Abstract
OBJECTIVE To describe a case of a patient with an epithelioid trophoblastic tumor (ETT) and review the literature regarding this new type of uterine trophoblastic tumor which is being reported with increasing frequency. There have been only 42 cases described in the world literature.
METHODS Routine sections of the tumor were prepared and stained with H&E. Using the SP method, immunohistochemical staining, for AE1/AE3, hPL, PLAP, and α-inhibin antigens was conducted.
RESULTS The patient was a 34 years old female who had delivered 12 years previously. She presented with amenorrhea for three months and vaginal irregular bleeding for 2 months. Her serum hCG level was 2,240 IU/L. After diagnostic curettage, an ETT was identified, and total abdominal hysterectomy and bilateral salpingo-oophorectomy (TAHBSO) performed. On microscopic examination it was found that the tumor was composed of chorionic-type intermediate-trophoblastic cells. The tumor cell nests were distributed in a geographical pattern. Some cells were filled with an eosinophilic hyalinized degenerative material. Study of the immunophenotype of the tumor showed that AE1/AE3, hPL, hCG, and α- inhibin were positively expressed.
CONCLUSION This is the 4th case report of an ETT in China. The tumor was identified as a new type of trophoblastic tumor combined with a focal chorioepithelioid carcinoma, a condition that is extremely rare. It consists of chorionic-type intermediate-trophoblastic cells, and is considered to have a lowgrade of malignancy. ETT should be differentiated from a placenta-site trophoblastic tumor, placenta-site nodule, chorioepithelioid carcinoma, and cervical squamous cell carcinoma.
keywords
An epithelioid trophoblastic tumor is a tumor resembling a carcinoma, but it is different from a placenta-site trophoblastic tumor (PSTT) or choriocarcinoma. In 1998, it was given the name of "epithelioid trophoblastic tumor (ETT)" by Shih and Kurman.[1] Before that time, it had been called an "atypical choriocarcinoma" and "multiple nodules of intermediate trophoblast".[2] Up to 2004, 42 cases had been reported in the literature.[1-12] Only one case was not clearly identified, 35 cases (three cases in China) were uterine or cervical lesions, three cases occurred in the lungs, one case in the vagina, one case in the bowel, and one case in the broad ligment of the uterus. Whereas in one case there was no previous pregnancy, the others all had histories of full-term pregnancy, abortion, hydatidiform mole, or chorioepithelioid carcinoma for one to 18 years. Mazur and Kurman[13] assumed that the primary disease of the lung might be the specific reactions to chemotherapy for a hydatidiform mole, or chorioepithelioid carcinoma. Because this tumor is rare, and because it has carcinoma-like morphologic features, it may be misdiagnosed.
Materials And Methods
Clinical features
The patient in this report was a 34 years old female who entered the emergency room because of amenorrhea for three months, irregular bleeding for two months, and one-day pelvic pain. She had delivered a full-term baby uneventfully 12 years ago. A gynecological examination was performed with the finding that the vulva was normal, but dark-red blood was flowing from the vagina. A 3 cm × 2 cm × 2 cm mass was palpated at the upper part of the cervix. Based on the size of uterus she was approximately two months pregnant. No mass could be palpated on both uterine appendages, and with mild pressure pain might be felt. A urine pregnancy test was positive, her blood hCG level was found to be 2,240 IU/L (normal value < 144 IU/L), and her β-hCG level was 50 ng/mL (normal value < 5 ng/mL) prior to the operation. A doppler examination also was performed. The uterine morphology was normal. The uterus was 6.1 cm × 4.7 cm × 4.1 cm in size, and an echo was even when given a sonogram. Her cervix was significantly enlarged, and the wall was uneven, with the thickest part being 4.5 cm. There was no definite embryonal cyst in the cervical canal. The endometrium was thickened to 1.0 cm in the uterine bottom and body. No mass was found in both uterine appendages. The clinical diagnosis was made as follows: 1) cervical pregnancy, and 2) chorioepithelioid carcinoma of uterus. A diagnostic curettage was performed. The pathological diagnosis was determined to be "epithelioid trophoblastic tumor of the uterus". A total hysterectomy and bilateral uterine appendage resection were performed three days later.
Methods
The tissue specimens were fixed in 10% neutral formaldehyde solution. Routine paraffin sections were prepared, with a thickness of 5μ,. H&E and the immunohistochemical staining using a SP method were performed. The first antibodies were hPL, hCG, α-inhibin, and AE1/AE3. All were purchased from the Maixin Reagent Co.
Results
Gross examination
The body of the uterus was 7 cm × 5 cm × 4 cm in size and the cervix was 3.5 cm × 2 cm × 2 cm. Purple necrotic meat-like fragile tissue adhered to the front wall of the uterine cavity and extended to the cervical canal. The entire volume of the tumor was 5.5 cm × 3.5 cm × 3 cm. It invaded the myometrium of the uterus, to a depth of 0.6 cm and the bilateral tubes and ovaries were non-remarkable.
Microscopical examination
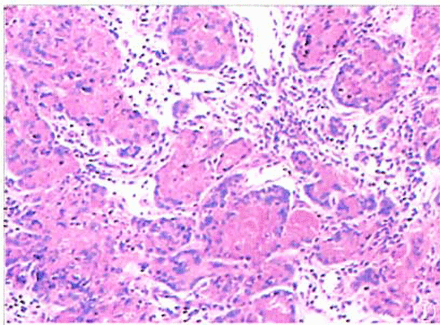
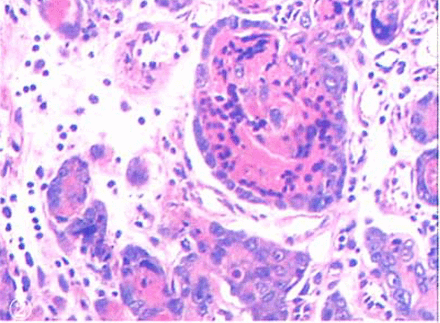
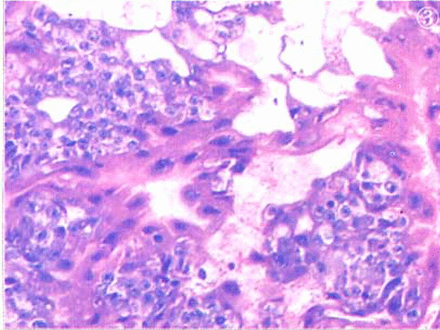
The tumor cells were round, oval, or irregular in shape. Round nuclei were found in the central part of the cells that had eosinophilic cytoplasm. The mitotic count was ≤ 2/10 HPF. There were no abnormal mitoses. The tumor cells were arranged in nests or coalesced in sheets. In some areas, the tumor-cell nests were distributed in a geographical pattern, and in the center of some tumor cell nests the cells were full of eosinophilic hyalinized degenerative and necrotic substances, resembling keratinized material. Blood vessels of different sizes were distributed throughout the areas between the tumor cell nests (Figs. 1, 2). The foci of an accompaning chorioepithelioid carcinoma were found (Fig.3).
ETT cell nests were distributed in a geographical pattern.
The center of some tumor cell nests are full of eosinophilic hyalinized material with lymphocyte cohesion.
ETT accompanied with chorioepithelioid carcinoma.
Immunophenotype
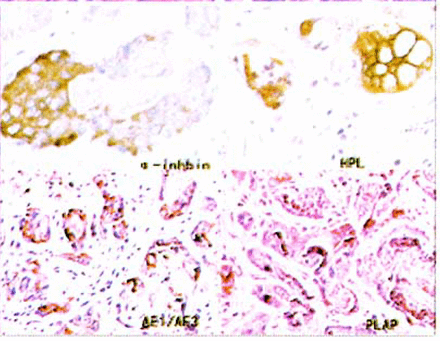
The tumor cells showed positive expression for alpha-inhibin, hPL, hCG, and AE1/AE3 (Fig.4).
ETT immunophenotype positive expression as follows: α-inhibin, HPL, CK, AE1/AE3, PLAP (S-P method).
The diagnosis was determined to be an epithelioid trophoblastic tumor of the uterus, infiltrated in the cervix, and accompanied by a focal chorioepithelioid carcinoma.
Discussion
Clinical manifestations[1-12, 14, 15]
The ages of most patients with this tumor vary from 15 to 48 years with an average of 36.1 years. Our patient was 34 years old. Only Coulson et al.[11] described a 66-year old postmenopausal woman with this tumor. Most of them have a history of previous pregnancy with a term delivery, abortion, hydatidiform mole, or chorioepithelioid carcinoma. Our patient had a history of full-term labor. The intervals between previous pregnancies and the determination of the diagnoses vary from one to 18 years, with an average interval of approximately six years. Our patient was pregnant 12 years ago. Almost all patients suffer from abnormal vaginal bleeding, and patients without symptoms are rare. Preoperative serum hCG values are slightly increased in all patients. Occasionally, this value is as high as 12,541 IU/L. In our patient, the preoperative hCG value was 2,240 IU/L, suggesting a diagnosis of chorioepithelioid carcinoma. Then diagnostic curettage was performed, and the diagnosis was confirmed. Preoperative diagnostic curettage is very important for diagnosis of this tumor.
In 2002, Kamoi et al.[9] reported one case in which atypical multinucleated giant cells were found with a preoperative endometrial douching method. This was an important clue leading to the diagnosis. In 1998, Shih and Kurman [1] reported that of 14 cases, in two there was no primary tumor in the uterus. Therefore, they were extrauterine ETT, hCG went up again after a hydatidiform mole occurred, and chemotherapy failed to be effective. More attention should be paid to this possibility. Occasionally, an ETT diagnosis has been made based on metastases, because of lack of detecting a primary tumor. In 2003, Shen et al. [7] described one case which was accompanied by a chorioepithelioid carcinoma, similar to our case which is quite rare. There hace been occasional reports of extrauterine ETT in the literature. This should be noted more by the research community.
Pathologic features[1-3]
Gross Examination
Almost all ETTs occur either in the uterine body, lower segment of the uterus, or in the cervical cannel. The diameters of tumors have varied from 0.5 cm to 5.0 cm. They are solitary or composed of scattered nodules, and often show invasion into the cervix and/or the myometrium. In our patient, it was coalesced into a sheet, attached to the uterine body, and extended into the cervical cannel, showing an expansive growth pattern and invasion into the superficial muscle layer. There has been one report of a tumor invading to the vaginal orifice, manifesting a polypoid mass. The cut surface of the tumor either was solid or solid with cystic areas. Solid areas were brown in color, accompanied by bleeding and with different degrees of necrosis.
Microscopical examination
ETTs manifest a nodular and expansive growth pattern, composed of chorionic type intermediate trophoblast cells. In typical ETTs, the nuclei are round, and uniform, the cytoplasm is eosinophilic or transparent (rich in glycogen), the nuclear chromatin is fine and scattered, nucleoli might be marked, and cytoplasmic borders are clear. The mitotic count varies from 0 to 9/10 HPF, with an average of 2/10 HPF. Abnormal mitoses are found in a few cases. The tumor cells are arranged into nests and cords which coalesce into sheets. In half of the cases, the tumor cell nests are surrounded by lymphocytes. Often, eosinophilic hyalinized degenerative material and necrotic debris are found in or around the nests. If they are found in the center of the nests, they might be like keratin, as was the case in our patient. Tumor cell islands might be found in the necrotic areas; in their center, there are single blood vessels.
Occasionally, a few amorphous fibrinoid and calcified materials might be deposited on the wall of the vessels, or tumor cells might infiltrate into the vessel wall. These structures seemed to be distributed in a geographical pattern. Most ETT cells (i.e. chorionic-type intermediate-trophoblastic cells ) are smaller than implantation-site intermediate-trophoblastic cells, of which placental site trophoblastic tumors are composed, but the ETT cells are larger than cytotrophoblastic cells. Occasionally, clusters of single implantation-site intermediate-trophoblastic cells are found in the ETT cell nests, or are embedded in the extracellular hyalinized degenerative matrix. Additionally, there is a rare feature, e.g. in about half of the cases, tumor cells replace the epithelial cells of the endocervix. A sudden shift between tumor cells and epithelial cells of the endocervix or squamous cells of the external orifice of the cervix is seen. The tumor cells in these areas often are arranged in two or three layers, in parallel, or at a right angle to the basal membrane. They are larger in size than squamous cells of the cervix. They have abundant eosinophilic cytoplasm and deep chromatic pleomorphic nuclei, much resembling dysplastic squamous cells. Therefore, this might give a pathologist a false impression, e.g. the tumors might arise from squamous epithelium of the cervix. In only a few cases are there foci resembling placental site nodules, PSTT, and chorioepithelioid carcinoma mixed with tumors.
Immunophenotype[16-24]
With the use of various monoclonal antibodies, the tumors show positive expression for cytokeratin (AE1/AE3), epithelial membrane antigen (EMA), E-cadherin, and epidermal growth factor receptor (EGFR), identifying their likely epithelial origin. Additionally, inhibin-α and prolyl 4-hydroxylase are also expressed in the tumors. Shih and Kurman [1] reported that of 14 cases of ETTs, nine cases (64%) were hPL positive, six cases (43% ) were hCG positive, and eight cases (5%) were PLAP positive; in our patient, apart from positive expression of all above mentioned three antibodies, AE1/AE3 was also positively expressed, suggesting that the tumor might have originated from epithelial tissue. Moreover, of the 14 cases, ten cases (71% ) were Mel-CAM positive.[5] This antibody seems to be highly expressed in the implantation-site-intermediate-trophoblastic cells in PSTT. In these tumors, chorionic type intermediate trophoblastic cells are Mel-CAM, hPL, hCG, and PLAP positive, but positive cells usually account for less than 20%, and are arranged in a scattered or clustered pattern. Expression of both E-cadherin and EGFR is highly positive in ETT cells in the tumor nests or on the epithelial surfaces of the endocervix, but expression was only focally positive in implantation-site-intermediate-trophoblastic cells. However, hPL or Mel-CAM were positively expressed in the latter cells.
HLA-G was detected in the tumors and only strongly positively expressed in the intermediate-trophoblastic cells. Therefore, it has been chosen as a valuable marker for these tumors. Ki-67 indexes vary from 10% to 25%, with an average of 17.7% ± 4.5%. They are similar to those cells in the chorion leave, but higher than those of implantation-site-intermediate-trophoblastic cells. The eosinophilic hyalinized degenerative materials in the ETTs have immunoreactivity against collagen IV, fetal and adult fibronectin. The diffusing arrangement pattern of apoptotic cells in the ETTs are identified with in situ terminal deoxynucleotidyl transferase and nick translation assays(TUNEL).
Ultrastructure[10]
Most tumor cells are polygonal in shape. They have single oval nuclei, rich in euchromatin. Nucleoli are prominent. Cytoplasms are moderate in volume, including free ribosomes and extended endoplasmic reticulum, rich in glycogen. There are characteristic intermediate microfilament bundles around the nuclei. The tumor cells are arranged compactly and have multiple intercellular bridge connections. Peripheral cells of the tumor nests have focal incisures of the basal membrane. There are rare sparse short villi on the free surfaces. Original multi-nucleated tumor giant cells are occasionally found. Their nuclei are rich in heterochromatin and the nucleoli are not clear. Cytoplasms are abundant, including organellae and microcysts. There are abundant microvilli on the free surfaces of the cells. The intercellular bridge connections are not identified. Both types of tumor cells mentioned above are arranged along the basal membrane, much resembling the duplicates of trophoblastic layers.
Histogenesis[1, 13, 15, 23]
The tumors are composed of intermediate trophoblastic cells, which in their morphological features are the trophoblastic cells intermediate in appearance between syncytiotrophoblast cells and cytotrophoblast cells. The observations are based on optical and electron microcopic studies. The intermediate trophoblastic cells in this tumor are obviously different from those in PSTT. The latter are similar to the implantation-site intermediate trophoblasts which invade into the myometrium during pregnancy. However the intermediate trophoblasts in this tumor resemble the intermediate trophoblasts in the chorion laeve. In this type of intermediate trophoblast, PLAP, E-cadherin and EGFR are positively expressed; hPL and Mel-CAM are expressed less. But in PSTT, it might be reversed, because hPL and Mel-CAM are strongly expressed. Therefore, the trophoblasts in PSTT and exaggerated placental site (EPS) may be termed as an “implantation-site intermediate trophoblast”, and the trophoblast in the ETT and placental nodule may be termed as a “chorionic-type intermediate trophoblast”.
Another type of intermediate trophoblast may originate from the intermediate trophoblast in the trophoblastic column of the placenta, and be termed as a “villous-intermediate trophoblast”. In 2002, the tumor was analyzed by Oldi and colleagues [23] using molecular genetic assays. Allelic loci of Y-chromatin genes and new allelic genes (might stem from a paternal line) in tumor cells were shown, but in the adjacent normal tissues, there was a lack of those genes. In this tumor, wild K-ras was included in codon 13, suggesting that its mutation has no important effects on development of the trophoblastic tumors and supporting that they may originate from pregnancy, especially following normal pregnancies or abortions. Indeed, tumors which developed after the formation of a hydatidiform mole and chorioepithelioid carcinoma accounted for only less than 10%.
In summary, the clinical manifestations, pathologic morphology, immunophenotypes, and molecular pathologic features all support the concept that ETT originates from chorionic type intermediate trophoblasts. The reasons are as follows: 1) ETTs express particular hormones or enzymes, such as hPL, hCG, and PLAP, of trophoblasts. In addition, they also express Mel-CAM (CD146) and α-inhibin, which are often, but not specifically expressed by trophoblastic diseases. 2) Statistics show that of these tumors, 67% follow full-term pregnancy, 16% follow an abortion, 16% are secondary to a mole, and another 1% are secondary to a chorioepithelioid carcinoma. 3) In almost all ETTs, serum hCG is raised. 4) A few ETTs are acomplicated by placental site nodules, placental site trophoblastic tumors, or chorioepithelioid carcinomas.
Differential diagnoses[1, 20]
Placental-site trophoblastic tumor
In PSTT, the cells have the characteristics of implantation-site intermediate trophoblasts, and invade between muscular bundles, showing an infiltrating pattern. However, in ETTs, the tumor cells have the features of chorionic-type intermediate trophoblasts, and are arranged into cords or nests, among them accompanied by geographical necroses, and calcified material depositions. In PSTT, homogeneous fibrinoid materials are found between the tumor cells. Tumor cells invade into the blood vessel wall and replace endothelial cells. And in this tumor, there are eosinophilic hyalinized-degenerative materials resembling keratin around or in the tumor nests. Moreover, they seldom invade blood vessels. And in PSTT, expression of hPL and Mel-CAM is massive, but in ETT, they only are focally expressed in a spotted pattern.
Placental site nodule
Although this nodule is similar to ETT, because both tumor cells originate from chorionic-type intermediate trophoblasts, they have focal lesions, with fewer tumor cells than those in ETT, atypia are few, nuclear mitoses are lacking or less than those in ETT, and Ki-67 indexes are low. In ETT, however, the lesions are diffuse, tumor cells are more abundant than those in the nodule, atypia are significant, and nuclear mitotic counts and Ki-67 indexes both are higher than those in ETT. All the above criteria can be used to differentiate these two entities.
Chorioepithelioid carcinoma
This cancer is a type of two-phase differentiated (including syncytiotrophoblastic and cytotrophoblastic cells) trophoblastic tumor, whereas, in ETT, tumor cells are a single-phase differentiated-chorionic-type intermediate trophoblasts, which are different from the tumor cells in chorioepithelioid carcinoma. Chorioepithelioid carcinoma has a strongly invasive potential, often accompanied by massive hemorrhage. But this tumor only shows a nodular expanded growth pattern.
Epithelioid leiomyoma
In this tumor, muscle-originating antibodies such as SMA are strongly expressed. In ETT, they are negative, but hCG, hPL, PLAP and α-inhibin may be expressed.
Keratinized squamous cell carcinoma of the cervix or metastatic squamous cell carcinoma
Differential diagnosis of these two tumors is very difficult, because ETT is apt to infiltrate into the lower segment of the uterus or cervix, replacing the surface epithelium. Detection of α-inhibin and hCG may be helpful in differential diagnosis of these two tumors, because in ETT, these two marker types are both positively expressed. However in cervical and metastatic squamous cell carcinomas, they are both negative. Ki-67 indexes of these tumors (10%-25%) are much lower than those of cervical squamous cell carcinomas (75%).
Treatment
Total hysterectomy should be performed immediately after determination of diagnosis by curettage. In young women, uterine appendages on both sides may be preserved based on the merits of each case. Postoperative adjuvant chemotherapy should include actinomycin, VP-16, methotrexate and formyltetrahydrofolate, because routine chemotherapeutic regimen against chorioepithelioid carcinomas is ineffective in treating these tumors. Postoperative monitoring of serum hCG should be performed.
Prognoses[16-24]
In the literature, 42 cases of ETTs have been reported. Apart from 18 cases with lost follow-up, three patients died from this tumor; five developed metastases (including three with lung metastases, one with vaginal metastasis,[4] and one with synchronous metastasis of both lung and bone). Another 16 patients survived, with the survival intervals varing from one to 120 months. In one patient with uterine ETT, hysterectomy was not immediately performed after curettage. Instead, methotrexate chemotherapy was used. However, serum hCG steadily increased, then a total hysterectomy was performed, and after three months of follow-up, she was still surviving. In our patient, uterine ETT involved the cervix, with the complication of focal chorioepithelioid carcinoma. Three courses of postoperative adjuvant chemotherapy were performed with cisplatin and etoposide. After eight months of follow-up, there was no recurrence.
- Received January 15, 2005.
- Accepted March 23, 2005.
- Copyright © 2005 by Tianjin Medical University Cancer Institute & Hospital and Springer