Abstract
OBJECTIVE To investigate the anti-irradiation effects of a. polypeptide from Chlamys farreri (PCF) on γ-ray induced damage to mouse thymocytes.
METHODS Thymocytes were randomly divided into 6 groups including an untreated control, a model group, groups treated with 5, 2.5 and 1.25 mg PCF/ml and a 0.1% vitamin C group. Cell viability, morphology, nucleic acid and enzymatic changes of the cells 3 h after 3 Gy γ-ray irradiation at a dose rate of 0.6 Gy/min were determined.
RESULTS γ-Ray irradiation decreased the cell viability and SOD activity and increased MDA levels and the apoptotic number of cells. PCF increased cell viability and SOD activity and decreased MDA levels and the apoptotic number of the cells in a dose-dependant manner.
CONCLUSION PCF pretreatment can attenuate cytotoxicity, inhibit apoptosis, reduce the level of MDA and maintain the activity of SOD. Therefore PCF has anti-irradiation effects on γ-ray induced damage of mouse thymocytes.
keywords
Ionizing radiation (IR) reacts with living tissues through a series of molecular events.[4] In addition to a direct action on DNA, IR also interacts with intracellular or intercellular water as well as oxygen (O2), generating various aqueous free radicals and reactive oxygen species (ROS). These products then can react with cellular macromolecules, such as DNA, RNA, protein and membrane constituents etc.[6,7] These interactions may cause radiation damage such as DNA-hydroxylation, chromosomal aberrations, micronuclei, lipid peroxidation (LPO) [8] and apoptosis. [9]
Over the past several decades, many natural antioxidants, such as vitamin C, vitamin E, β-carotene and others from herbs and plants have been investigated regarding their ability to inhibit radiation damage.[10,1] However there have been few reports concerning radioprotective polypeptides, especially from marine products. Our previous work [l2] showed that a novel marine polypeptide (PCF, Mr=912) isolated from the gonochoric Chinese scallop Chlamys farreri had radioprotective activity. PCF, an octapeptide consisting of Pro, Asn, Ser, Thr, Arg, Hyl, Cys and Gly, has effective capability of scavenging ROS induced by UV light,™ and has protective effects on nude mice exposed to UVA. [14] Based on these results we suggest that PCF is a potential radioprotector. In this study, we investigated the effects of PCF on thymocytes from Kunming mice irradiated by cobalt-60 γ-rays. We examined the effect of PCF on irradiation damage using a cell viability assay, DNA “ladder” measurements, ultrastructural assessment, flow cytometric analysis and assays of superoxide dismutase and lipid peroxidation activity. Our results for the first time suggest that PCF has anti-irradiation effects against γ-ray-induced damage to mouse thymocytes.
Materials and Methods
Animals and cell culture
Kunming mice (6-8 weeks old, 20-30 g) were purchased from the Animal Center of Qingdao University. Single-cell suspensions of thymocytes in phosphate-buffered saline (PBS) were harvested from a single thymus from one mouse for each test. Cells were cultured in RPMI 1640 medium supplemented with 10% (v/v) heat-inactivated fetal bovine serum, 100 U/ml penicillin, 100 μg/ml streptomycin, and seeded in 6-well culture plates at a density of 1 ⨯ 106 cells/ml, with 3 ml per well as a final volume. The cells were cultured at 37°C in a 5% CO2 humidified atmosphere.
PCF treatment
PCF (pH 6.5-7.0) was dissolved in sterile deionized water and stored at 4°C. Immediately after seeding the cells in 6-well culture plates, PCF was added at concentrations of 5, 2.5 and 1.25 mg/ml for the experimental groups and 0 mg/ml for the model group. Cells cultured in medium alone and in 0.1% vitamin C served as the control and positive groups respectively. All group treatments were conducted in triplicate.
γ-Ray irradiation
After culturing the cells with PCF or vitamin C for 2 h, the cells were exposed at room temperature to 3 Gy of γ-rays from a cobalt-60 source of a dose rate of 0.6 Gy/min. The control group was not irradiated. The cells then were cultured under the same condition for another 3 h and then harvested for the following assays.
Assay of cell viability
Cell viability was measured using the MTT assay according to the manufacturer’s instructions. At the time for assaying, cell suspensions were transformed from the 6-well culture plates to 96-well culture plates in triplicate wells for each group and then MTT was added to each well at a final concentration of 0.5 |μg/|μl. Then cells were cultured for another 4 h. For the assay, 100 μl of dimethylsuloxide (DMSO) was directly added to each well. After the formazan crystals were solubilized, the products obtained were spectrophotometrically quantified using an ELISA reader (λ= 540 nm).
Measurement of DNA fragments
DNA samples were prepared as described by Aggarwal et al.,[15] loaded onto 1.5% agarose ethidium bromide-stained gels with loading buffer and electrophoresed at room temperature for 1 h at 75 V. The gel was then photographed using a UV transilluminator.
Preparation of the cells for transmission electron microscopy (TEM)
Cells to be subjected to TEM were prepared as previously described. [16] Briefly, the cells were harvested, centrifuged, washed with ice-cold PBS and fixed sequentially with 2% glutaraldehyde (v/v) and osmium tetroxide. After staining with uranyl acetate, dehydration and embedding in Epon-812, the samples were cut into thin sections which were then re-stained with lead citrate, examined and photographed using a JEM 1200 ES electron microscope.
Analysis of cell apoptosis
After the cells were harvested and washed, they were treated with propidium iodide (PI) as previously described. [17] Apoptosis of the thymocytes was monitored using FACS Vantage flow cytometry (Becton-Dickinson, USA). The number of cells with a sub-G1 DNA content were identified as apoptotic cells emptying as a MODFIT LT program (Verity Software House, Topsham, ME).
Determination of cellular malondialdehyde and superoxide dismutase
The level of malondialdehyde (MDA) and the activity of superoxide dismutase (SOD) were measured according to the manufacturer’s instructions in assaying kits from the Nanjing Institute of Jiancheng Biological Engineering, China.
The determination of MDA was based on its reaction with thiobarbituric acid to develop a red substance absorbing at 535 nm. Cell homogenate (0.5 ml) from each group was mixed with 0.5 ml of the thiobarbituric acid reagent consisting of 0.67% thiobarbituric acid and acetic acid (1:1, v:v). The reaction mixture was heated at 95°C for 1 h cooled, and 1 ml of n-butanol added. The mixture was shaken vigorously for 30 s, centrifuged at 3000 g for 10 min and the absorbance of the n-butanol layer read at 535 nm using a fluorescence spectrophotometer. The value of fluorescence was calculated by comparing with standards prepared from 1,1,3,3-tetraethoxypropane.
SOD activity was measured its ability to inhibit the oxidation of oxymine by the xanthine-xanthineoxidase system. The red product (nitrite) produced by the oxidation of oxymine has an absorbance at 550 nm. One unit of SOD activity was defined as the activity which reduced the absorbance at 550 nm by 50%.
Statistical analysis
The data were expressed as the mean ± SD. Evaluations were conducted by one-way ANOVA followed by the Student’s t-test to detect inter-group differences. P< 0.05 denoted a statistically significant difference.
Results
Effect of PCF on γ-ray-induced inhibition of cell viability
In this study, cell viability of the model group decreased more than that of the untreated control group. The viability of thymocytes pretreated with 1.25, 2.5 or 5 mg PCF/ml increased in a concentration-depend manner at doses tested (Table 1).
Effect of PCF on cell apoptosis
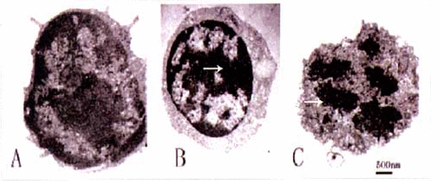
TEM is the most reliable method for assessing apoptosis, so we determined the effect of PCF pretreatment on induction of apoptosis by examination of ultrastructural changes. Typical signs of cell apoptosis including cell shrinkage, blebbing of the plasma membrane, chromatin condensation, margination and fragmentation were presented in the model group (Fig.l). In contrast, thymocytes pretreated with PCF and the untreated control group did not display apoptotic morphologic changes.
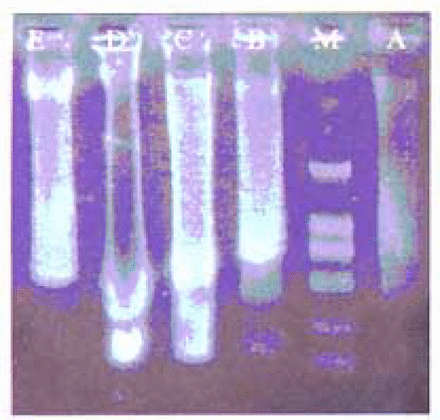
The typical DNA “ladder” (Fig. 2. lane B) appeared in the model group and fewer strips were found in the “ladder” from PCF-pretreated groups, The group treated with 5 mg PCF/ml showed “no” “ladder” (Fig. 2. lane E).
Ultrastructure of the cultured thymocytes. Irradiated cells show typical signs of apoptosis (B, C): cell shrinkage, blebbing of the plasma membrane, chromatin condensation and fragmentation, and vacuolization of micro-organelles. The ceHs pretreated with PCF did not display the morphologic features of apoptosis (A). Scale bar: 500 nm.
Influence of PCF pretreatment on a DNA “ladder”. (A) Untreated control group; (B) Model group; (C) Treated with PCF (2.5 mg/ml) before irradiation; (D) Treated with vitamin C (0.1%) before irradiation; (E) Treated with PCF (5 mg/ml) before irradiation. (M) DNA marker.
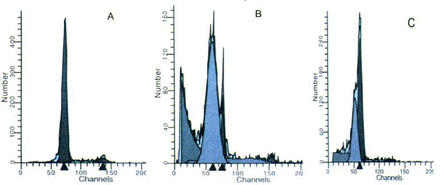
To exactly assess the apoptotic changes we next used FCM analysis to quantify the rates of apoptosis among different groups. The untreated control group contained a very low level (2.44%) of pre-Gl cells, which is representive of the apoptotic population, whereas the cell number in the model group was about 40 times higher (71.81%) than that of the untreated control group (P<0.01). PCF pretreatment decreased the pre-Gl population in a concentration-depend manner, with the maximal inhibitory effect occurring at a concentration of 5 mg PCF/ml. There was a significant difference between the 5 mg PCF/ml group and model group (Fig. 3).
Apoptosis of the thymocytes detected by FCM. The pre-G1 population is representative of the apoptotic cells shown as a gray histogram in the Figure. Groups:(A) Untreated control; (B) Model; (C) Treated with PCF (5 mg/ml) before irradiation.
Effect of PCF on γ-ray-induced lipid peroxidation
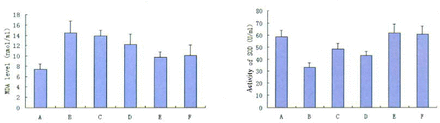
The MDA level as a marker of lipid peroxidation was increased nearly 2 times more in the model group compared to the untreated control group. Also there was another concentration-dependent decrease in the MDA level due to PCF pretreatment. The MDA levels of the PCF-treated groups were 13.97 nmol/L with 1.25 mg PCF/ml, 12.29 with 2.5 mg PCF/ml and 9.75 with 5 mg PCF/ml, respectively (Fig.4).
Effects of PCF on thymocyte membrane damage induced by γ-ray irradiation. The levels of malondialdehyde were measured by the reaction with thiobarbituric acid (described in Materials and Methods). Groups:(A) Untreated contro; (B) Model; (C) 1.25 mg PCF/ml; (D) 2.5 mg PCF/ml;(E) 5 mg PCF/ml;(F) 0.1% vitamin C. Data are represented as the means ± SD of three experiments. *, ** Significantly different from model group at P<0.05 and P<0.01 respectively.
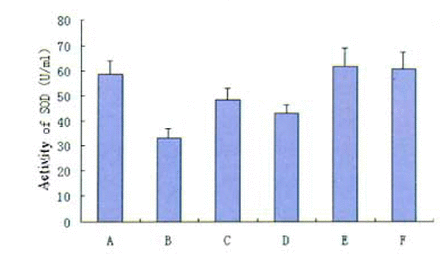
Effects of PCF on the activity of SOD
The activity of SOD was lower in the model group compared to the untreated control group (33.29 ± 2.34 vs 58.64 ± 1.03). PCF pretreatment inhibited the decrease of SOD activity and the activity of SOD in the 5 mg PCF/ml group was even maintained at a normal level (61.48 ± 1.04 vs 58.64± 1.03) (Fig.5).
Changes in the activity of SOD in thymocytes after γ-ray exposure. Groups:(A) Untreated control; (B) Model; (C )1.25 mg PCF/ml; (D) 2.5mg PCF/ml; (E) 5mg PCF/ml; (F) 0.1% vitamin C. Data are represented as the means ± D of three experiments.
*, ** Significantly different from model group at P<0.05 and P<0.01 respectively.
Discussion
In this study, our results showed that in vitro PCF pretreatment of thymocytes from Kunming mice 2 h before irradiation could attenuate the cytotoxic effect of irradiation, protect the cells from apoptosis, alleviate membrane damage and maintain the activity of SOD in a concentration-dependent manner.
Ionizing radiation produces various free radicals that cause cell dysfunction and mortality. [6] Our results showed that the viability of irradiated thymocytes decreased, which is in accordance with previous studies. [18] In groups pretreated with PCF, however, cell viability was enhanced with an increase in PCF concentration indicating attenuation of cytotoxity-induced γ-irradiation. In addition, exposure of thymocytes to γ-irradiation induced cellular apoptosis, [9,18,19,20,23] demonstrated by cell shrinkage, blebbing of the plasma membrane, chromatin condensation and margination, etc. This morphologic method is the most reliable means to assess apoptosis.[9] The development of a DNA “ladder” at a molecular biochemical level was also used to verify cellular apoptosis. Our results are in agreement with a report of Shevchenko, etc. [20] who demonstrated an increase in DNA fragmentation in thymocytes after irradiation. Surprisingly, we observed that the group pretreated with 5 mg PCF/ml did not show an obvious DNA ladder. Consequently we further examined apoptotic changes using FCM analysis and found that the degree of apoptosis of that group was 16.14%. These data support the view [19] that DNA fragmentation will only be detectable when apoptosis occurs at a level of more than 20% of the total cell population.
Further FCM analysis also revealed that after irradiation the increase of the pre-G1 population paralleled the decrease of the S population (data not shown) and that the higher concentration of PCF correlated with the higher S population and the lower pre-G1 population. IR induces arrest at a number of cell cycle checkpoints including a G1-S, G2-M, and a replication-associated S-phase checkpoint. It is widely assumed that these cell cycle checkpoints function to delay cell cycle progression until the DNA damage has been repaired or critical metabolic events, such as replication and chromosome segregation, have been completed.[21] In results similar to ours, Kastan et al.[22] showed that there was only a G1-S phase arrest. PCF may enhance G1-S phase arrest induced by γ-irradiation, thus allowing repair of cell damage and reduction but not triggering γ-ray-induced apoptosis. Dai et al. [23] reported that intracellular pH was increased during γ-Ray-induced p53-dependent apoptosis of thymocytes, and that this apoptotic effect could be inhibited by an acidic condition of the culture. PCF, whose isoelectric pH is between 6.5-7.0, is an acidic marine product which may exert its protective action via a pH effect.
Membrane damage caused by ionizing radiation is of particular importance because significant changes in structure and function of membranes result in cell death via apoptosis.[24,5] Alcouffe et al.[26] indicated that lipid peroxides induce apoptosis in SMCs, macrophages, activated T lymphocytes and the Jurkat T-cell line Malondialdehyde (MDA), is a commonly measured substance from lipid damage after ionizing radiation resulting from the interaction of free radicals with polyunsaturated fatty acids.[6] In our study, an elevated level of MDA, which followed the γ-irradiation, was an indication of γ-ray-induced damage and PCF pretreatment relieved membrane damage by lowering the level of MDA. Therefore apoptosis induced by membrane damage was reduced.
Radiation injury to living cells is, to large extent, due to oxidative stress.[27] Superoxide dismutase (SOD), is one of several enzymes involved in antioxidative defense. It is well documented that SOD has protective effects in living cells against oxidative radiation damage as SOD degrades superoxides from H2O2 which then is removed by catalase.[28] The activity of SOD was lower in the model group compared to the untreated control group and PCF pretreatment upgraded the activity of SOD, though the values in the PCF treatment group were not higher than the model group. The level of SOD in the group treated with 0.5 mg PCF/ml and the untreated control group showed no significant difference, indicating that PCF treatment maintained the activity of SOD against radiation damage. Previous studies have shown that down-regulation of SOD activity is associated with apoptosis of neuronal cells.[29] Thus, PCF may prevent γ-irradiation-induced apoptosis by maintaining the activity of SOD which scavenges superoxides.
In conclusion, our results showed that PCF pretreatment inhibits γ-irradiation-induced apoptosis of thymocytes. PCF displayed its radioprotective effect by attenuation of cytotoxity, alleviation of lipid peroxidation and maintaining the activity of SOD. These results suggest that PCF may be an effective radioprotector for medical applications. Further studies are required to clarify the optimum doses required for human radioprotection.
Footnotes
This work was supported by the grant of the Science and Technology Bureau of Qingdao City, China (No.04-2-NY-49; 04-2-HH-69).
- ABBREVIATIONS
- PCF:
- Polypeptide from Chlamys farreri
- IR:
- ionizing radiation
- ROS:
- reactive oxygen species
- SOD:
- superoxide dismutase
- LPO:
- lipid peroxidation
- MTT:
- 3—(4, 5-dimethl-thiazol-2-yl)-2,5-diphenyl-diphenyl-tetra-zoliumbromide
- DMSO:
- dimethylsuloxide
- TEM:
- transmission electron microscopy
- FCM:
- flow cytometry
- MDA:
- malondialdehyde
- Received July 9, 2004.
- Accepted December 22, 2004.
- Copyright © 2005 by Tianjin Medical University Cancer Institute & Hospital and Springer