Abstract
OBJECTIVE To determine whether Bcl-2 protein down-regulation can render NCI-460 cells more susceptible to gamma radiation-induced apoptosis by treatment with antisense oligonucleotide (ASODN) against the coding region of Bcl-2 mRNA.
METHODS Cell survival was determined using the trypan blue dye exclusion. Expression of the Bcl-2 protein was assayed using immunofluorescence labeling with fluoresce isothiocyanate. Apoptosis was determined by Giemsa staining and flow cytomertry.
RESULTS It was found that Bcl-2 ASODN combined with radiation significantly reduced the number of viable cells (P<0.05). There was no difference in cell survival between a nonsense oligodeoxynucleotide/radiation combination and cells treated with radiation alone. Bcl-2 ASODN combined with radiation significantly inhibited expression of the Bcl-2 protein in the NCI-H460 cells (P<0.05). Using Giemsa staining, cells treated with Bcl-2 ASODN combined with radiation at 72 h displayed classic apoptotic changes. Apoptotic rates of the NCI-H460 cells treated with Bcl-2 ASODN combined with radiation significantly increased (P<0.05), compared with either a nonsense oligodeoxynucleotide/radiation combination or radiation-treatment cells alone.
CONCLUSION ASODN against the coding region of Bcl-2 mRNA increases radiation-induced apoptosis in NCI-H460 cells.
keywords
The Bcl-2 gene is the prototype of a class of oncogenes that contributes to neoplastic progression by enhancing cell survival through inhibition of apoptosis. Expression of Bcl-2 has been observed in the majority of human cancer specimens and cell lines and Bcl-2 plays a major role in the response of malignant cells to a variety of stresses that produce cellular damage, including chemotherapy. Malignant cell lines transfected with the Bcl-2 gene, with resultant overexpression of the protein product, demonstrate increased resistance to various chemotherapeutic reagents.[1-3] It has been shown that antisense-mediated reduction in Bcl-2 protein levels could ultimately induce a lower apoptotic threshold and restore chemosensitivity in a variety of hematologic malignancies, including acute leukemia and non-Hodgkin lymphoma. [4-6] The Bcl-2 antisense oligonucleotides, G3139 (Genasense, oblimersen), which has been developed by Genta Inc, is currently in phase ll-III clinical trials.[7,8] In our lab, a novel antisense sequence has been identified in the coding region of Bcl-2 mRNA that effectively down-regulates expression of the Bcl-2 protooncogene.[9,10] Whether the novel antisense oligodeoxynucleotide (A-SODN) of Bcl-2 mRNA can overcome radiation-resistance in cancer therapy has not been elucidated. This study was conducted to further investigate the effect of Bcl-2 A-SODN on gamma radiation-induced apoptosis in nonsmall-cell lung cancer NCI-H460 cells, which might be helpful in developing new radiosensitization drugs.
Materials and Methods
Cell culture
The non-small-cell lung cancer NCI-H460 cell line was purchased from the Shanhai Cell Bank. The cells were cultured in RPMI medium supplemented with 10% heat-inactivated fetal calf serum at 37°C under 5% CO2 in a humidified incubator.
Synthesis of oligonucleotides and reagents
The sequence of the ASODN targeting the coding region of the Bcl-2 mRNA was 5’-ATC CTC CCC CAG TTC ACC-3’. For control, nonsense oligodeoxynucleotide (NSODN) was used. The sequence was 5’-CAC CCC AAT TCT TCC GCC-3’. The phosphorothioate oligodeoxynucleotid was synthesized and purified by Shanghai Sangon Biology Engineer Corporation. RPMI-1640 and newborn calf serum were purchased from Gibco BRL.
Assay of viable cells
To determine cell viability, NCI-H460 cells were seeded at a density of 1 × 104 cells per well in 96-well plates. Bcl-2 ASODN, or NSODN was added to the cells at a final concentration of 10 μmol/L. Four hours later, radiation alone, radiation plus Bcl-2 ASODN, and radiation plus NSODN groups were irradiated at room temperature using 60Co gamma radiation of 70 cGy/min within a dose range of 2 and 4 Gy. After treatment, the cells were incubated at 37°C for 72 h. Viable cells were counted by the Trypan blue dye exclusion assay.
Quantitation of the Bcl-2 protein by flow cytometry
All cells were collected by centrifugation (2000 × g) for 5 min, and then washed twice and fixed in 4% formaldehyde (Sigma) for 30min on ice, followed by permeabilization with 0.1% Triton X in phosphate-buffered albumin for 10 min at 4°C. The cells were then washed in cold phosphate-buffered saline (PBS) before the addition of 10 μl of anti-BcI-2 antibody (1: 100) (DAKO, Carpinteria, CA) for 30 min at room temperature in the dark. After washing twice with PBS, 10 μl of fluorescent isothiocyanate (FITC)-conjugated second antibody (1:100) was added. After again washing twice with PBS again, the cells were subjected to flow cytometry (Becton Dickinson) and the percentage of cells with positive staining for Bcl-2 protein was determined.
Measurement of apoptotic cells
NCLH460 cells of each group were harvested, and fixed in methanol. Morphology was examined using Gimesa stain under a light microscope. Cellular DNA content was determined by staining cells with propidium iodide, followed by flow cytometric analysis. The cells were collected by centrifugation (2000 × g for 5 min), washed with PBS and resuspended in a final volume of 200 μl of ice-cold PBS. One mL of 70% ethanol in PBS was added with vigorous mixing to the resuspended cells. The fixed cells were incubated in the dark at 4°C overnight, then rehydrated in 500 μl of PBS for 25 min and stained for 30 min with propidium iodide before flow analysis. Assessment was conducted with respect to cellular red fluorescence profile (575 nm) using 488 nm excitation at 150 mW from a Coherent Enterprise Laser of a FACS vantage instrument (BD). DNA histograms were produced using LYSIS II software.
Statistical analysis
Results were expressed as x̄±s and analyzed using ANOVA, using a significance level of P<0.05.
RESULTS
Combined effects of Bcl-2 ASODN and radiation on the NCI-H460 cells
To investigate whether down-regulation of Bcl-2 expression has the potential to sensitize NCI-H460 cells to radiation, a combination treatment with Bcl-2 A-SODN and radiation was applied. The cells were exposed to 10 μmol/L Bcl-2 ASODN, followed by gamma irradiation (2 and 4 Gy). A summary of experiments measuring cell death at 24, 48 and 72 h is shown in Table 1. Compared to treatments with either NSODN plus radiation or radiation alone, the viability of the cells was effectively reduced by combining Bcl-2 ASODN and radiation in a dose-dependent fashion. There was no difference on the viability of the cells between NSODN combined radiation and radiation alone.
Cytotoxic effects of the Bcl-2 antisense oliogonucleotide combined with radiation on NCI-H460 cells (%, × ± s, n=3)
Cooperative effect of Bcl-2 ASODN and radiation on Bcl-2 protein levels
By flow cytometry, Bcl-2 levels in the NCI-H460 cells decreased on day 2 of incubation with 10 ptmol/L Bcl-2 ASODN plus radiation, and decreased further on day 3. Expression of Bcl-2 significantly decreased in the cells treated with a combination of 10 μmol/L Bcl-2 ASODN plus radiation for 72 h as compared with either NSODN plus radiation or radiation alone. There was no difference between NSODN plus radiation and radiation alone, P>0.05 (Table 2).
Effects of the Bcl-2 antisense oliogonucleotide combined with radiation on expression of the Bcl-2 protein in NCI-H460 cells (%, × ± s, n=3)
Bcl-2 ASODN combined with radiation induces apoptosis of NCI-H460 cells
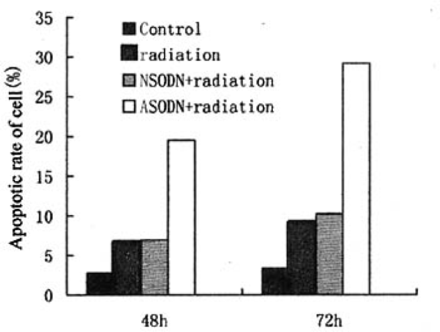
At 72 h after treatment with 10 μmol/L Bcl-2 ASODN followed by irradiation (2 Gy), the NCI-H460 cells took on morphologic characteristics of apoptosis (membrane blebbing, nuclear condensation and fragmentation and formation of apoptotic bodies) (not shown). After treatment with both Bcl-2 ASODN (10 μmol/L) and radiation (2 Gy) for 48 or 72 h the cells exhibited a subGl peak with an apoptotic rate that was significantly different, compared to radiation alone or NSODN combined with radiation, respectively, P<0.05. No significant difference in apoptosis occurred between those cells treated with NSODN plus radiation or radiation alone (P>0.05, Fig.1).
Effects of Bcl-2 ASODN and 2Gy radiation combination on induced apoptosis in NCI-H460 cells.
DISCUSSION
Antisense oligonucleotides have been reported to specifically inhibit expression of various different genes and delay tumour progression in many preclinical models. Antisense drugs that target a number of distinct growth regulatory genes, or interfere with signalling pathways involved in cell proliferation and growth, or target the tumour’s microvasculature are particularly promising in combination with conventional anticancer treatments. Bcl-2 is a prominent member of the Bcl-2 family of proteins that regulate the induction of apoptotic cell death by a wide variety of stimuli. Increased tumor expression of Bcl-2 is considered among the major determinants of resistance to chemotherapeutic drugs and radiotherapy.[1-3] Blocking of Bcl-2 expression and function may represent a relevant therapeutic strategy, and Bcl-2 antisense oligonucleotides have been successful in several preclinical and clinical studies. The currently ongoing phase II-III clinical studies using antisense Bcl-2, known as G3139, aim at the treatment of patients with prostate cancer, melanoma, and non-Hodgkin’s lymphoma. [5,7,8]
Our results indicate that expression of Bcl-2 significantly decreased in NCI-H460 cells treated for 72 h with a combination of 10 μmol/L Bcl-2 ASODN and radiation as compared to either a combination of NSODN and radiation or radiation alone. The viability of the cells exposed to 10 p, mol/L Bcl-2 ASODN, followed by irradiation (2 and 4 Gy), was effectively reduced in a dose-dependent manner, compared to the treatments with either NSODN and radiation or radiation alone. There was no difference in the viability of the cells between those treated with NSODN plus radiation versar and radiation alone. Similarly, the apoptotic rate was also increased in combination-treated cells compared to either NSODN plus radiation treated cells or those treated only with radiation.
Our data indicate that down-regulation of the Bcl-2 protein by Bcl-2 ASODN increased gamma radiation-induced apoptosis in NCI-H460 cells, and that antisense Bcl-2 may be usefill for the treatment of lymphoma with overexpression of Bcl-2. This observation is in agreement with other findings, in which suppression of Bcl-2 can enhance radiosensitization.[11,12]
Footnotes
This work was supported by the Natural Science Program Foundation of the Guangdong Province (021195); Natural Science Program Foundation of the Guangdong Province (04010446).
- Received October 14, 2005.
- Accepted January 19, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer