Abstract
OBJECTIVE To study the expression of phosphorylated p38 mitogen-activated protein kinase (p-p38) and uPA and the correlation of their expression with breast cancer clinicopathological characteristics, and to investigate the role of the p38MAPK-signaling pathway in regulating uPA expression in breast cancer cells.
METHODS Immunohistochemistry (S-P) was used to test the expression of p-p38 and uPA in 60 specimens of breast cancer tissues. Western blots were adopted to detect expression of the p-p38 and uPA proteins in MDA-MB-231 and MCF-7 breast cancer cells, and uPA expression after treatment with SB203580, a specific inhibitor of p38 MAPK.
RESULTS The positive rate of the p-p38 protein and uPA protein expression in the breast cancer tissues was 56.7% and 60.0%,respectively. The expression of p-p38 was positively related to the expression of uPA (r = 0.316, P < 0.05). The expression of p-p38 and uPA was related to lymph node metastasis and the TNM stage (P < 0.05), but it was not related to the patient’s age or tumor size (P > 0.05). The expression of p-p38 and uPA in MDA-MB-231 cells was higher than that in MCF-7 cells. SB203580 inhibited the p38 MAPK pathway and reduced uPA protein expression.
CONCLUSION The p38 MAPK-signaling pathway promotes breast cancer malignant progression by up-regulating uPA expression, and it may be an important process in breast cancer invasion and metastasis.
keywords
Introduction
Although combined therapy of breast cancer has advanced over the last several decades, over 30% of breast-cancer patients die due to a recurrence and metastasis. Therefore an understanding of the detailed mechanisms of invasion and metastasis in breast cancer would be helpful in improving treatment. Since breast cancer invasion and metastasis are multifactorial processes, studies related to these processes have received current emphasis.
uPA is a serine protease and, when bound to its receptor, uPA initiates the activation of MMPs as well as the conversion of plasminogen to plasmin. uPA confers the ability for cells to degrade the extracellular matrix. Up to now, a growing body of evidence has shown that over-expression of the urokinase-type plasminogen activator (uPA) can be detected in various malignant tumors, and that it plays a key role in tumor invasion and metastasis. However, there is a need to demonstrate the molecular regulatory mechanisms for uPA expression. p38 MAPK is an important member of mitogen-activated protein kinases (MAPKs). The MAPKs have been shown to transduce extracellular signals into cellular responses and play important roles in cell proliferation, apoptosis, differentiation, cell migration, and cytoskeleton remodeling. Recently, it has been suggested that the p38MAPK-signaling pathway is involved in the process of tumor invasion and metastasis by a positive regulatory mechanism. Therefore, we investigated the expression of p-p38 and the uPA protein in breast cancer tissues and cells, as well as the effect of the p38 MAPK-signaling pathway on the expression of uPA. Our studies suggest that an inhibitor of p38 MAPK might be a potential therapeutic target for anti-breast cancer treatment.
Materials and Methods
Specimens
A total of 60 paraffin-embedded breast cancer tissues were obtained from surgical resections at the Pathology Department of the First Affiliated Hospital of China Medical University from 2001 to 2003 (Shenyang, Liaoning Province, China). According to the 2003 World Health Organization breast carcinoma histological classification criteria, all specimens were invasive ductal carcinomas. Human MDA-MB-231 and MCF-7 breast cancer cells were obtained from the Pathology Department of the China Medical University.
Reagents
DMEM and RPMI 1640 were purchased from Gibco. The first antibodies were mouse monoclonal antibody to phosphorylated-p38 and rabbit polyclonal antibody to uPA. SB203580, a specific inhibitor of p38MAPK, was purchased from Calbiochem.
Immunohistochemistry
IHC was performed according to the indirect streptavidin-biotin-hyperoxidase method, following the manufacture’s protocol. For the negative controls, the primary antibody was replaced by RIPA buffer, but all incubation steps were identical. Previously identified strongly staining tumor tissue sections were used as positive controls. We used an intensity-adjusted scoring system to evaluate the immunostaining indices. The sections were scanned by light microscopy. Five fields were randomly selected and 100 tumor cells in each field were counted. The staining intensity was graded 0~2, corresponding to weak, moderately strong, and intense staining, respectively. With respect to the invasive cell count, < 10% positive cells were scored as 0, 10~50% were scored as 1, and > 50% were scored as 2. By multiplying these two factors, an immunoreactive score ≥ 2 was considered to be positive.
Western blots
The cells were lysed in RIPA buffer, sonicated and centrifuged at low temperature. The protein concentration was measured by the Bradford method and cell lysates electrophoresed in 12% polyacrylamide SDS gel followed by transfer onto polyvinylidene difluoride membranes, protein bands were blocked and incubated with the first and second antibody and visualized with a DAB kit. Protein contents were calculated by densitometry.
Statistical analysis
All statistical calculations were carried out using SPSS 10.0 statistical software. The results were compared using the chi-square and Pearson tests. P < 0.05 was considered statistically significant.
Results
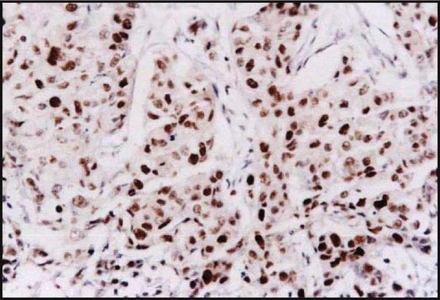
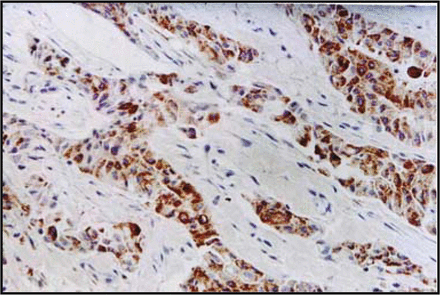
Correlation between p-p38 and uPA protein expression in breast cancer tissues Immunohistochemical results showed that the p-p38 protein was detected in the nucleus of the breast cancer cells. It showed dark staining with a positive rate of 56.7% (34/60) (Fig.1). The uPA protein was expressed in the cytoplasm with dark staining, with a positive rate of 60.0% (36/60) (Fig.2). The uPA protein was expressed positively in 25 of 34 cases, which were also positive for p-p38 protein expression, and 15 cases showed negative expression of the uPA protein in 26 cases with p-p38 negative expression. Statistical analysis suggested a positive relationship between expression of p-p38 and uPA (r = 0.316, P < 0.05).
Expression of p-p38 in breast-infiltrating duct carcinoma showing nuclear staining (S-P method, × 200).
Expression of uPA in breast-infiltrating duct carcinoma showing cytoplasmic staining (S-P method, × 200).
Relationship between p-p38 and uPA protein expression and clinical pathological characteristics
There was a significant correlation between the level of uPA and p-p38 expression and lymph node status and clinical stage (P < 0.05). The level of uPA and p-p38 expression was not significantly related to the patients’age or tumor size (P > 0.05) (Table 1).
Correlation of p-p38 and uPA expression with clinicopathological characteristics of breast cancer.
Expression of the p-p38 and uPA protein in two different metastatic breast cancer cells
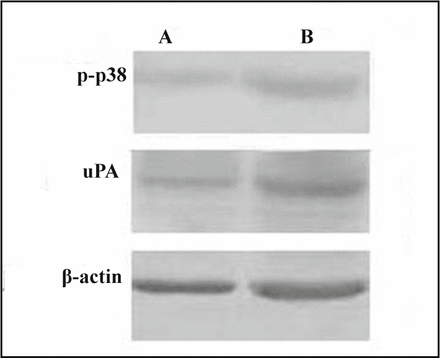
Western blot analysis indicated that the expression of p-p38 and uPA in the highly metastatic MDA-MB-231 cells was higher than that in lowly metastatic MCF-7 cells (Fig.3).
A, MCF-7 cells; B, MDA-MB-231 cells.
The change of uPA protein expression after the p38 MAPK-signaling pathway was blocked by SB203580
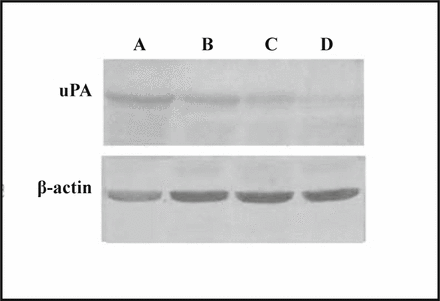
The expression level of the uPA protein in MDA-MB-231 cells decreased after the addition of SB203580, a specific inhibitor of p38 MAPK, was added into the culture medium. SB203580 treatment of the cells reduced uPA protein expression in a concentration-dependent manner (Fig.4).
A, Control; B, C, D, SB203580 2, 5, 10 μmol/L respectively.
Discussion
The MAPKs are serine/threonine kinases, which generally are found in various cells. MAPKs have been shown to transduce extracellular signals involving cell proliferation, differentiation and malignant transformation, and play important roles in tumor generation and development. p38 MAPK is an important member of the MAPK family. p38 MAPK undergoes phosphorylation at both tyrosine and threonine sites and can be activated by a wide spectrum of stimuli, including inflammatory cytokines, growth factors and cellular stress. The p38MAPK-signaling pathway has been implicated in cell growth, apoptosis, cellular motion with an invasive phenotype that mediates cell migration, tumor invasion and metastasis[1-4]. Urokinase plasminogen activator (uPA) is a serine protease that initiates the conversion of plasminogen to plasmin. These proteases confer cellular ability to degrade the extracellular matrix. uPA has been clearly demonstrated to be essential in the maintenance of invasive and metastatic phenotypes[5-9].
At the present time, only a few studies have demonstrated the relationship between uPA expression and a signal-transduction pathway. Recent research has indicated that the p38 MAPK-signaling pathway participates in regulating uPA expression in gastric, lung, leukemic and breast cancer cells[10-13]. Our studies suggest that there is a significant correlation between the level of uPA and p-p38 expression and lymph node status and the clinical stage of breast cancer, and we demonstrated that the expression of p-p38 and uPA in the highly metastatic MDA-MB-231 cells was higher compared to lowly metastatic MCF-7 cells. Our results indicate that over-expression of p-p38 and uPA might be related to breast cancer malignant progression, invasion and metastasis. Immunohistochemical results showed a positive relationship between the expression of p-p38 and uPA, suggesting that uPA protein expression might be regulated by the p38 MAPK-signaling pathway. We also investigated the relationship between uPA expression and the p38 MAPK-signaling pathway. Western blot analysis showed that SB203580 treatment of the MDA-MB-231 cells decreased uPA protein expression in a concentration-dependent manner, demonstrating that the p38 MAPK-signaling pathway might be involved in regulating uPA expression.
In summary, we suggest that the p38 MAPK-signaling pathway promotes breast cancer malignant progression, invasion and metastasis. Although the precise mechanism remains unknown, an inhibitor of the MAPK-signaling pathway represents a novel target for cancer intervention strategy. Our study suggests that the p38 MAPK-signaling pathway may be a potential therapeutic target for breast cancer treatment.
- Received October 18, 2007.
- Accepted February 28, 2008.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer