Abstract
OBJECTIVE To investigate the effect of neoadjuvant chemotherapy in treatment of Stage II breast cancer.
METHODS The data from 113 patients with breast cancer of the same pathologic type in Stage II, during the period of 1995 to 2001, were analyzed retrospectively. Among the patients, 47 were treated with neoadjuvant chemotherapy, and 66 received no adjuvant therapy before surgery (control group). After the patients of the neoadjuvant chemotherapy group had received 2 courses of chemotherapy with the CMF regimen, the surgical procedure was conducted.
RESULTS Complete remission (CR) was attained in 9 of the 47 cases receiving neoadjuvant chemotherapy and partial remission (PR) was reached for 22 cases. The rate of breast-conserving surgery was enhanced from 22.73% to 46.81% (P<0.05) in the neoadjuvant treatment group.
There was no difference in the 5-year overall survival (OS) and disease-free survival (DFS) rate between the two groups (P>0.05), but the 5-year OS and DFS of the cases with clinical tumor remission was higher compared to the control group (P<0.05).
CONCLUSION Neoadjuvant chemotherapy can enhance the rate of breast conservation for Stage II breast cancer and may improve the prognosis of the cases with clinical remission.
keywords
INTRODUCTION
Neoadjuvant chemotherapy involves systemic chemotherapy conducted before an operation or radiotherapy, the effects being different from postoperative adjuvant chemotherapy. It has an ideal therapeutic efficacy on local advanced breast cancer[1,2], but the effects of chemotherapy on the Stage II breast cancer are still under discussion. In this study, the data from Stage II breast cancer patients was reviewed retrospectively to determine the effect of neoadjuvant chemotherapy on this disease.
MATERIAL AND METHODS
Clinical data
Data from a total of 113 patients who had been hospitalized during the period from June 1995 to June 2001, with the Stage II breast cancer of infiltrating ductal carcinoma (IDC), were analyzed. A group of 47 patients who received neoadjuvant chemotherapy were confirmed to have breast cancer by preoperative biopsy and cytological examination of a breast lump. All 47 patients underwent two courses of the CMF regimen before operation (CTX 600 mg/m2, venous bolus injection, D1 and D8; MTX 30 mg/m2, venous bolus injection, D1 and D8; 5-FU 500 mg/m2, venous bolus injection, D1 and D8). The other 66 patients did not receive a preoperative biopsy or neoadjuvant chemotherapy. In all cases no distant metastases were found before treatment.
Observation of the curative effect
In the neoadjuvant-chemotherapy group, alteration of the tumor size before and after chemotherapy was recorded in detail. The mode of operation and status of the lymph node and receptors in all cases were recorded; regular return visits or a doctor’s survey by phone calls were advised, with a overall followup rate of 93%. The recurrence and death time of the patients were accurately recorded.
Criteria for judgment of curative effect
Based on the evaluation criterion for the effect of chemotherapy on breast cancer stipulated by the International union against cancer (UICC)[3], the size of the lump in the breast (SLB) before and after neoadjuvant chemotherapy was measured for analysis (product of multiplication between the longest lump diameter and the longest longitude line vertical to the diameter). The calculation method for clinical tumor retraction (TR) after chemotherapy is shown as follows:
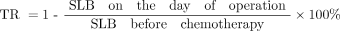
Based on the effect of neoadjuvant chemotherapy, the condition was divided into 5 groups, i.e., a) the group with pathological CR (PCR) meaning there was no histological tumorous residue after chemotherapy; b) the group with clinical CR, meaning no tumorous residue was found after clinical examination; c) the group with clinical partial remission (PR) meant that retraction of the tumor size was more than 50% after chemotherapy; d) the group with no change (NC) of tumor size indicated that retraction of the tumor size was less than 50% or with an augmentation of less than 25% after chemotherapy and e) the progress of disease (PD) showing that after chemotherapy, the enlargement of the tumor was more than 25%. The CR cases in combination with the PR cases were called the total effective rate.
Statistical analysis
SPSS11.0 was used for the chi-square test, with P<0.05 as a significant difference.
RESULTS
General information
In this study, there was a total of 47 cases receiving neoadjuvant chemotherapy and 66 cases without neo-adjuvant treatment. For clinical data of the groups, see Table 1. Statistical data showed that the clinical data between the two groups were similar.
Clinical data from 113 patients with breast cancer.
Short and long-term effect of neoadjuvant chemotherapy
In the group receiving neoadjuvant chemotherapy, CR occurred in 9 of the 47 cases (19.15%). Among the 47 cases, 4 attained PCR (8.51%) and 22 reached PR (46.81%), whereas NC occurred in 11 cases (23.40%) and PD in 2 (4.26%). The clinical-effect rate amounted to 65.96% and achievement ratio of the breast-conserving surgery was enhanced up to 46.81%, whereas in the controls, the rate of breast-conserving surgery was only 22.73% (χ2=7.23, P<0.01). Tables 2 and 3 showed the 5-year OS and DFS which were used as an analytical index of the prostecdtive efficacy.
Analysis of prostecdtive efficacy for neoadjuvant chemotherapy.
Survival-time analysis of 31 cases with clinical remission.
DISCUSSION
The short-term effect of neoadjuvant chemotherapy on breast cancer is positive, and clinical CR, even PCR, may be attained in some patients. The clinical remission rate (CRR) may reach a range from 74.30% to 83.33%[4,5]. Data from our study showed that the CRR using neoadjuvant chemotherapy for the Stage II breast cancer may attain 66%, which was lower compared to previous reports from the literature. The reason may relate to the fewer courses of chemotherapy, for only 2 courses were conducted in this study whereas 3 to 4 courses are often given. However, this study demonstrated that after neoadjuvant chemotherapy, breast conservation operability for Stage II breast cancer was significantly raised, i.e. from 22.73% to 46.81% (P<0.05). These results demonstrate that the short-term efficacy of neoadjuvant chemotherapy for Stage II breast cancer is also manifest.
The prospective efficacy of neoadjuvant chemotherapy on advanced breast cancer has been established, with enhancement of the 5-year DFS and DS, however, the value of the therapy for the disease still remains. It has been reported that neoadjuvant chemotherapy may enhance the OS rate[6], but findings of our study demonstrated that there was no effect of neoadjuvant chemotherapy on the 5-year OS and DFS for Stage II breast cancer patients (P>0.05, P>0.05). However, 5-year OS and DFS of the clinical remission cases were all increased (P<0.05, P<0.05), which was in accord with the conclusion from of results of the NSABP[7]. Upon analysis, the reasons might be as follows: for patients with an overt effect of neoadjuvant chemotherapy, most tumor cells were killed after preoperative chemotherapy and distant tumor metastasis caused by trauma during the operation was reduced and avoided, thus increasing the OS and DFS rate; on the other hand, preoperative chemotherapy may minimize rapid growth of the tumor cells in systemic micrometastasis, caused by excision of the primary tumor[8].
To summarize the above, neoadjuvant chemotherapy can not only reduce the size of Stage II breast tumors and enhance the rate of breast-conserving operability, but can improve the 5-year DFS and OS rate and prognosis of the patients with an established effect.
- Received December 15, 2006.
- Accepted January 5, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer











