Abstract
OBJECTIVE To investigate the expression of the high mobility group box1(HMGB1) in human cervical squamous epithelial carcinoma (CSEC) and to explore the relationship of HMGB1 expression to the differentiation degree, size, invasion and metastasis of CSEC.
METHODS Immunohistochemical staining of tissue microarrays and Western blot analysis were conducted to detect the expression of HMGB1 in the following tissue samples: 30 carcinoma in situ, 90 invasive CSEC without metastasis, 30 invasive CSEC with metastasis, 30 cases of normal cervical squamous epithelia.
RESULTS The positive-expression rate of HMGB1 was 58.7% (88/150) in CSEC, showing a significant difference compared to normal cervical squamous epithelia. The expression of HMGB1 was correlated with tumor size, invasion and metastasis of CSEC (respectively, P<0.01), but had no relationship with the degree of differentiation (P>0.05).
CONCLUSION The over-expression of HMGB1 in CSEC might be a useful parameter as an indication of tumor invasion, metastasis, prognosis and overall biological behavior of human CSEC, as well as a noval target site for gene therapy.
keywords
- cervical squamous epithelium carcinoma (CSEC)
- high mobility group box1 HMGB1
- immunohistochemistry
- Western blot
- tumor invasion
Introduction
With about 400,000 new cases, and nearly 250,000 deaths each year, cervical cancer contributes significantly to worldwide cancer-related morbidity and mortality. In China, CSEC is the most common gynecological malignant tumor. As with most tumors, the occurrence and development of CSEC is regulated by multiple gene alteration. HMGB1 is a small, highly conserved nonhistone DNA-binding protein. HMGB1 expression is correlated with tumor growth, invasion and metastasis, and is over expressed in many tumors. In this study, we investigated the expression of HMGB1 in CSEC and explored its relationship with the CSEC degree of differentiation, size, invasion and metastasis.
Materials and Methods
Clinical data
Tissue sections for immunohistochemistry were comprised of 150 CSEC tissue specimens (30 carcinoma in situ, 90 invasive CSEC without metastasis, 30 invasive CSEC with metastasis) and 30 normal cervical squamous epithelia. The specimens were obtained from routinely processed paraffin-embedded tissue samples resected at the Tianjin Medical University Cancer Institute & Hospital. The criteria for inclusion in the study were that the paraffin blocks were suitable for immunohistochemistry and there was adequate associated clinical information. The ages of the patients ranged from 35 to 64, with a median age of 50. All of the patients received no pre-operation therapy. For Western blots, the tumor tissues (30 invasive CSEC, 20 CSEC, 20 normal cervical squamous epithelia) were obtained immediately after surgical excision and were stored at -80°C before use. Tissues for both immunohistochemistry and Western blots were obtained from hysterectomy specimens. Institutional ethical approval and informed consent was given by the patients from whom the clinical samples were obtained.
Immunohistochemical analysis
Formalin-fixed and paraffin-embedded tissues were used to make tissue microarrays for HMGB1 immunonstaining. Deparaffinization and rehydration were performed using xylene and alcohol. The sections were treated with 0.3% hydrogen peroxidase for 15 min, and with blocking antibody for 30 min. The antibody against HMGB1 (monoclonal antihuman HMG-1 antibody R&D, Germany) was at 1:100 (v/v). An avidin-biotin complex methodology was used with diaminobenzidine as the chromogen and hematoxylin for counterstaining. The expression of HMGB1 was categorized as over-expressed when there was definite nuclear staining in more than 30% of the tumor cells and/or cytoplasmic expression, and as normal with definite nuclear staining in less than 30% of the tumor cells.
Western blot analysis
All tissues used were lysed using Tris buffer (pH 8.0; 50 mM Tris, 150 mM NaCl, and 5 mM EDTA with protease inhibitor cocktail; Roche Diagnostics, Mannheim, Germany). To perform Western blot analysis for HMGB1, 15 μg of protein from each sample was separated by 12% sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis. The bands were then electrically transferred onto PVDF membranes using Semi-Dry TransBlot Cell (Bio-Rad Laboratories, Hercules, CA). Membranes were soaked in 5% nonfat dry milk in TBST (20 mM Tris base, 500 mM NaCl, and 0.05% Tween 20, pH 7.5) for 1 h at 37°C and incubated overnight at 4°C with monoclonal anti-human HMG-1 antibody (1:300) and with anti-β-actin antibody 1:5000 (Abcom, UK). They were washed thrice with TBST and incubated for 1 h with horseradish peroxidase-labeled anti-rabbit secondary antibody (Pierce Biotechnology, Rockford, IL) diluted in TBST (1:1000) at room temperature. The membranes were then washed and developed with chemiluminescent substrates (Pierce Biotechnology).
Statistical analysis
All experimental data were entered into the data library of the SPSS 13.0, χ2 test, One-way ANOVA was used to analyze the associations between different variables. Significance was accepted at a value of P<0.05.
Results
Immunohistochemical analysis of HMGB1 expression in CSEC
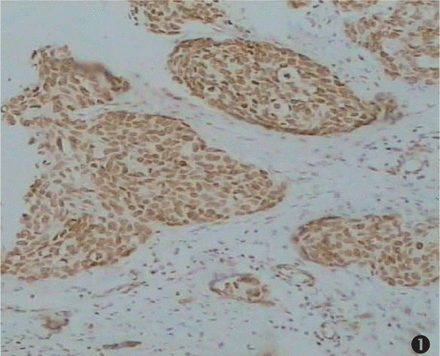
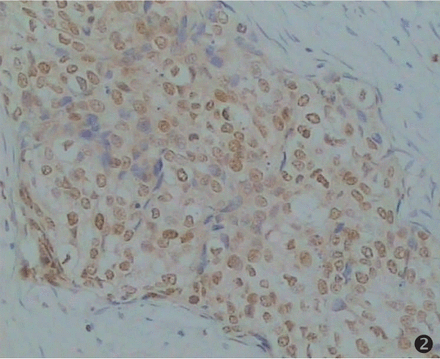
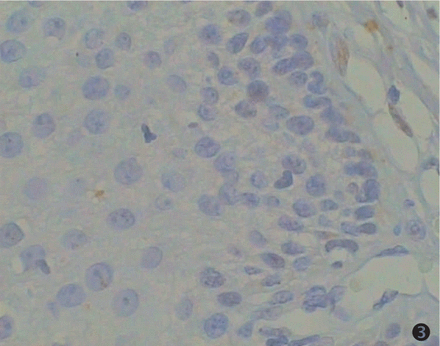
An immunohistochemical technique was chosen for HMGB1 protein analysis because it allows a rapid and sensitive screening of CSEC pathological tissues, and is amenable to regular use as a routine diagnostic test. We analyzed tissue specimens from 150 CSEC cases, constituted by 30 carcinomas in situ, 33 cases with a pathological grade I; 47 cases with pathological grade II; 40 cases with pathological grade III, 30 cases of invasive CSEC with metastasis and 30 normal cervical squamous epithelial tissues. The results of the immunohistochemical study of the CSEC specimens are summarized in Table 1. We found that most of the CSEC samples showed a strong HMGB1 nuclear immunoreactivity in the majority of the cells, along with a little cytoplasmic staining (special HMGB1 staining in brown, counterstaining in blue). Twenty-five of 30 (83.3%) invasive CSEC with metastasis displayed over-expressed HMGB1 levels, showing extremely strong nuclear expression (Fig.1). The rates of positive staining in pathological grades I, II and III cases were respectively 54.6%, 61.7%, 67.5% (P>0.05, Fig.2). Most of cells from normal cervical squamous epithelia showed no or faint staining (16.7%, Fig.3).
HMGB1 expression in CSEC and its relationship with clinical pathological characters.
Expression of HMGB1 in invasive CSEC with metastasis. Nearly all the tumor cells were stained. Strong nuclear positive staining was detected (×100).
Expression of HMGB1 in pathological grade II of CSEC. Special HMGB1 staining in brown, counterstaining in blue (×200).
Expression of HMGB1 in normal cervical squamous epithelium. Little immunostaining was observated in normal cells (×400).
HMGB1 overexpression in CSEC samples
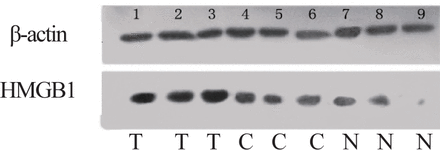
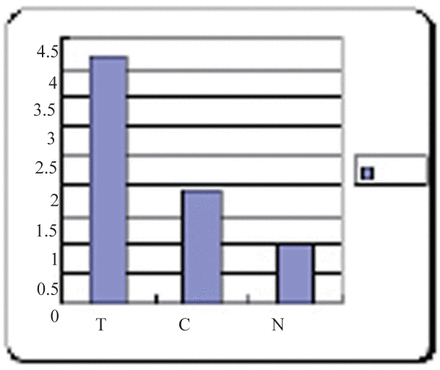
In a small number of samples (30 invasive CSEC, 20 CSEC, 20 normal cervical squamous epithelia), HMGB1 protein expression was also evaluated by Western blot analysis that essentially confirmed the immunohistochemical data. As shown in the Fig. 4, a HMGB1 specific single band with apparent molecular weight of 30 kDA was present. The Western blot analysis illustrated that the expression of HMGB1 in invasive CSEC with metastasis was more than two times higher than in CSEC without metastasis, and about four times higher than the expression in normal cervical squamous epithelia (P<0.01, Fig.5, Table 2).
T: invasive CSEC with metastasis C: CSEC N: normal cervical squamous epithelia. Western blot analysis indicated that the T samples displayed the highest levels of HMGB1 expression (lanes 1, 2, 3), C samples were lower (lanes 4, 5, 6), and N samples were the lowest (lanes 7, 8), and there was no HMGB1 expression in the 9th. β-actin lane shown as the protein-loading control.
The expression of HMGB1 in invasive CSEC with metastasis (T), CSEC (C), and normal cervical squamous epithelia (N).
Statistical analysis for HMGB-1 among: invasive CSEC with metastasis (T), CSEC (C) and normal cervix samples (N) by one-way ANOVA.
Discussion
HMGB1 is a small highly conserved non-histone DNA-binding protein that has been implicated in a variety of biologically important processes, such as transcription, DNA repair, recombination, differen-tiation, development, and extracellular signaling. In certain cell types, HMGB1 is found at the cell surface where it has been shown to contribute to cellular migration and tumour invasion[1,2]. Several publications have reported up-regulated HMGB1 in different tumor types, including uveal melanoma[3], colon cancer[1], prostate cancer[4], pancreatic carcinoma[5], GISTs[6], biliary cancer[7], breast cancer[8], etc.
HMGB1, along with other factors may be an important component of the disordered tumor micro-environment that induces gene mutation, encourages tumor development and local immunosuppression[9,10].
Our results showed over-expression of HMGB1 in CSEC (58.7%) compared with carcinoma in situ (46.7%) and with normal cervical squamous epithelia (16.7%, P<0.01). No significant correlations were found among pathological grades I, II, or III (P>0.05), results that are consist with a study of pancreatic carcinoma[5]. In our study, increased HMGB1 protein levels were observed in 69 of 89 (77.5%) CSEC (size ≥3cm) analyzed by tissue microarrays (P<0.01), that indicated HMGB1 may associated with the proliferation of CSEC. We also found strong up-regulation of HMGB1 in invasive CSEC with metastasis (83.3%) compared to those tumors without metastasis CSEC (52.5%, P<0.01). Western blot analysis showed that the expression of HMGB1 in invasive CSEC with metastasis was more than two times higher than in CSEC without metastasis and about four times higher than the expression in normal cervical squamous epithelia, suggesting that HMGB1 may be involved in metastasis of CSEC cells.
HMGB1 may support tumor growth by its ability as an anti-apoptotic protein. Over-expression of HMGB1 inhibits Bak (and UV)-induced cell death by preventing Casp-9 (and consequently Casp-3) activation[1]. HMGB1 has been suggested to regulate the processes of invasive extension and cell migration in tumor cells through binding to a receptor for advanced glycation end products (RAGE), a multiligand transmembrane receptor belonging to the immunoglobulin superfamily. This results in stimulation of multiple signaling pathways crucial for cell migration, such as Ras-extracellular signal-regulated kinase 1/2, Cdc42/Rac, stress-activated protein kinase/c-Jun-NH2-terminal kinase, and p38 mitogen-activated protein kinase pathways. In addition, it can increase activity of MMP-2 and MMP-9 correlating with the metastatic potential of tumor cells[11,12].
Bandiera[13] et al. detected the expression of HMGB1 in four cell lines (C-4I, CaSki, ME-180, FRTL-5), showing that the expression of HMGB1 in a normal cell line was negative, but highly positive for the transformed cell line. However, the mechanism of HMGB1 induction of metastatic invasion ability of CSEC cells is unknown. Other findings showed that HMGB1 may promote human tumor cell invasiveness by potentiating specific EGF signaling cascades[14] and may have angiogenetic effects on endothelial cells. HMGB1 may have an indirect effect resulting in secretion of angiogenetic factors as, for example, VEGF, tumor necrosis factor-α, and interleukin-8, or it could act as an angiogenetic factor itself[15]. In any event, HMGB1 is associated with CSEC metastasis and further investigation is required to find the details of the signaling pathway, including the downstream targets, upstream activators and to elucidate the mechanism of its effects.
In our study, HMGB1 is associated with the development, growth, and metastasis of CSEC. Therapies based on inhibition of tumor cell invasion may be eventually developed to complement other forms of cancer treatment. Such anti-invasion therapy might be especially valuable in the case of CSEC that char-acteristically destroy surrounding tissue by local invasion. Furthermore the study of HMGB1 will offer an experimental foundation for the further study of CSES pathogenesis.
- Received May 29, 2007.
- Accepted November 12, 2007.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer