Abstract
OBJECTIVE To investigate the therapeutic potential of amphiregulin antisense RNA delivered by adenoviral vector in a human breast cancer model.
METHODS Human amphiregulin cDNA was subcloned in the opposite orientation to the cytomegaloviral promoter and inserted into an E1/E3 - deleted type 5 adenoviral vector to obtain an AdA4 construct which expresses amphiregulin antisense mRNA. Both in vitro and in vivo anti - proliferative effects of the antisense RNA were studied by infecting transformed human breast epithelial NS2T2A1 cells and tumors.
RESULTS Amphiregulin protein expression was inhibited dramatically in the NS2T2A1 cells after infection with AdA4. The in vitro cell growth was inhibited significantly at day 4 post-AdA4 infection compared with control empty virus AdC1 at a MOI of 200 and 400 pfu/cell to 69.3% and 49.8%, respectively (P< 0.02, P <0.005). After 3 intra-tumoral injections of 109 pfu AdA4, tumor volumes were reduced to 40.6% of that of the control group at day 35 (P< 0.005).
CONCLUSION The transfer of amphiregulin RNA antisense by adenoviral vector is effective for amphiregulin targeting strategy, leading to an inhibition of in vitro cell proliferation and in vivo tumor growth in this breast cancer model.
keywords
Growth factors play an important role in the pathogenesis and progression of breast cancer. Amphiregulin, one of the ligands of epidermal growth factor receptor (EGFR), has been found associated with poor prognostic parameters in breast cancer.[1] In an experimental model, we found that amphiregulin expression augmented with tumorigenicity, and the transformed phenotype could be reversed after transfection with an amphiregulin antisense RNA expressing vector.[2,3] To potentiate the therapeutic effectiveness of antisense strategy, we constructed a recombinant adenovirus carrying amphiregulin antisense cDNA.
Materials and Methods
Preparation of recombinant adenovirus
The 1.1 kb cDNA fragment of human amphiregulin was subcloned in the opposite orientation to the cytomegalovirus (CMV) immediate-early gene promoter and inserted into an El/E3-deleted type 5 adenoviral vector to obtain an AdA4 construct which expresses amphiregulin antisense RNA. Control AdCl contained no insert whereas Ad-β-gal carried the Escherichia coli β-galactosidase (β-gal) gene driven by a RSV promoter. Recombinant adenovirus was constructed and amplified by using El-expressing 293 cells. Viral stocks were purified by CsCl-gradient ultracentrifugation and titrated as plaque-forming units (pfu), and stored at -80°C.
Cell culture and viral infection
Transformed NS2T2A1 cells, derived from normal human breast epithelial cells transfected by SV40 T antigen and then selected by successive passages in nude mice, were cultured in DMEM/F12 (1/1) medium supplemented with 10 μ g/ml insulin, 5 μ mol/L hydrocortisone, 2 ng/ml EGF and Ca2+-chelated 5% horse serum.[4] Human 293 cells were cultured in MEM medium supplemented with 10% fetal bovine serum. Cells were maintained in 5% CO2 in air at 37 °C in a humidified incubator and passed every 3 or 4 days per week after trypsinization. The optimal multiplicity of recombinant virus infection (MOI) of recombinant adenovirus was determined by staining for LacZ after infection with Ad-β3-gal. For NS2T2A1 cells, 200-400 pfu per cell were determined as an optimal compromise between a maximal efficiency of infection and a minimal virus-induced cytopathic effect.
RNA analysis
The amphiregulin cDNA Eco RI-Nae I fragment was subcloned into the pGEM 4 vector (Promega, Madison, WI) to obtain a pGEM-AR construct which was linearized with Xba I and Eco RI respectively to synthesize an amphiregulin sense riboprobe with SP6 polymerase. Linearized pTRI-GAPDH-human antisense control vector (Ambion, Austin, TX) was digested with Dde I to synthesize a GAPDH antisense riboprobe-with T7 polymerase. The probes were labeled with 800 Ci/mmol α-32P UTP using a MAXIscript in vitro transcription kit (Ambion, Austin, TX). The probes were purified by 5% acrylamide gel containihg 8 mol/L urea.
Cellular RNA was isolated 4 days after infection with recombinant adenovirus by the guanidine cesium chloride method and stored at -80 °C. Ten μg cellular RNA was mixed with 3 × 104 cpm labeled probes to perform the RNase protection assay using the Hybspeed RPA kit as described by the manufacturer (Ambion, Austin, TX). The 230 base amphiregulin antisense RNA fragment was protected by the amphiregulin sense riboprobe, and the 154 base GAPDH mRNA fragment was protected by the GAPDH antisense riboprobe as internal control.[3]
Western blot analysis
Total cellular protein was prepared 4 days after recombinant adenovirus infection. Twenty-five μg of protein was loaded on a 12% SDS-PAGE gel for amphiregulin detection. Proteins were then transfered to a nitrocellulose filter and incubated with 1 μg/ml affinity-purified polyclonal rabbit anti-amphiregulin antibody AR-Ab2 and an affinity-purified polyclonal rabbit anti-actin antibody overnight at 4 °C. The blot was then incubated for 1 h with horseradish peroxidase conjugated Fab fragments of anti-rabbit IgG antibody, and amphiregulin protein was visualized with ECL reagent (Amersham Life Science, Buckinghamshire, UK) with actin as internal control.[3]
In vitro growth assay
NS2T2A1 cells (7 × 104) were seeded in 9.6 cm2 cell culture dishes on the day before infection. Virus infection was carried out in triplicate in 0.5 ml of medium for 4 h at a MOI of 200 and 400 pfu/cell, respectively. After PBS rinsing, cells were incubated in EGF-free medium containing 2% of carbon-dextran depleted fetal bovine serum, 4 mmol/L glutamine. Cell growth was evaluated at day 2 and day 4 post-infection by cell counting after trypsinization.
In vivo tumor treatment
NS2T2A1 cells cultured at approximately 80% confluence were harvested and suspended at 107 cells in 0.2 ml PBS and subcutaneously injected into the right flanks of 5-week-old female swiss nu/nu mice (Iffa Credo, L’Arbresle, France) maintained in SPF-P3 condition in the animal experimental centre of Gustave Roussy Institute. The growing tumors were measured once a week with a caliper, and their volumes were calculated as π× (average diameter)3/6. Animal randomization was carried out 7 days after grafting when tumor volume had reached 70.86 ± 23.9 mm3. Three groups were formed, each with 10 animals. Three injections of 100 μl of PBS buffer containing 109 pfu of AdA4 or AdCl or PBS alone were injected directly into the tumors at 5-day intervals. Tumor, volumes were calculated weekly.
Statistical analysis
Data were analyzed using the Student t-test.
Results
AdA4 inhibits amphiregulin protein expression in NS2T2A1 cells
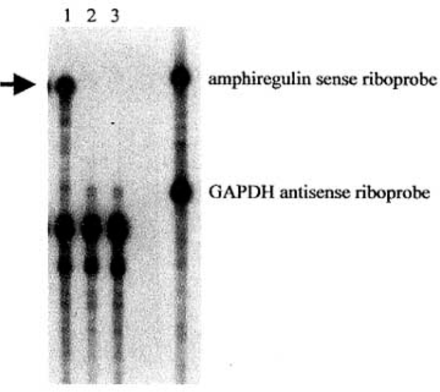
To detect the expression of amphiregulin antisense RNA after AdA4 infection, an RNase protection assay was performed. Total RNA obtained from AdA4 or AdCl infected NS2T2A1 cells was hybridized to a 32P-labeled amphiregulin sense riboprobe that protected a 230 base amphiregulin antisense RNA fragment. As expected, amphiregulin antisense RNA expression was detected only in the cells infected by AdA4(Fig.l).
Adenovirus-mediated expression of amphiregulin antisense RNA. RNase protection assay was performed with cellular RNA from NS2T2A1 cells infected with AdA4 (lane 1), AdC1 (lane 2) or treated with PBS alone (lane 3). The protected amphiregulin antisense RNA fragment is noted by the arrow.
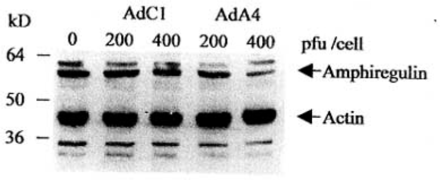
Expression of the amphiregulin protein in the NS2T2A1 cells infected by AdA4 was studied by Western blotting with actin detection as internal control. The production of the 55-60 kD amphiregulin protein was greatly reduced in cells infected by AdA4 when compared with cells infected by AdCl (Fig.2). Therefore amphiregulin protein expression was inhibited in the NS2T2A1 cells after infection with recombinant amphiregulin antisense adenovirus.
Amphiregulin expression after AdA4 infection. Expression of amphiregulin was detected by Western blot analysis.
AdA4 reduces the growth of NS2T2A1 cells in vitro
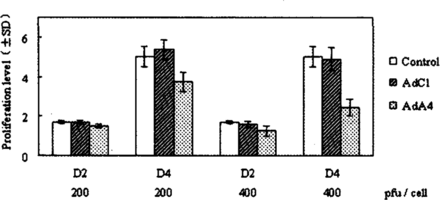
To determine whether AdA4 could inhibit NS2T2A1 cells growth in vitro, viral infection was performed. As shown in Fig. 3, cellular proliferation was significantly inhibited 4 days after AdA4 infection compared with AdCl in a dose-dependent manner. We observed 30.7% cell growth inhibition following AdA4 infection at a MOI of 200 pfu/cell and a 50.2% reduction at a MOI of 400 pfu/cell, respectively ( t = 4.017, P< 0.02; t = 5.887, P < 0.005).
Inhibition of N2T2A1 cell proliferation in vitro by AdA4. Proliferation level is expressed as relative values to the cell number at DO
AdA4 reduces the tumor growth of NS2T2A1 cells in vivo
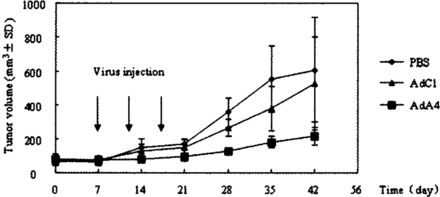
We studied the effect of AdA4 infection on subcutaneously implanted NS2T2A1 tumors in nude mice after three successive intra-tumoral injections of 109 pfu recombinant adenovirus every 5 days. No critical side effects were observed in nude mice m any group. At day 35 postinjection, the volume of the tumors treated with AdA4 was 40.6% of that receiving AdCl (t =3.570, P < 0.005) (Fig.4).
Inhibition of N2T2A1 tumor growth in vivo by AdA4.
Discussion
Amphiregulin is a member of the EGF family, originally isolated from serum-free conditioned medium of the human breast carcinoma cell line MCF-7 treated with the tumor promoter phorbol 12-myristate-13-acetate (PMA). Amphiregulin is commonly expressed by various normal and malignant human epithelial cells, and is increased in mammary tumors developed in transgenic mouse strains overexpressing oncogenes such as TGFα, neu and int-3. These findings suggest a role for amphiregulin in the promotion and/or progressive stages of mouse mammary tumorigenesis. [5] Using an expression cloning strategy, Berquin et al.[6] demonstrated that amphiregulin transforms normal human mammary epithelial cells to EGF independence as aggressive EGFR-overexpressing breast cancer cells. Inhibition of amphiregulin expression reduces not only the proliferation of malignant tumors, but also the intra-tumoral neovascularization.[3,7] In human breast cancers, immunoactive amphiregulin was detected in 35 ~77% of infiltrating breast carcinomas, and correlated with aggressive or poor prognostic forms of tumors.[1]
We have successfully immortalized normal human mammary epithelial cells using SV40 T antigen. After successive passages in nude mice, a highly tumorigenic cell line was selected, NS2T2A1, in which we demonstrated a dramatic increase of amphiregulin expression. We demonstrated that a striking inhibition of amphiregulin expression in the NS2T2A1 cell line following amphiregulin antisense RNA expressing-vector transferction reverses the malignant phenotype of this cell line.[3] In the same model, we found that amphiregulin also induces the expression of urokinase-type plasminogen activator (uPA), TGF-betal (TGFβl), matrix metalloproteinases (MMPs) and extracellular matrix metalloproteinase inducer (EMMPRIN).[8,9] All these results suggest that amphiregulin over-expression induces the invasiveness and metastatic capacity of breast cancer cells, and indicate that this protein could represent a new therapeutic target for breast cancer.
The aim of antisense therapy is to block the expression of the targeted protein, inhibiting its biological effects that are essential for cancer growth and progression. However, clinical applicability of antisense oligonucleotide administration or liposome-mediated antisense delivery approaches seems to be limited by their relatively low efficiency of gene transduction, slow cellular uptake and physiological instability. At the present time, adenoviral vector seems to be a useful vector in clinical oncology because of its simplicity in preparation and high efficiency of gene transduction regardless of tissue type or cell cycle.[10]
In this study, we showed that an adenoviral vector is effective for antisense strategy in our breast cancer model. After infection with recombinant adenovirus, expression of amphiregulin antisense RNA was detectable only in NS2T2A1 cells treated with the amphiregulin antisense adenoviral construct AdA4, which inhibited amphiregulin protein expression. Then, we demonstrated that in vitro proliferation of NS2T2A1 cells was inhibited by AdA4 with a dose-response effect compared with the control empty adenovirus, and injection of AdA4 into the established tumors reduced the in vivo tumor growth to near 60% comparing to the control. The above results demonstrate that adenoviral vector is effective for antisense strategy in our breast cancer model and suggest the necessary to study and develop this technique for possible tumor therapy.
Footnotes
This study was supported by the Advanced Research Program France-China (PRA-B00-08) and the National Science Foundation of China (No. 30171066)
- Received September 3, 2004.
- Accepted March 2, 2005.
- Copyright © 2005 by Tianjin Medical University Cancer Institute & Hospital and Springer