%), with a total responsive rate of 92.3%. Of the 7 patients who achieved CR, 5 reached a continued complete remission and have still survived now; 1 suffered CNS and bone-marrow invasion 8 months after reached CR and was lost to the follow-up17 months after CR; 1 died of PD 3 months after reaching the CR. Of the 4 patients with CRu, 3 died of PD, and 1 has still survived now. One patient suffered PD 3 months after reaching PR, and then died even a salvage chemotherapy was applied. One patient who had developed PD died. By the end of the follow-up, 6 of the patients died of PD. The 1-year OS rate 1-year PFS and 1-year DFS were respectively 56.98%, 32.31%, and 39.77% in all patients in this group (Fig. 1).
Survival curves of 13 BL and BLL patients.
Adverse reactions
Evaluation of the adverse reactions was conducted based on the WHO standards. The grade II/III myelosuppression was found in 9 of the patients (69.2%). All the 9 patients received the supportive treatment, such as granulocyte stimulating factor or/and the infusion of red cell suspension, and platelet, etc., and among them, 6 occurred concomitant infections at various degrees and at different sites. Grade-IV thrombocytopenia inducing epistaxis was found in 2 patients and were controlled by nasal packing and by platelet infusion. Over amount of pleural fluid was seen 1 of the other 7 patients. The TLS occurred after a high-dose of MTX (methotrexate) chemotherapy, resulting in the acute renal failure. Although the salvage treatments of hydration, alkalization, diuresis, and intensified calcium leucovorin were applied, which won a recovery of renal function, the patient still died of the grade-IV mucositis, low digestive tract hemorrhage, and TP.
Discussion
BL and BLL are the highly invasive lymphomas, with a low incidence rate. The 13 patients in our group accounted for 0.92% of the NHL patients concurrently admitted to our hospital, which is similar to the incidence rates reported overseas. At admittance, 61.5% of the 13 patients were at stage-III/IV, with a median age of 15 years and a majority in the males. The common site of tumor was SFLN and abdomen, and jaw bone involvement was seen in only 1 of the patients. Previous reports had it that the incidence of bone marrow or CNS involvements in adults was in a range of 30%-38% and 13%-17%, respectively[2]. Recent reports have shown that the rate of the involvements of bone marrow or CNS has somewhat increased, and at diagnosis, the involvement of the marrows or meninges has reached up to 70% and 40%, respectively[3]. In the patients of our group, the incidence rate of the bone marrow or CNS invasions was 30.8% (4/12) and 38.5% (5/12), respectively.
The malignancy degree of BL and BLL are extremely high, with the advanced stages at the diagnosis. The treatment effect on the invasive lymphoma is less preferable based on the traditional views and experiences. At overseas, the high-intensity and short-course therapeutic strategy with the multi-drug combination and preventive treatment for CNS invasion have been employed, with the 5-year event-free survivals (EFS) of the children with BL up to 90%[6-12]. After the revision, several therapeutic regimens for children with BL or BLL have been used for the adult patients, including the LMB and NHL-BFM series, and the CODOX-M/IVAC protocol etc. The treatment effects for these patients have been significantly improved after using the new regimens, and the 2-year EFS has at least reached up to 50%-73.2%[13-20]. Other common regimens include the Stanford regimen, Hyper-CVAD, CALGB 9251 protocols, etc., in which the 3-year OS may achieve a range from 49% to 54%[21-23]. The chemotherapeutic intensity in these protocols is high, and its regimen has a short intermission and conspicuous adverse reactions. The concomitant grade-III/IV myelosuppression has been often seen, and chemotherapy-related death even occurs. Because the incidence rate of BL is low and it tends to occur in children and adolescents, quantities of such cases have only been seen in a few cancer centers over the last 10 years in China. The median age of the patients is below 10[24-27], and the 3-year EFS is over 80%. Reports on the adult BL has been less. Lin et al.[25] employed the CODOX-M/IVAC protocol to treat 11 adult BL patients, and the outcome of the treatment achieved 80% of CR and 75% of 2-year EFS. A total of 13 BL or BLL patients were treated in our center during the period of 12 years, and the CHOP and CHOP-lilke regimens were mainly used in chemotherapy owing to an insufficient knowledge about the disease in the past years. Since 2006, the chemotherapeutic Hyper-CVAD protocol started to be used for the patients in our hospital. Although the CR + CRu rate has reached 84.6% in all the patients, with a total responsive rate of 92.3%, the difference still remains in the long-term survivals compared with that reported overseas. The 1-year OS, PFS, and DFS rates were respectively 56.98%, 32.31%, and 39.77% in our cases. Relapse and PD were commonly seen in the patients within a year after CR, with an unfavorable efficacy of the salvage chemotherapy.
After analysis, we figure out possible reasons as follows: i) it correlates to the CHOP and CHOP-lilke regimens that were employed owing to insufficient knowledge about the disease in the past years; ii) though the Hyper-CVAD protocol was used in the 5 patients who were admitted to our hospital over the past 2 years, the dose of Cytarabine was reduced to 1 g/m2 owing to an anxiety induced by worsened myelosuppression, based on part B of the protocol. Nevertheless, grade-IV myelosuppression still occurred in all 5 patients. The severe myelosuppression usually results in infection, therefore, chemotherapy was postponed, and may in some degrees lead to a proliferation of the drug-resistant tumor-cell lines. It suggests that the chemotherapy of sufficient intensity under an active supportive treatment should be guaranteed in the BL and BLL therapy, and at the same time, the intermission of treatment be shortened. Based on the references of the successful experience in treatment for children with BL in other centers, we are now trying to employ LMB89 protocol as the first-line therapy in the treatment for adult BL or BLL patients.
BL and BLL are frequently complicated with TLS, owing to the short time for tumor cells doubling within 25 h[2], and to the sensitivity to the cyto-toxic drugs and high tumor load in the patients. In our study, TLS occurred in 1 patient with over amount of pleural fluid during the second-line chemotherapy using the LMB89 (COPADM2) protocol, resulting in the acute renal failure. Then after active treatments, such as hydration, alkalization, diuresis, etc. for the patients, the renal function recovered. However, the complication with total gastrointestinal mucositis occurred in the patient, with a massive hemorrhage of lower gastrointestinal tract. Finally, the patient died of TP on account of the severe complication that delayed the chemotherapy. Lin et al.[25] reported that 2 BL patients who died of TLS also suffered the complication of over amount of thoracico-abdominal fluid (TAF). Analysis of the reasons shows that it may be caused by the accumulation of MTX in the TAF[28]. Therefore, TLS should be cautiously prevented and treated in the centers with more clinical experience. Concerning the patients suffering profuse TAF, if the tumor load is very heavy, and the serum LDH and uric acid are obviously increased, a drainage of TAF before administration of large dose MTX should be done as needed, in order to reduce the possible accumulation of MTX.
The main cause of death of the patients in our group was TP. Availability of other therapeutic measures should be discussed concerning the patients who suffered TP or failed to achieve CR after the first-line intensive chemotherapy, or those with relapse within a short period after CR. Ladenstein et al.[29] found that the 5-year EFS rates respectively could reach 56.6% and 48.7% in the patients with PR and a sensitive relapse who underwent the initial treatment following the autologous hemopoietic stem-cell transplantation, while those with primary refractory disease and resistant replase after the autologous transplantation all died within 1 year. Thomas et al.[30] reported that the 3-year OS, EFS and DFS rates were respectively 89%, 80%, and 88% in the adult BL patients treated with Rituximab combined with Hyper-CVAD protocol, and the 3-year DFS rate also achieved 88% in 9 patients at the age of over 60, which was obviously 30% higher than that in the patients applied with simple Hyper-CVAD protocol, suggesting that the addition of Rituximab can improve the treatment effect, especially in the aged patients.
It indicates in our study that the CHOP and CHOP-lilke regimens are not the best way to treat BL and BLL, and the intensive short-course chemotherapeutic regimen is recommended, which can ensure the sufficient dose. The active prevention and treatment for TLS and the treatment for adverse reaction induced by chemotherapy are needed to guarantee the uninterrupted and timely chemotherapy. For the patients with unfavorable prognosis, the best possible regimen of salvage chemotherapy should be further investigated.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received February 24, 2010.
- Accepted April 12, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer