Abstract
OBJECTIVE To study the changes in the matrix metalloproteinases-2 and 9 (MMP2, MMP9) induced by 60Co γ-ray external irradiation of human prostate cancer PC-3 cells.
METHODS Human prostate cancer PC-3 cells were irradiated with different doses of 60Coγ-rays. Cell migration and invasiveness were evaluated and the expression of MMP2, and MMP9 was investigated by RT-PCR, Western blotting and flow cytometry(FCM).
RESULTS Irradiation enchances invasive protential at the doses of 1,3 and 5 Gy, whereas it significantly inhibits cell migration.
CONCLUSION The different doses of 60Co γ-ray external irradiation for prostate cancer may have different effects through the changes of MMP2, and MMP9 expression.
keywords
INTRODUCTION
Prostate cancer is the most common malignancy in North American men. Because most patients with this disease miss the opportunity for complete surgical resection at the time of diagnosis, radiotherapy remains as a major means to control tumor progression. However, prostate cancer often shows resistance to radiation. The precise mechanism underlying this radio-resistance is unknown, but several factors have been reported to be involved including cell types, degree of differentiation [1], the cell cycle[2], DNA ploidy[3] and DNA-dependent protein kinase activity[4].
The progression of prostate cancer depends not only on rapid proliferation of tumor cells, but also on other biological characteristics including motility, invasiveness, and metastatic potential. In this respect, conventional colony formation assays or cell viability assays may be insufficient to evaluate the therapeutic effects of radiotherapy. Changes in motility or invasive potential after irradiation have been poorly understood.
In the present study, we examined the effects of irradiation on proliferation, motility, and invasiveness of prostate cancer cells. Based on finding enhancement of invasive potential by irradiation in a subset of prostate cancer cells, we expanded our investigations into the MMP2 and MMP9 proteolytic system known to be relevant to tumor-invasive phenotypes. Collectively, our data suggest that the current concepts of involved-field radiotherapy for prostate cancer need to be reconsidered, and that the inhibition of migration and invasion during radiotherapy may represent a new therapeutic approach to improve the therapeutic efficacy of radiotherapy for prostate cancer.
METERIALS AND METHODS
Cell culture and reagents
The PC-3 prostate cancer cell line used in this study was provided by the Academy of Military Medical Sciences. The cells were maintained in RPMI-1640(Sigma Chemical Co.) supplemented with 10% fetal bovine serum at 37°C with humidified 95% air and 5% CO2. The number of cells was counted with a particle distribution counter.
Irradiation
The cells were irradiated with does of 1,3 or 5 Gy at room temperature using a 60Co source(γ-rays, FC-7000A, ShangDong Xinhua Ltd. China).
Cell proliferation assay
Cell proliferation was evaluated by the MTT assay[5]. Briefly, cells were seeded in 96-well plates at a density of 3×104/well. After overnight incubation, the cells were irradiated and cultured for 7 days. DMSO(150 μl) was added to each well 4 hr after the addition of MTT (20 μl), and the culture media examined by a multiwell plate-reader at 490 nm. The cell proliferation rate was calculated based on the OD at 490 nm of each well at the time point indicated at the day of irradiation.
Matrigel invasion assay
Invassion of the PC-3 cells was measured by the invasion of cells through Matrigel-coated transwell inserts [6,7]. Briefly, transwell inserts with 8 μm pores were coated with Matrigel(40 μg/well). Cell suspensions(500 μl;1×105/ml)were added to the upper chambers while 750 μl of the same medium was placed in the lower wells. Thereafter, the cells were irradiated and incubated for 24 h. Cells that had invaded into the lower surface of the Matrigel-coated membrane were fixed with 70% ethanol, stained with H&E, and counted in five random fields under a light microscope.
Flow cytometry
For the MMP2 and MMP9 analysis, the PC-3 cells were irradiated, washed with PBS, incubated with trypsin for 30 s at room temperature and harvested. The cells(5×105) were incubated with 200 μl(1:50) of either MMP2 or MMP9 mouse monoclonal antibodies(Zhongshan Biotechnology Co Ltd. China) for 120 min at 4°C with protection from light. Then 100 μl(1:100) of mouse IgG isotype antibody with FITC(Zhongshan Biotechnology Co Ltd. China) was added followed by incubation for 45 min at 4°C in the dark. The cells were then washed twice with PBS and analyzed on a FITC calibur flow cytometer using Cell Quest acquisition and analysis software(Stata 7.0 American).
RT-PCR
Total RNA was purified with ISOGEN(Nippon Gene, Tokyo, Japan) RNA extraction reagent. Complementary DNA(cDNA) was made by reverse transcription(RT) of 1 μg total RNA for each assay by using a Ready-To-GoTMT-Primed First-Strand Kit(Amershan Pharmmacia Biotech, Inc., Piscataway, NJ). Each cDNA sample was amplified with 10 μl of PLATINUM polymerase chain reaction(PCR) Supermix(Life Technologies, Inc., Rockville, MD). The PCR reactions for glyceraldehydes-3-phosphate dehydrogenase(GAPDH; 94°C for 15s; 56°C for 30s; 72°C for 30s), MMP2 and MMP9 (94°C for 2min; 92°C for 30s; 62°C for 30s; 72°C for 1min) were carried out by using the following forward and reverse primers to detect MMP2, MMP9 and GAPDH(housekeeping gene, positive control). The primer pairs were as follows:
MMP2
5ˋ-AGA GTT GGC AGT GCA ATA CC-3ˋ
5ˋ-TTG CCA TAC TTC ACA CGG AC-3ˋ
MMP9
5ˋ-CGG AGC ACG GAG ACG GCT AT-3ˋ
5ˋ-TGA AGG GGA AGA CGC ACA GC-3ˋ
GAPDH
5ˋ-ACC ACA GTC CAT GCC ATC AC-3ˋ
5ˋ-TCA CCA CCC TGT TGC TGT TA-3ˋ
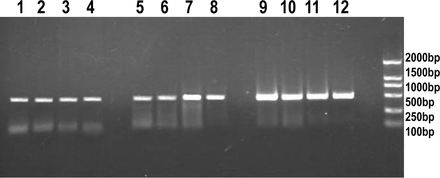
Each of the amplified products for MMP2, MMP9 and GAPDH were of the expected sizes (520, 537 and 543 bp, respectively) as determined by 1.5% agarose gel electrophoresis.
Western blotting
The proteins (80 μg/lane) from the soluble fraction of the PC-3 cells were fractionated by 10% SDS-PAGE and transferred to a polyvinylidene difluoride membrane (Millipore, Bedford, MA). The membrane was incubated with 1:500 dilutions of polyclonal antibody for human MMP2 and MMP9 (Zhongshan Biotechnology Co Ltd. China) and then probed with antigoat IgG conjugated with horseradish peroxidase (Zhongshan Biotechnology Co Ltd. China). Immunoblots were detected by enhanced chemiluminescence (Amersham International, Buckinghamshire, United Kingdom).
Statistical analysis
Statistical analyses were performed by using ANOVA and unpaired Student’s t test. All of the statistics were performed on a two-sided test. P<0.05 was considered as significant. Each experiment was repeated three times.
RESULTS
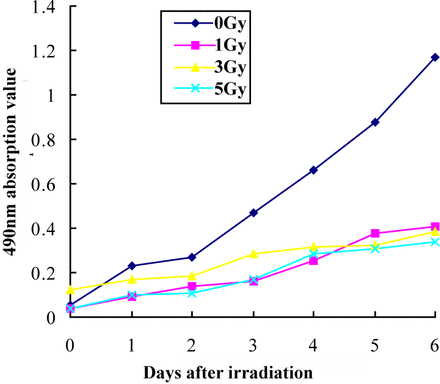
Irradiation inhibits proliferation of PC-3 cells
We examined the proliferation of the PC-3 cells after irradiation. Irradiation suppressed proliferation of the PC-3 cells in a dose-dependent manner, and almost complete inhibition was observed at doses of 1, 3 and 5 Gy (Fig.1).
Effect of irradiation on the growth of PC-3 cells: Irradiation suppressed the proliferation of thePC-3 cells at doses of 1, 3 and 5 Gy.
Irradiation promotes invasive potential of the PC-3 cells
We examined changes in the invasive capacity of the PC-3 cells after irradiation using the Matrigel inva-sion assay. In contrast to the decline in migration ability, invasive potential of the PC-3 cell line was significantly increased after irradiation at doses of 1,3 and 5 Gy(Table.1).This increase in invasive potential appeared to be dose-dependent over the 1 and 3 Gy range.
Results of the Matrigel invasion assay with the PC-3 cell line under graded doses of γ-irradiation.
Flow cytometry
Flow cytometry analysis showed that the expressed level of MMP2 and MMP9 in the PC-3 cells altered under graded doses of γ-irradiation (Table 2), suggesting that different doses of γ-irradiation can influence migration and invasiveness of the PC-3 cell line.
Flow cytometric analysis of MMP2 and MMP9 expression in the PC-3 cells after irradiation.
The results show that the level of the MMP2 and MMP9 proteins increased after 1 and 3 Gy of irradiation whereas the expression level in the 5 Gy-treated group tended to decrease. The experiment was repeated thrice with similar results.
RT-PCR
The expression of MMP2 and MMP9 mRNA was detected in the PC-3 cells under the PCR conditions used. MMP2 and MMP9 mRNA expression increased after the irradiation with 1,3 and 5 Gy compared to the control group, whereas the 5 Gy group showed a lower level compared with the 3 Gy-treated group(Fig.2).
RT-PCR analysis of MMP2 and MMP9 gene expression in PC-3 cells after γ irradiation :MMP2 (lane 1 control; lane 2, 1 Gy; lane 3, 3 Gy; lane 4, 5 Gy); MMP9 (lane 5, control; lane 6, 1 Gy; lane 7, 3 Gy; lane 8, 5 Gy); GAPDH (lane 9, control; lane 10, 1 Gy; lane 11, 3 Gy; lane 12, 5 Gy); M (lane M, A 2000-bp DNA ladder was used as a size marker).
Irradiation increases MMP2 and MMP9 expression
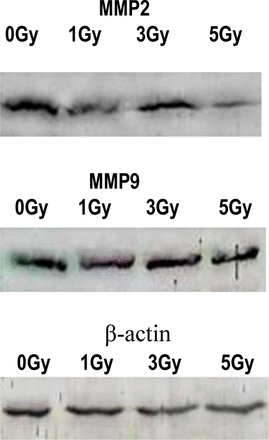
To determine the role of gelatinases related to the radiation-induced changes in invasive potential, we examined the MMPS expression in the PC-3 cells before and after irradiation. The cells were incubated 24 h after irradiation, and the conditioned medium subjected to the gelatin zymography. After irradiation, MMP2 and MMP9 expression was significantly increased(Fig.3, Table 3), thus suggesting that the increase in MMP2 and MMP9 production may play an important role in the enchanced invasiveness after irradiation.
MMP2 and MMP9 expression in the PC-3 cells before and after irradiation.
MMP2 and MMP9 protein absorbance mean value.
DISCUSSION
Prostate cancer is the leading cause of male cancerrelated deaths in the United states, and for many years the results for treating metastic prostate cancer have been disappointing. Radiotherapy, or the use of ionizing radiation directed toward malignant tissue, has proved its usefulness in the treatment of many cancers, and has a significant role to play in the management of genitourinary malignancies. In essence, radiation is effective at a cellular level by causing both single strand and double-strand DNA breaks within malignant cells. This leads to a loss of the reproductive integrity of the cell. Involved-field radiotherapy has remained the single most effective treatment of prostate cancer. However whole pelvis irradiation does not decrease the risk of recurrence or alter the site of a recurrent tumor, but seriously increases the risk of adverse reactions, e.g., diarrhea, rectal irritation dysuria, and frequency of urination [8].
Brachytherapy, i.e., stereotactic interstitial irradiation using temporary high activity isotopes, is used as an alternative to conventional radiotherapy with the aim of providing a high dose of irradiation to the targeted volume. This therapy can potentially enhance local control, with minimal toxicity to healthy tissue surrounding the tumor bed. Unfortunately, this therapeutic modality has led to a significant increase in recurrencies at distant sites[9,10]. The effects of sublethal doses of irradiation on migration and invasion reported suggest that ionizing irradiation from the implanted isotope might promote centrifugal migration and invasion of cells at the tumor border.
Enhanced migration and invasiveness of sublethally irradiated PC-3 cells involved enhanced MMP expression. The matrix metalloproteinases(MMP)-2 and MMP9 are known to degrade the extra-cellular matrix(ECM) and to be involved in the invasion and metastasis of cancer cells[11]. MMP2 is a protease capable of degrading type IV collagen, a major component of the cellular basement membrane. Fur-thermore, MMP2 and MMP9 have been shown to be associated with invasion and metastasis of cancer cells[12].
Our results have shown that irradiation promotes invasive potential at least in the PC-3 cell line, whereas cell proliferation and motility were significantly suppressed at 1, 3 and 5 Gy; and we found that the increased invasiveness after irradiation was associated with increased expression of MMP2 and MMP9. Qian et al. [13] reported that irradiation enchances invasive potential in some pancreatic cancer cells. They have shown that simultaneous administration of a MMP inhibitor during radiotherapy could be a potent adjuvant therapeutic approach to improve the efficacy of pancreatic cancer radiotherapy. Wild-Bode et al. [14] showed that sublethal doses of irradiation enhanced the migration and invasiveness of human glioblastoma cells in association with enhanced expression/activity of MMP2 and MMP9. Furthermore, Camphausen et al. [15] reported that radiation therapy of a primary Lewis lung carcinoma accelerated metastatic growth in mice. These findings support our present results with prostate cancer cells.
Importantly, radiation-induced enhancement in invasiveness was observed at doses of radiation used clinically as in fractionated irradiation of carcinomas. These results suggest that the feasibility of the current radiotherapy needs to be reconsidered. Additional investigations are needed for elucidating the relevant molecular mechanisms behind the different responses toward irradiation.
- Received February 27, 2007.
- Accepted March 16, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer