Abstract
Stereotactic radiosurgery is a non-invasive procedure that utilizes precisely targeted radiation as an ablative surgical tool. Conventional radiosurgery devices, such as the Gamma Knife, rely upon skeletally attached stereotactic frames to immobilize the patient and precisely determine the 3D spatial position of a tumor. A relatively new instrument, the CyberKnife (Accuray, Inc., Sunnyvale, CA), makes it possible to administer radiosurgery without a frame. The CyberKnife localizes clinical targets using a very accurate image-to-image correlation algorithm, and precisely cross-fires high-energy radiation from a lightweight linear accelerator by means of a highly manipulable robotic arm. CyberKnife radiosurgery is an effective alternative to conventional surgery or radiation therapy for a range of tumors and some non-neoplastic disorders. This report will describe CyberKnife technology and oncologic applications in neurosurgery and throughout the body.
keywords
HISTORY of RADIOSURGERY
Radiosurgery is an ablative technique that combines stereotactic localization with multiple cross-fired beams from a highly collimated high-energy radiation source. This surgical procedure was first conceived by Swedish neurosurgeon Lars Leksell in 1951.[1,2] In Leksell’s initial implementation, an orthovoltage x-ray tube was coupled with his first-generation guiding device (Leksell Stereotactic frame) to focus radiation on the Gasserian ganglion for treatment of trigeminal neuralgia. Over the following decade the principles espoused by Leksell were adopted by others using heavy particles. In 1954, John Lawrence initiated charged particle irradiation and ablation of the pituitary gland in breast cancer patients with intractable pain,[3] and in 1961, Raymond Kjellberg began performing proton Bragg peak radiosurgery at the Boston(Harvard) cyclotron unit. Shortly thereafter, arterioyenous malformation (AVM) irradiation was initiated in Boston.[3]
Given the technical limitations of early medical linear accelerators (LINACs) and the tremendous expense of heavy particle sources, Leksell sought to develop an alternative radiosurgical technology. In 1967, Leksell and Larsson built the first dedicated stereotactic radiosurgical device, the Gamma Knife (current manufacturer, Elekta, Inc., Stockholm, Sweden).[4] The prototype unit, installed at the Sophiahemmet Hospital in Stockholm, was designed to create discoid-shaped lesions for the treatment of movement disorders and intractable pain.[5,6] In the early 1970s Gamma Knife radiosurgery was expanded to treat vascular malformations and selected small brain tumors that could be imaged and targeted on simple x-ray.[7] With the advent of computerized tomography (CT) and the installation of a redesigned second-generation Gamma unit at the Karolinska Hospital, radiosurgical treatment of brain tumors became much more practical and commonplace, In 1984, the third-generation 201-source Gamma unit was developed. The first Gamma Knife in North America was installed at the University of Pittsburgh Medical Center in 1986.[8] It was in Pittsburgh that many clinical applications for radiosurgery were refined.
Paralleling the emergence of Gamma Knife radiosurgery in the 1980s was the development of modified LINACs for radiosurgery. In the intervening years, many of the technical shortcomings that had originally discouraged Leksell from using them had been overcome. By the 1980’s, medical LINACs had become potentially more flexible, more widely available, and less expensive. Akin to the Gamma Knife, the LINAC-based radiosurgical devices that were developed also utilized stereotactic frames for target localization. However, the greater number of moving parts in LINAC radiosurgery necessitated a more complex process of calibration so as to ensure precise radiation delivery.[9-18] Working separately, Betti and Derechinsky in Buenos Aires [19] and Colombo in Italy 1201 pioneered the development and application of LINAC radiosurgery to small brain tumors and vascular malformations, as first reported in 1982.[20] A few years later, Sturm in Germany, [21,22] and Winston and Lutz in Boston [23] described further LINAC adaptations for radiosurgery. The latter description led to a commercially available system for equipping nearly any modem medical LINAC with stereotactic radiosurgical capabilities. With the growing availability of both the Gamma Knife and modified LINAC devices over the past two decades, radiosurgery has gradually become a mainstream neurosurgical procedure.
RADIOSURGICAL TECHNOLOGIES
Gamma Knife (models A, B, and C)
The first Gamma Knifes (the A or U prototype)[24,25] contained 179 cobalt-60 sources arranged in a hemispheric array. This prototype system was superseded by a redesigned “A" model sited at the Karolinska Institute in Stockholm. However, these initial units had problems with loading and reloading of the cobalt-60. In response to this limitation, the B model, in which the cobalt sources were arranged in an annular configuration, was developed.[8] The innovations leading to the C model (in 1999) were prompted by the fact that the treatment of non-spherical targets with the Gamma Knife requires the placement of multiple isocenters (shots) within the target region, a process which in turn necessitated several, sometimes many, time-consuming changes in the patient’s head position. The newer “C” technology, combining advanced dose planning with a robotic, automated positioning system (APS) for repositioning, enabled greater dose conformality without a significant increase in treatment time.
LINAC-based radiosurgery
LINAC-based radiosurgery fires multiple intersecting arcs of photon radiation to create a very high dose within the targeted volume and a surrounding steep dose gradient to ensure minimal impact on adjacent normal tissue. This cross-fire approach is analogous to the Gamma Knife’s intersecting beams of cobalt radiation. The most widely available modified LINAC radiosurgical devices include the X-Knife (Radionics Inc., Burlington, MA), the Novalis (BrainLAB, Heimstetten, Germany) and the Trilogy System (Varían Inc, Palo Alto, CA).
Heavy-particle radiosurgery
Proton beam or charged-particle radiosurgery takes advantage of the unique physical characteristics of heavy particles as they transit matter. This phenomenon, termed the Bragg peak, results in a markedly diminished exit dose of radiation.[26] Beam targeting at most heavy particle facilities is generally less precise than most photon radiosurgical instruments. The exceptionally high cost of equipment and maintenance for a heavy particle radiosurgical facility severely limits its availability. For a few unusual conditions such as chordoma and chondrosarcoma, heavy particle radiosurgery, because of its high Linear Energy Transfer (LET), may have advantages over photon/x-ray equivalents.[27]
CYBERKNIFE
Origins of the CyberKnife
The concept of the CyberKnife (Accuray Inc., Sunnyvale, CA) can be traced to Lars Leksell’s vision of radiosurgery. In 1985, while working as a fellow under Leksell, neurosurgeon John Adler realized the radiophysical principles of cross-fire could be applicable to lesions in the spine, chest, and abdomen if the problem of targeting these lesions could be solved. It was also apparent, however, that frame-based targeting, as employed by the Gamma Knife, could not practically be applied to targets outside the brain. This critical understanding proved to be the genesis of image-guided radiosurgery and the CyberKnife. Guthrie and Adler published the first description of this concept in 1991.[28] The prototype device was installed at Stanford in 1994 Fig. 1, where in the following years investigators pioneered a diverse spectrum of CyberKnife applications. In particular, they explored the clinical application of hypo-fractionated or multi-session radiosurgery to a range of pathologic lesions. This experience contributed to the 2001 American Food and Drug Administration (FDA) clearance of the CyberKnife for the treatment of lesions “anywhere in the body where radiation treatment is indicated." To date, more than 100 CyberKnifes have been installed worldwide.
1a: Photograph of the original alpha system CyberKnife installed at Stanford in 1994.
1 b: Photograph of a contemporary CyberKnife: (1 : diagnostic x-ray source; 2: linear accelerator; 3: amorphous silicon x-ray camera; 4: treatment couch)
1c: A schematic image that depicts the position of a patient with respect to the CyberKnife’s orthogonal x-ray imaging cameras. In this instance the head is positioned near the “nominal" isocenter of the system, an approximate point in space defined during the process of system calibration.
Technical characteristics
The CyberKnife is composed of a lightweight (120 kg) and compact 6-MV LINAC mounted on a robotic manipulator that can position and point the LINAC with 6 degrees of freedom and 0.3 mm precision. The critical targeting innovation is real-time image guidance; the CyberKnife is the first-ever radiation instrument to incorporate image guidance. This fully automated targeting process determines the location of a lesion with respect to adjacent skeletal anatomy all within the coordinate system of the robot and LINAC. Changes in target position are detected and beam pointing is corrected throughout treatment.129301 Several recent competing systems currently attempt to emulate the x-ray image-to-image correlation technology of the CyberKnife.
Frameless targeting
Targeting is accomplished by comparing digitally re-constructed radiographs (DRRs) derived from the treatment-planning CT scan with orthogonal x-rays acquired repeatedly during both initial patient setup and treatment. Anatomic translation and rotation are measured by iteratively changing the position of the anatomy in the DRR until an exact match between the radiographs and DRRs is achieved.[31] The target’s coordinates are relayed to the robot manipulator, which adjusts the pointing of the LINAC before radiation is delivered. Image analysis takes less than a second, so the CyberKnife can detect and adjust to changes in target position in near real time. With thin-slice planning CT scans, overall error can be less than 1 mm.[32,33]
Treatment planning
Conventional radiosurgical systems are restricted by their restrictive kinematics, which are either fixed (Gamma Knife) or based on simple gantry rotation medical LINACs. Such devices rely on the concept of an isocenter,[10-34,35] i.e., the single point in space through which all beams pass. The CyberKnife is not so limited; beams can originate at arbitrary points in the workspace and target arbitrary points within the lesion undergoing ablation, thereby allowing complex non-spherical targets to be treated non-isocentrically. The treatment planning system of the CyberKnife is designed to take advantage of this unique mechanical flexibility.
Treatment planning with the CyberKnife system occurs in steps. First, regions of interest are delineated manually on CT or MR images by the treating surgeon or radiation oncologist. The amount of radiation required for tumor ablation and that will be tolerated by critical regions is specified by the user. Next, the system utilizes contour data to create a 3-D representation of the lesion. Based on this geometry, an initial set of beam configurations is defined which originate from a set of discrete points in space (nodes) where the robot stops to aim the LINAC. At each node, a series of beams are aimed from random orientations towards points that are evenly distributed over the surface of the tumor. Finally, optimization methods determine both the selection and weighting of each beam such that the specified dose constraints are met. During patient treatment the LINAC stops at each node and checks target position before firing. Total treatment time depends on the complexity of the plan and dose, but typically ranges from 20 minutes to 1.5 hours.
CLINICAL EXPERIENCE
The CyberKnife has been used to ablate a broad spectrum of brain tumors including all the lesions commonly treated with conventional stereotactic radiosurgical devices like the Gamma Knife. However, because the CyberKnife is frameless, it is also possible to incorporate fractionation or multiple sessions into radiosurgery in ways that appear to improve clinical outcome. Fractionation appears to enhance treatment safety for larger metastatic brain and skull-based tumors, acoustic neuromas and perioptic lesions.
Metastatic brain tumors
The vast majority of brain tumors are metastatic. Extensive clinical experience with the Gamma Knife shows these lesions to be best treated in a single radiosurgical session, which results in a high rate of local control. Chang et al.[36] published the early Stanford > CyberKnife experience with brain métastasés. Seventy-two patients with 84 lesions were treated, all with a single 10 to 36 Gy dose. Comparable to other types of radiosurgery, the tumor control rate was 95% and a 4% incidence of radiation injury was observed.
Shimamoto et al.[37] reported the Osaka CyberKnife experience with ablating 66 metastatic lesions in 41 patients using doses of 9 to 30 Gy. Lower marginal doses (9 to 20 Gy) resulted in an acceptable tumor control rate, but a successful outcome was significantly more likely in patients treated with 24 Gy or greater (23 of 28 tumors decreased in size, 12 were eliminated, and only one tumor progressed). Although the six-month overall survival rate of patients with a performance status (PS) of 0 or 1 was 83%, in patients with a PS of 2 or more, survival was only 13%. Prolonged peritumoral edema persisted in some patients but no severe complications were reported.
Young et al.[38] reported their experience with radio-surgical ablation of selected non-small cell lung cancer brain métastasés using both the Gamma Knife and CyberKnife. In the Gamma Knife arm of this study the single-fraction dose was 14-20 Gy, prescribed to the 50% isodose line. In contrast, the CyberKnife dose, within a cohort of slightly larger brain métastasés all treated in 1-3 sessions, was 14-30 Gy; the prescription isodose line was selected to encompass 95% of the lesion volume. This comparative study reported an overall tumor control rate with the Gamma Knife of 97.8%, and for the group of larger lesions treated with the CyberKnife, 96.7%.
Acoustic neuroma
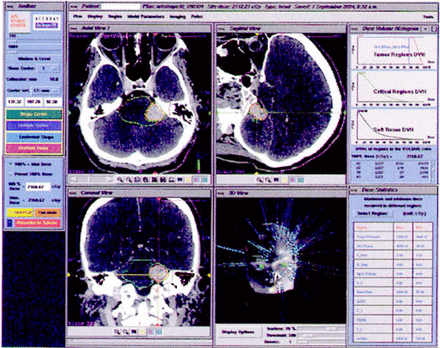
A large literature now supports both the safety and efficacy of radiosurgical ablation of acoustic neuromas. [39] The CyberKnife program at Stanford has sought to improve the rate of hearing preservation after radiosurgery by incorporating the principles of fractionation. An initial estimate of equivalent dose, based on the linear quadratic model and numerous published retrospective studies of single-stage acoustic neuroma radiosurgery, has been refined through years of experience. Since 1998, a 3-session regimen has been utilized, initially with 21 Gy and, since 2000, 18 Gy Fig. 2 prescribed to the 70-80% isodose and corresponding to the edge of the target volume.
A radiosurgical plan for a left acoustic neuroma, involving the left internal auditory canal and cerebellopontine angle cistern; the prescription dose for this three-session procedure is 18 Gy with a Dmax of 21.7 Gy. The dose distribution has been specifically contoured to minimize irradiation of the cochlea and brain stem.
Between 1999 and 2001, 61 patients with unilateral acoustic neuromas were treated at Stanford using three fractions.[39] The mean pretreatment transverse tumor diameter was 18.5 mm (range, 5-32 mm). For the first 14 patients, a total dose of 21 Gy resulted in an excellent clinical response, so the remaining 47 patients were treated with only 18 Gy; the aim of lowering the dose was to further reduce the risk of injury to adjacent cranial nerves.[50] Overall, 29 (48%) of 61 tumors decreased in size and 31 tumors (50%) were stable, resulting in a tumor control rate of 98%. Importantly, after a mean follow-up of 4 years, 74% of patients with serviceable hearing (Gardner-Robinson Class I-II) maintained serviceable hearing, and no patient with at least some hearing before treatment lost hearing on the treated side. In no patient did new trigeminal dysfunction develop, nor did any patient experience permanent injury to their facial nerve; two patients experienced transient facial twitching that resolved in 3 to 5 months.
Ishihara et al.[51] utilized the CyberKnife to treat 14 acoustic neuroma patients with Gardner Robertson class I or II hearing (serviceable hearing) and 24 with Gardner Robertson class III or IV hearing (nonserviceable hearing). The mean tumor volumes were 4.7 cm3 in the hearing patients and 8.2 cm3 in the others. It should be noted that the latter group of lesions were much larger than nearly all prior radiosurgical studies. Radiosurgery was administered in 1-3 fractions. The total marginal dose was 15.0 to 20.5 Gy (mean 17.0 Gy) in the hearing patients and 11.9 to 20.1 Gy (mean 16.9 Gy) in the other patients. While the tumor control rate was 94%, hearing preservation occurred in 93% of cases with serviceable hearing, including the cohort of larger lesions. The authors concluded that improved tumor dose homogeneity and fractionated treatment may improve hearing preservation in patients with acoustic neuroma.
Perioptic lesions
A number of tumors arise in close proximity to the anterior visual pathways and are largely unresectable, using conventional surgical techniques. Such lesions include many pituitary adenomas, meningiomas, craniopharyngiomas, and malignant skull-base tumors. Although single-fraction radiosurgery now has a proven role in managing many of these tumors, the limited radiation tolerance of the optic nerves and the optic chiasm makes it impossible to treat those perioptic lesions that are immediately adjacent to or surrounding the anterior visual pathways.[52-54]
Mehta et al.1521 were the first to report on treatment of lesions involving anterior visual pathways with multi-session CyberKnife radiosurgery. Thirteen patients, 10 with good or excellent pretreatment vision, were analyzed in this preliminary investigation. Among the treated patients, five received 25 Gy in 5 fractions, five were treated with 21 Gy in 3 sessions, and two received 20 Gy in 2 fractions. Doses were prescribed to the 75~95% isodose line. After a median follow-up of 18 months (range 12 to 54), four patients experienced an improvement in either or both visual acuity and visual fields. No visual deterioration or tumor progression was observed.
In 2004, Pham et al.[53] selectively drew from their experience using CyberKnife radiosurgery to treat 399 patients with meningiomas or pituitary adenomas. Thirty-four of these tumors were parasellar meningioma m or pituitary adenoma [14] within 2 mm of the optic apparatus, thereby qualifying as perioptic lesions. In this highly selected group, radiosurgery was delivered in two to five sessions using a mean total dose of 20.0 Gy (range, 15.0-30.0 Gy), as defined at an average 71% isodose line (range, 67-95%). After a mean follow-up period of 29 months (range, 15-62), 32 patients (94%) experienced either a decrease or stabilization in tumor size. Although there was no change in visual field or acuity in 20 patients, improvement in vision was documented in 10 cases. Three patients experienced visual loss; massive tumor progression was the cause in two patients with atypical meningioma. Meanwhile, a prior course of radiotherapy and multiple radiosurgical treatments proved permanently injurious to vision in one patient with multiply recurrent Cushings disease. Most importantly, the vision in 91% of the patients in this series was preserved at pre-treatment levels despite the immediate proximity or even displacement of the optic apparatus. The authors concluded that fractionated CyberKnife radiosurgical ablation can effectively treat many small parasellar lesions while preserving visual function in nearly all cases.
In the largest study of multi-session CyberKnife ablation for perioptic lesions, Adler and colleagues[54] retrospectively analyzed 49 patients with meningioma, [27] pituitary adenoma,[19] craniopharyngioma P1 or a mixed germ cell tumor tl! situated within 2 mm of the optic apparatus. A critical caveat in this report is that the estimated length of optic nerve involvement was less than 2.5 cm. Once again CyberKnife radiosurgery was administered in 2 to 5 sessions. A cumulative marginal dose of 20.3 Gy was delivered to a mean tumor volume of 7.7 cm3. After an average follow-up of 49 months (range: 6-96 months), the visual field was unchanged in 38 patients, improved in eight (16%), and worse in three (6%). Forty-six patients (94%) experienced either a decrease of more than 20% or stabilization (15 cases) in tumor volume throughout followup. Only one previously irradiated patient in this series suffered visual loss that was attributed to radiosurgery. This investigation confirmed that multi-session radiosurgery is generally safe and effective for parasellar lesions in close proximity to a short segment of the anterior visual pathways.
Head and neck
Nasopharyngeal carcinoma (NPC)
NPC has been traditionally treated by radiotherapy alone or in combination with chemotherapy. The present local-regional failure rate is about 20-50% for patients treated by radiotherapy alone. Over the past decade CyberKnife radiosurgical boost after conventional chemoradiotherapy has become a standard part of the management of patients with NPC at Stanford University. Le et al.[55] reported universal local control when stereotactic radiosurgical boost was included in the treatment regimen of patients with NPC. During this prospective study 45 patients with stage II~IV NPC received 66 Gy of conventional external beam radiotherapy (EBRT) delivered at 2 Gy/fracfion. Thirty-six also received concurrent cisplatin-based chemotherapy. Four to 6 weeks after EBRT CyberKnife radiosurgery was delivered in a single 7-15 Gy session to the primary site. The 3-year local control rate was 100% and the overall survival 75%. In addition, the rate of freedom from distant metastasis was 69% while progression-free survival was 71%. Late toxicity included transient cranial nerve weakness in 4 patients, radiation-related retinopathy in one, and asymptomatic temporal lobe necrosis in 3 patients, all of the latter originally having had intracranial tumor extension.
Chen et al.[56] reported their experience with fractionated CyberKnife boost therapy in 44 patients with NPC. All but 5 of these cases received 36 fractions of standard EBRT to 64.8 Gy, followed by 12-15 Gy in 4-5 fractions with the CyberKnife. Forty-one patients also received concurrent or neoadjuvant cisplatin-based chemotherapy. The 2-year rate of local control was 95.4%, the freedom from distant metastasis rate was 81.3%, and the overall survival rate was 87.3%. There were no grade 3 or higher radiation-related complications.
Glomus jugulare tumors
Glomus jugulare tumors are highly vascularized lesions arising from chemoreceptor glomus cells within the adventitial dome of the jugular bulb’s paraganglia. Although histologically benign, these tumors can be locally very aggressive. The goal of intervention is to prevent further tumor growth and neurological compromise. Standard treatment options for affected patients include microsurgical resection, vascular embolization, conventional fractionated EBRT, or some combination. Because surgical extirpation frequently causes injury to adjacent cranial nerves, there has been considerable interest in radiosurgical ablation as an alternative. Preliminary studies with this approach have been uniformly favorable.[57] However, some of these tumors grow interiorly enough in the head so as to be difficult to target with stereotactic frames, thereby limiting the application of conventional radiosurgery to only the more superiorly situated lesions. In contrast, CyberKnife radiosurgical ablation enables surgeons to target these lesions anywhere in the head. Lim et al.[58] described the outcomes after CyberKnife radiosurgery in 13 patients with 16 glomus tumors. Using a prescribed dose of 14~27 Gy, they reported a 100% rate of tumor control with no permanent morbidity.
Spinal radiosurgery
Spinal tumors
Most brain tumors occur within or adjacent to the spine. The frameless CyberKnife targeting system makes it a relatively straightforward process to apply the principles of radiosurgery to these lesions. Nevertheless, the close proximity of the radiation-sensitive spinal cord poses a unique challenge. Despite the risks and uncertainty of the advantages, our team at Stanford began investigating spinal radiosurgery almost a decade ago. These effects were directed primarily towards patients who harbored either unresectable or otherwise poorly treated lesions. During this time, our team developed considerable confidence in the targeting accuracy of the CyberKnife for virtually all paraspinal lesions. Even more importantly, we have acquired a much more nuanced understanding of the spinal cord’s tolerance to ionizing radiation, and in particular, hypofractionated CyberKnife radiosurgery.
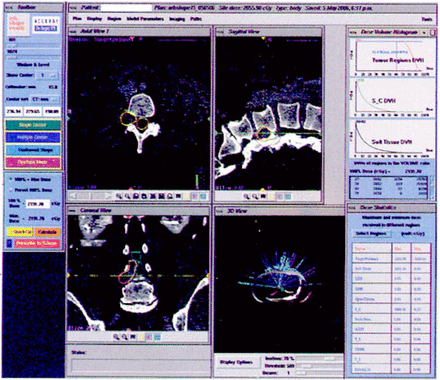
The initial targeting method used by the CyberKnife for spinal radiosurgery was fiducial-based. Murphy et al.[59] first demonstrated that the CyberKnife could accurately track spine lesions based on implanted fiducials (percutaneously placed small stainless steel screws or more simple gold seeds). Targeting accuracies were near a millimeter and competitive with stereotactic frame-based targeting schemes. A recently developed spine tracking technology that comes with the. latest generation of CyberKnife, Xsight ® eliminates the need for implanted fiducials. The Xsight ® system localizes spinal targets by direct reference to the adjacent vertebral elements. Stanford participated in the clinical assessment and development of this technology and has now treated more than 100 spinal patients without implanted metal fiducials Fig. 3. In addition, rigorous phantom studies with the Xsight system now demonstrate targeting accuracies of better than 1 mm.[60,61]
A radiosurgical plan for a right L4-5 neural foramina nerve sheath tumor prior to targeting with the fiducialess Xsight ® system. The marginal prescription dose was 16 Gy in one session.
Ryu et al.[62] first reported the feasibility of treating spinal lesions with the CyberKnife. Target localization was based on stainless steel screws implanted into adjacent vertebral segments. In this retrospective analysis, 16 patients with spinal hemangioblastomas, vascular malformations, metastatic carcinomas, schwannomas, a meningioma, and a chordoma were treated with doses of 11 to 25 Gy in one to five fractions. Among patients followed for at least 6 months there was no evidence of tumor progression or treatment-related complications.
Gerszten et al.[63,64] (who at the University of Pittsburgh have now treated over 700 spinal tumors with the CyberKnife) reported their experience in 125 patients with paraspinal lesions, treated exclusively with a single-fraction technique. This initial series was composed of 17 benign tumors and 108 metastatic lesions. Twelve to 20 Gy (mean, 14 Gy) was prescribed to the 80% isodose line. No acute radiation toxicity or new neurological deficits occurred during the followup period (range, 9~30 months; median, 18 months). Axial and radicular pain improved in 74 of 79 patients.
Recently, Gerszten et al.[65] combined spinal radiosurgery with kyphoplasty, a minimally invasive means for stabilizing vertebral bodies after pathological fractures. Twenty-six patients underwent kyphoplasty followed by single-fraction CyberKnife radiosurgery (mean time after kyphoplasty 12 days). The tumor dose ranged from 16 to 20 Gy (mean' 18 Gy) to the 80% isodose line. No acute radiation toxicity or new neurological deficits occurred during the follow-up period (range: 11~24 months), and axial pain improved in 92% patients. The authors concluded that this combination of minimally invasive procedures can be of great value in patients with pathologic spine fractures, many of whom cannot otherwise tolerate an open spinal operation.
Degen et al.[66] at Georgetown University reported the results of a prospective study that measured safety, pain and quality of life outcomes among a group of patients with spinal tumors treated by the CyberKnife. Patients completed visual analog scale (VAS) pain assessments and the Short Form Health Survey (SF-12). Between March 2002 and March 2003, 51 patients with 72 lesions (58 metastatic and 14 primary) were treated with a mean dose of 2116 cGy at an average 70% isodose line in 1~5 fractions. Many of the patients had received prior radiation therapy. Despite this fact, pain was improved across the board; at 4 weeks the mean VAS score decreased from 51.5 to 21.3. Meanwhile, physical and mental quality of life measures were maintained throughout a study period that averaged one year. No serious side effects were reported.
Dodd et al.[67] published their experience treating 55 benign spinal tumors (30 schwannomas, 9 neurofibromas, 16 meningiomas) with CyberKnife radiosurgery. This collection of lesions was treated with 16-30 Gy to an average 80% isodose line delivered in 1-5 fractions (mean 2 sessions). Tumor volumes varied from 0.136 to 24.6 cm3. Prescribed doses varied depending on histology, from a mean of 2031 cGy for spinal meningiomas to 1870 cGy for spinal schwannomas. Tumors caused pain in 78%, 66%, and 53% of patients with neurofibromas, schwannomas, and meningioma, respectively. Pain was reduced in 25-50% of the patients 12 months after CyberKnife radiosurgery (over half of this group of patients had greater than 24 months follow up). At last follow-up, all lesions were either stable (61%) or smaller (39%). An incomplete radiation-induced myelopathy occurred 8 months after radiosurgery in one patient.
Intramedullary spinal cord AVMs
Intramedullary spinal cord AVMs (SCAVMs) are high-risk lesions which, because of their location within the spinal cord parenchyma, are rarely amenable to traditional endovascular embolization and microsurgical resection. Because there are so few treatment options for most patients with SCAVM, spinal radiosurgery is now proving to be an important new therapeutic tool. Despite initially being uncertain about the potential for catastrophic spinal cord injury, our team at Stanford embarked on an investigation of CyberKnife radiosurgery for SCAVMs almost a decade ago. Twenty-one patients with intramedullary SCAVMs (11 cervical, 7 thoracic, 3 lumbar) have been treated as part of this dose escalation study. The initial results from this experience have been reported.[68] In our series of spinal cord AVM patients, radiosurgery was delivered in 1-5 sessions to an average lesion volume of 1.8 cm3 (range: 0.14-4.94 cm3); the average marginal dose was 19.5 Gy (range: 15.0-21.1 Gy). Patients received clinical and MRI follow-up at 6-month intervals and spinal angiography at three years. Clinical outcome was improved or stable in all patients. After a mean clinical follow-up of 29 months (range, 3-93 months), seven patients have been studied with posttreatment angiography; AVM obliteration (absence of all high flow shunt vessels) was partial in 4 and complete in three patients. To date, twelve patients have been imaged with postoperative contrast MRI alone. Significant AVM obliteration has been observed in nearly all cases who were more than 1 year from radiosurgery. AVM involution appears complete in three of these patients, each of whom awaits confirmatory angiography. No patient experienced a post-radiosurgical hemorrhage. To date it appears that the higher the biologically effective dose, which basically means little or no fractionation, the greater the rate of AVM obliteration.
Intra-thoracic and Intra-abdominal Tumors
Lung tumors
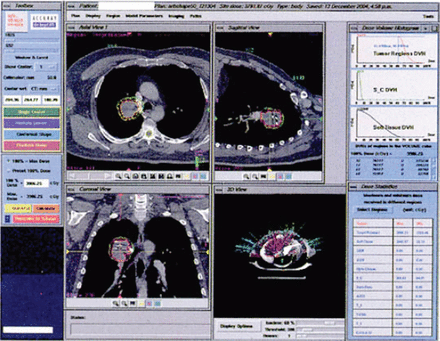
Focal treatment of lung neoplasms can be beneficial, and even curative, in many clinical situations. Cyber-Knife-radiosurgical ablation of lung lesions is a minimally invasive alternative to other more invasive techniques such as mini-thoracotomy and radiofrequency ablation. Like all radiosurgical procedures, lung radiosurgery is intended to deliver the most accurate, conformal and aggressive radiation treatment possible. CyberKnife radiosurgery currently requires fiducials implanted in or near lung lesions for target identification. Other advanced approaches to more precise radiation treatment of lung tumors employ breath-holding, respiratory gating, or abdominal compression exerted by body frames. In contrast, the CyberKnife uses Synchrony ® a method for targeting and tracking tumors in real time that move throughout the respiratory cycle Fig. 4. Dynamic feedback from this system is used to continuously reorient the linear accelerator so that treatment beams accurately follow moving targets.
This radiosurgical plan was designed to ablate in a single session a squamous cell carcinoma of lung with Synchrony ® respiratory tracking. The marginal dose is 25 Gy as prescribed to the 69%ile; the corresponding Dmax is 39 Gy.
A pilot study of CyberKnife radiosurgical ablation for primary lung tumors was initially conducted at Stanford and the Cleveland Clinic. Whyte et al.[69] reported the clinical results of the first-dose increment, 1500 cGy. Tumor motion was addressed either using a breath-holding technique (Stanford) or, at the Cleveland Clinic, by tracking light-emitting diodes placed on the patients' skin (which at the time represented an alpha version of Synchrony). Twenty-three patients received 1500 cGy of radiation in a single session. Although there were four complications, all related to fiducial placement (3 pneumothoraces, 1 emphysema), no grade 3 or higher radiosurgery-related complications were noted. At 1-3 months of follow-up, the radiologic response was deemed complete in 2 patients, partial in 15 and stable in 4; the imaging in two cases demonstrated progressive disease. Although this study demonstrated the feasibility of using CyberKnife radiosurgery to ablate lung lesions, the high recurrence rate showed the need for more aggressive dosing.
In a subsequent paper, Le et al.[70] reported the final experience from the above dose escalation study. Enrolled patients had stage I non-small cell lung cancer (NSCLC) or a solitary metastasis and were judged not to be surgical candidates. Nine to 20 patients were treated per dose cohort starting at 15 Gy/fraction followed by a dose escalation interval of 5~10 Gy to a maximal dose of 30 Gy/fraction. Among patients with NSCLC the 1-year freedom from local progression was 91% for doses >20 Gy but 54% for doses <20 Gy. Patients with primary NSCLC had significantly higher freedom from relapse (FFR) and a trend towards better overall survival than patients with metastatic tumors. One-year FFR was 67% for NSCLC versus 25% for metastatic patients. One-year overall survival was 85% for NSCLC versus 56% for metastatic patients. Perioperative complications stemming from fiducial placement included six cases of pneumothorax, three (9%) of which required chest-tube placement. Radiation-related complications, including four cases of grade 2~3 pneumonitis and one pleural effusion, were noted at doses >25 Gy; there were three possibly treatment-related deaths in patients with a history of prior thoracic radiation therapy. The authors concluded that single-session radiosurgery is feasible for ablating lung lesions and that doses in the range of 25 Gy were both efficacious and well tolerated in previously unirradiated patients. However, this dose level appeared too toxic in a setting of previous thoracic radiation, especially when the lesion was centrally located. This conclusion led to the hypofractionated lung radiosurgery regimen that was subsequently adopted at Stanford.
Hepatocelluar carcinoma (HCC) and liver metastases
There are several modalities currently used to treat HCC, including transarterial chemoembolization, surgical resection, radiofrequency ablation, radioisotope injection, chemical ablation, and radiation therapy.[71] However, none of these has become standard practice. Because it is less invasive, CyberKnife radiosurgery is a potentially more attractive option for such tumors.
Choi et al.[72] were the first to report their experience with CyberKnife ablation in cases of small inoperable or advanced HCC. Nineteen patients with 20 lesions were treated as part of this pilot study. Within Group A, consisting of 15 lesions, the entire primary HCC lesion was ablated. In five patients (Group B) only the component invading the intrahepatic vascular system and producing portal vein tumor thrombosis (PVTT) was targeted. Radiosurgical doses across both patient groups ranged from 30-39 Gy at the 70-85% isodose line, and were delivered in three sessions using Synchrony respiratory tracking. After a median follow-up of 4.3 months, a complete response (CR) was achieved in 8/15 patients in Group A; a partial response (PR) was observed in three cases and stable disease (SD) was recorded in four. Within group B, a CR, PR, or SD were noted in 2/5, 1/5 or 2/5 patients, respectively. One patient suffered severe treatment-related complications.
Pancreatic cancer
Pancreatic cancer continues to be one of the most lethal of all cancers. With early stage tumors, surgical pancreatectomy and aggressive radiation therapy offer at best limited prospects for cure or palliation; even these modest objectives come at the expense of significant rates of morbidity. Given the shortcomings inherent to state-of-the-art treatment, Stanford University has over the past six years been investigating the potential benefits of CyberKnife ablation for pancreatic cancer.
Koong et al.[73] conducted a phase I dose escalation trial in which 15 patients with locally advanced pancreatic cancer were treated in a single CyberKnife session with 15-25 Gy. Tumors were tracked using 3-5 gold fiducials implanted into or near the tumor; breathholding was used to minimize respiratory motion. Twelve patients experienced clinical benefit with significantly decreased pain and increased weight gain. Among the patients treated with 25 Gy, follow-up CT imaging demonstrated 100% local control of the primary pancreatic tumor. For the entire series, the median overall survival was 11 months. No grade 3 or higher toxicities were noted. The authors concluded that 25 Gy was the optimal dose to achieve local control of the primary lesion without inducing significant gastrointestinal toxicity.
A phase II investigation that studied boost radiation delivered to the pancreas by the CyberKnife was conducted by Koong et al.[74] Treatment for this cohort of patients with locally advanced pancreatic cancer consisted of 45 Gy intensity-modulated radiotherapy (IMRT) and concurrent 5-FU, followed by a 25 Gy CyberKnife boost to the primary tumor. Sixteen patients completed the trial. Two experienced Grade 3 toxicity. Fifteen patients were free from local progression until death. Median overall survival was 33 weeks. The authors concluded that the addition of IMRT resulted in a higher rate of complications without any survival or palliative benefit. Because of these findings, IMRT is no longer part of the treatment regimen at Stanford for patients with locally advanced pancreatic cancer.
Prostate cancer
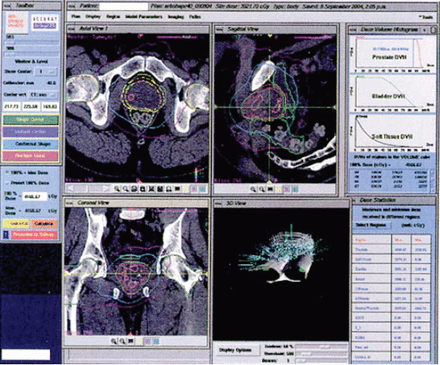
Prostate cancer cells are believed to have a very low a/β ratio, i.e., less than 2. When analyzed by the linear quadratic model, such a value would argue strongly in favor of larger dose fractions than those currently used in conventional radiation therapy. Theoretically such an approach could produce the same or improved tumor control rates, with an even lower incidence of radiation-related complications. The accuracy and conformality achievable with CyberKnife irradiation represent an opportunity to investigate this concept Fig. 5
This radiosurgical plan was delivered to a patient with early-stage prostate adenocarcinoma. The prescription dose is 36.25 Gy at the 87% ile and was administered in 5 fractions.
King et al.[75] reported a case study in which the dosimetry achievable with the CyberKnife was compared to that of IMRT. Three fiducials were placed within the prostate gland for target localization during CyberKnife treatment. Based on dose-volume histograms (DVHs), CyberKnife radiosurgery plans were better than the best IMRT plans in terms of sparing the rectum and bladder while covering the target lesion. This finding suggests that CyberKnife radiosurgery might allow further dose escalation without exceeding the radiation tolerance of adjacent normal tissues. Based on this analysis, a number of CyberKnife facilities in the United States are currently investigating the long-term outcome in men with early stage prostate cancer who have been treated with a one-week course of radiosurgery using five fractions of 7.25 Gy, or a total dose of 36.25 Gy.[76] In addition, Medbery et al.[771 are conducting a phase I trial of CyberKnife radiosurgery for early-stage localized prostate cancer using 38 Gy delivered in four fractions.
Renal tumors
It is debatable whether conventional radiotherapy should play any role in treatment of primary renal cell carcinoma (RCC). Although a few early series [78,79] suggested an improvement in survival among patients with RCC when preoperative adjunctive radiotherapy was used, this benefit was associated with a much higher complication rate.[80,81] Despite this marginal endorsement, Ponsky et al.[82] investigated the utility of renal tissue ablation using CyberKnife radiosurgery in a porcine model; the authors reasoned that the accuracy and dose gradient of the CyberKnife might enable dramatically better outcomes more akin to radiofrequency ablation. In this study small volumes within the pig’s kidney were targeted using a prescribed dose of 24~40 Gy in a single fraction. Gross and histological evaluations were completed at 4, 6, or 8 weeks. After 8 weeks, the lesions showed complete fibrosis without injury to any of the normal adjacent kidney elements. The authors concluded that CyberKnife radiosurgery represented a potentially important new therapy for discrete renal masses.
CONCLUSION
The CyberKnife combines image-guidance technology and computer-controlled robotics to enable state-of-the-art frameless radiosurgery virtually anywhere in the body. In addition to being highly accurate, this procedure makes possible homogeneous irradiation of tumors with complex shapes and the delivery of fractionated “multi-session" treatments. There is now extensive clinical experience that documents the application of CyberKnife radiosurgery to lesions throughout the brain, head and neck and spinal axis. For intracranial lesions, clinical outcomes with the Gamma Knife and CyberKnife are comparable. However, for many of the current brain indications for CyberKnife radiosurgery, such as larger lesions or perioptic tumors, conventional “single-session" Gamma Knife treatment is not an option. As a consequence, CyberKnife radiosurgery for head and neck tumors as well as spinal lesions is growing in importance. Minimally-invasive CyberKnife ablation of tumors within the chest, abdomen and pelvis is moving beyond the stage of feasibility and finding growing acceptance as treatment parameters are refined. Should further clinical investigation validate early evidence of efficacy, CyberKnife ablation is likely to become an important tool for managing nearly all cancers.
- Received August 5, 2006.
- Accepted August 21, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer