Abstract
Fatty acid synthase (FAS) attracts more and more attention recently as a potential target for metabolic syndrome, such as cancer, obesity, diabetes and cerebrovascular disease. FAS inhibitors are widely existed in plants, consisting of diversiform compounds. These inhibitors exist not only in herbs also in many plant foods, such as teas, allium vegetables and some fruits. These effective components include gallated catechins, theaflavins, flavonoids, condensed and hydrolysable tannins, thioethers, pentacyclic triterpenes, stilbene derivatives, etc, and they target at the different domains of FAS, showing different inhibitory mechanisms. Interestingly, these FAS inhibitor-contained herbs and plant foods and their effective components are commonly related to the prevention of metabolic syndromes including fat-reducing and depression of cancer. From biochemical angle, FAS can control the balance between energy provision and fat production. Some studies have shown that the effects of those effective components in plants on metabolic syndromes are mediated by inhibiting FAS. This suggests that FAS plays a critical role in the regulation of energy metabolism, and the FAS inhibitors from plants have significant potential application value in the treatment and prevention of metabolic syndromes.
keywords
Introduction
Diseases associated with metabolic syndrome, such as cardio-cerebral vascular diseases, diabetes and cancer, have been the biggest killers of humans. All these diseases gradually develop over a relatively long period, and when clinical symptoms are observed, are usually difficult to treat. Therefore, early prevention of these diseases is vital. Obesity, in particular, central obesity, is the typical representative of metabolic syndrome. It has been established that obesity is one of main causes of cardio-cerebral vascular disease and fatty liver, and that obesity is the strongest risk factor for type 2 diabetes[1]. Unexpectedly, obesity is also highly correlated with cancer incidence. In 2003, after a 16-year follow-up of 900,000 U.S. adults, Calle and colleagues reported that body-mass index was positively correlated with cancer mortality[2].
Recently, fatty acid synthase (FAS), a very important enzyme involved in lipid metabolism, has attracted increasing attention from researchers. FAS catalyzes the synthesis of long-chain fatty acids, mainly palmitates[3]. The structure and function of FAS is complex, and a 3.2 nm resolution crystal structure of mammalian FAS was not reported until 2008[4]. Each peptide monomer of the mammalian FAS homodimer contains seven functional domains. Although it has been known for a long time that FAS is responsible for the synthesis of fatty acids, the effects of FAS on the regulation of metabolism remained largely unclear until recently. In 2000, Loftus and colleagues reported that the accumulation of the FAS substrate malonyl-CoA after FAS inhibition was an important intermediate that suppressed expression of the key orexigenic peptide neuropeptide Y (NPY) to inhibit appetite and thereby decrease the food intake and weight of obese mice. These results showed that FAS was centrally involved in the control of food intake, and revealed FAS as a potential target of anti-obesity drugs[5]. Our group previously reported that FAS activity positively correlated with body fat level in fowl, suggesting a close connection between FAS and obesity[6].
The obesity of mammals usually results from an increase in number and/or size of adipocytes. Adipocytes develop from mesenchymal stem cells of bone marrow, accompanied by a morphological change incell shape[7]. During development, these cells turn from spindle-shaped preadipocytes into spherical mature adipocytes that accumulate lipids. This development is regulated by a series of transcription factors, such as peroxisome proliferator-activated receptor γ (PPARγ) and CCAAT/Enhacer-binding protein (C/EBPα). These factors upregulate the expression of the genes involved in the synthesis of fatty acids, such as FAS[8]. The inhibition of FAS by FAS inhibitors or siRNA reduces lipid droplets of adipocytes and decreases the mRNA level of C/EBP and PPARγ. Therefore, during adipocyte development, FAS possibly plays very important roles that are not limited to fatty acid synthesis[9].
In addition, FAS is hyper-expressed in many cancers, and its inhibition leads to cell death[10]. Therefore, FAS is emerging as a potential therapeutic target for these cancers, although the mechanism still remain to be explored.
In view of the importance of FAS in human diseases, the safe and effective FAS inhibitors could be promising drugs to treat these diseases. Many herbs and functional plant-derived foods have been found to prevent and relieve metabolic syndrome during long-term use, and their active ingredients, such as tea polyphenols, resveratrol, flavonoids, allicin, proanthocyanidins and ursolics, have been indentified and found to possess the corresponding functions. These antioxidant compounds are different from each other in structure, but are typically thought to be beneficial due to their ability to scavenge free radicals. Generally metabolic syndrome results from long-term disorders in energy metabolism, however, so far there has been no report showing how free radicals affect energy metabolism. Do other mechanisms underlie the effects of these healthful plant-derived foods and their active compounds on metabolic syndrome? A series of studies have shown that all these active compounds have one thing in common: effective inhibition of FAS.
FAS inhibitors in plants
In the last century, cerulenin was the only known FAS inhibitor, but it was toxic to cells[11]. In 2000, Kuhajda and his colleagues reported one synthetic FAS inhibitor, C75[12], without apparent toxicity, but afterward it was found that C75 was obviously toxic when peripherally used at increasing concentrations. Therefore, the development of effective FAS inhibitors with low toxicity gained attention from researchers. Since 2000, our group successively reported various types of natural FAS inhibitors present in plants. They had very different molecular types and structures, and came from various species of plants. However, most of these FAS inhibitors could prevent or attenuate metabolic syndrome.
Esterified catechins in green tea
Epigallocatechin gallate (EGCG), one of the major bioactive compounds in green tea, was the first natural botanical compound found to inhibit FAS[13]. Green tea consumption has anti-obesity, lipid-lowering and anticancer effects, and has also been recommended as a healthy drink by the World Health Organization. Catechin polyphenols are the main bioactive constituents in green tea, among which EGCG is the richest one and has a very high antioxidant capacity and a number of physiological activities. Kao and colleagues reported that feeding of EGCG to mice led to the loss in their weight, but the mechanism remained to be explored[14]. Our experimental results showed that EGCG showed both reversible fast-binding and irreversible slow-binding inhibition of FAS. Differing from C75 that targeted the ketoacyl synthase domain of FAS, EGCG mainly acted on the ketoacyl reductase domain. Kinetic analyses indicated that EGCG inhibited FAS competitively with NADPH, and thereby, EGCG possibly bound to the binding-site of NADPH in FAS due to the structural similarity between EGCG and NADPH[13]. Actually, not only EGCG but also all the esterified catechins showed potent inhibition to FAS, and their interaction with FAS was closely related to their ester bond whose highly electrically positive carbon atom played a critical role. Non-esterified catechins did not strongly suppress FAS[15]. Extracts from different kinds of green teas showed various inhibition of FAS with a five-fold difference in their IC50 values[16]. Another esterified catechin, catechin gallate (CG), inhibited FAS 10-fold more potently than EGCG[17], but its content varied significantly in different kinds of green teas. This possibly accounted for the variant inhibition of green teas to FAS. Interestingly, in animal experiments, green tea extracts with stronger FAS inhibitory activities exerted better appetite-repressing and weight-reducing effects. This suggested that the anti-obesity function of green tea was possibly related to their inhibitory abilities of FAS[16].
Tea pigments in fermented tea
During the fermentation of tea, catechins areoxidized and polymerized to tea pigments, among which the types and structures of theaflavins that resulted from polycondensation of two catechins were relatively clear. Now the question is: does fermented tea still have the ability to inhibit FAS? Our results showed that two most widely-used black teas (fermented) in China, brick tea and Pu’er tea (semi-fermented) showed very good inhibition of FAS, more potent than green tea. The main bioactive components, theaflavins, of black tea suppressed FAS more potently than EGCG, by an order of magnitude. Similar to the esterified catechins, theaflavins also exerted both reversible and irreversible inhibition to FAS, and mainly targeted the ketoacyl reductase domain of FAS[18]. Extracted with the optimal solvents, black tea did not show better inhibition to FAS than other teas, but it inhibited most when extracted with boiled water[19]. This is to say, when we drink tea in boiling water, black tea is the best one based on their FAS inhibitory ability. Therefore, the fermented black tea is potentially promising in human health. Our animal experiments also showed that black tea had better anti-obese, appetite-suppressing and lipid-lowering effects on mice than green tea[18]. In the application in human health, black tea is not acknowledged as widely as green tea is. This is possibly due to many more studies on green tea with positive results than on black tea. Actually, compared with green tea, fermented black tea probably possesses even better protective effects on human health.
Herbs and their active flavonoids
We had reported that the extracts of a variety of Chinese herbs were able to potently inhibit FAS, decrease body weight and suppress appetite in mice. These herbs included parasitic loranthus[20], polygonum multiflorum[21], galangal[22], ginkgo leaf[23], night kodo[24], maple leaf[25], and others. The detailed active ingredients in these herbs still remained to be determined, but it was clear that some flavonoids present in these herbs showed potent inhibition of FAS. Epidemiological studies showed that high intakes of flavonoids decreased the mortality of coronary heart disease by 65%, and also reduced the incidences of stroke, lung cancer, rectal cancer, asthma and chronic obstructive pulmonary diseases[26]. Our previous results showed that quercetin, kaempferol, morin, isorhamnetin and rutin exhibited a range ofinhibitory activity toward FAS. The most potent one only had an IC50 of 1 μg/mL (about 2 μM), and it strongly and reversibly repressed the activity of FAS but did not show significant irreversible inactivation of FAS. Contrary to tea polyphenols, these flavonoids targeted the acyltransferase domain of FAS.
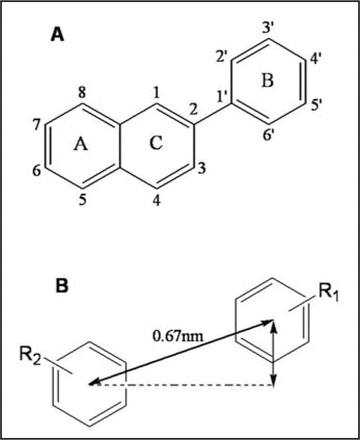
The variantly inhibitory activities of FAS by flavonoids were related to their structures, and the analysis of structure-activity relationship indicated: i) the hydroxyl group in B ring of flavonoids was necessary, the 7’-OH was very important and the two hydroxyl groups were the best; ii) the A ring and B ring should be in the same plane, and the distance between the centers of A ring and B ring should keep proper; and iii) 3’-OH in the C ring was not critical to FAS inhibition[27]. Based on these data, a common pharmacophore for polyphenol FAS inhibitors was put forward as shown in Fig.1, and this was further comfirned by our reruslts[28,29].
Flavonoid structure. A. Structure of flavonoid; B. Pharmacophore structure of polyphenol FAS inhibitors.
It is interesting to note that the herb extracts may contain other FAS inhibitors that are more potent than flavonoidsand remain to be characterized.
Thioethers in Allium plants
Most plants in the Liliaceae are edible, and due to their flavormany of them are used as dietary spices, such as green onion, garlic, allium tuberosum and onion. Recently, interest has been building in many Allium plants that may have preventive and therapeutic effects on cardio-cerebral vascular diseases, hypertension, hyperlipidemia, cancer and other diseases. For instance, dietary garlic can significantly lower blood cholesterol and triglyceride level in hyperlipidemic patients[30]. Onion can help reduce triglyceride level as well as act as a cancer preventative[31,32]
Sulfur compounds are widely present in Allium plants, and their precursor is S-alkyl-cysteine sulfoxide or S-alkenyl cysteine sulfoxide that is converted to thioether or ajoene via a series of reactions by alliinase after cell disruption[33]. It was demonstrated that the extracts of many Allium plants exhibited strong inhibition of FAS. For instance, petroleum ether extracts of green onion and garlic, and ethyl acetate extracts of onion inhibited FAS with IC50 values less than 5 μg/mL[34]. Diallyl trisulfide, commonly known as allitridin, is the most potent inhibitor of FAS among the thioether compounds. Thioether compounds irreversibly modified the essential sulphydryl group in both β-ketoacyl synthase and acyl-carrier protein domains of FAS through a thiols exchange reaction. They inactivated FAS by affinity-labeling, which differs from other FAS inhibitors in the inhibitory mechanism. This may allow thioether compounds to specifically inactivate FAS at lower concentrations that do not affect the activities of other enzymes with essential sulphydryl groups[35].
Diallyl trisulfide strongly inhibited both differentiation and lipid accumulation in 3T3-L1 preadipocytes, perhaps reflecting anti-obesity and anti-hyperlipidemic effects. Diallyl disulfide inhibited FAS much more weakly than diallyl trisulfide, and its suppressive effect on the development of 3T3-L1 cells also decreased. The weakest FAS inhibitor, dimethyl disulfide, had almost no influence on 3T3-L1 differentiation[35]. This suggests that the effects of thioesters on differentiation and lipid accumulation in preadipocytes may correlate with their inhibition of FAS.
Tannins in plants
Tannins are water-soluble polyphenols that are widely distributed in plants, and include condensed and hydrolysable tannins. One of the basic properties of tannins is to non-specifically bind to and aggregate proteins, such as enzymes, receptors, hormones and other biological factors, that are the most common drug targets. The non-specific interaction of proteins with tannins will disrupt the function of drugs. Besides, tannins bind irreversibly to chromatographic resins used to purify secondary products. Therefore, tannins are often removed in the first isolation step in the purification of bioactive constituents from plant tissue. Surprisingly, it was found that condensed as well as hydrolysable tannins are strong FAS inhibitors. We separated dimeric and trimeric condensed tannins from the traditional Chinese medicine catechu. Both of them exhibited potent inhibition of FAS. Trimeric condensed tannin inhibited FAS with IC50 of only 0.47 μg/mL, and moreover it also showed a strong time-dependent inhibition of FAS[36]. The kinetic analyses showed that trimeric condensed tannin inhibited FAS competitively with respect to the substrate acetyl-CoA with an inhibition constant Ki of only 0.40 μg/mL. This suggested that condensed tannins mainly acted on the acyl transferase domain of FAS, which was similar to flavonoids. Trimeric condensed tannin inhibited the β-ketoacyl reduction reaction of FAS competitively with respect to NADPH with a Ki of 1.55 μg/mL, and also showed a time-dependent inhibition on this reduction. This was similar to EGCG. In addition, condensed tannins exhibited strong inhibitory effects on the growth of human breast cancer cells and further results showed that the suppression of cancer cells by condensed tannin may be related to their ability to inhibit FAS. Notably, the concentrations of condensed tannin required for the inhibition of both FAS and cancer cell growth was much lower than those for FAS aggregation[36], which suggested that the biological activities of tannins are not related to their ability to induce aggregation of protein. Hydrolysable tannins also showed strong inhibition of FAS. Two typical hydrolysable tannins, tannic acid and ellagic acid, inhibited FAS with IC50 values less than 1 μg/mL and also exerted a time-dependent inactivation of FAS (Tian’s unpublished data).
Pentacylic triterpenoids, chlorogenic acid and stilbenes in fruits
Both ursolic acid and oleanolic acid are the members of the pentacylic triterpenoid family, and they are very commonly active components in fruits. It was found that ursolic acid has protective effects on hyperlipidemia[37], diabetes[38], cancer[39] and canker[40]. Ursolic acid shows strong inhibition of FAS with an IC50 of 6 μg/mL, and it acts mainly on the acyl transferase domain of FAS and also weakly on the β-ketoacyl synthase domain. Kinetic studies showed that ursolic acid inhibited the overall activity of FAS competitively with respect to both substrates, acetyl-CoA and malonyl-CoA, suggesting the binding-site of ursolic acid was related to those of both substrates. Ursolic acid inhibited FAS overall reaction rate uncompetitively against NADPH, but it was competitive with NADPH for the same binding-site of FAS in the β-ketoacyl reduction reaction. In addition, ursolic acid at high concentrations (> 25 μg/mL) inactivated FAS very rapidly. The second rate constant of the inactivation of FAS by ursolic acid was higher than that by C75. However, ursolic acid at low concentrations only inactivated FAS very weakly, and the sigmoid curve of second rate constants versus concentrations suggested the presence of a positive cooperativity. Interestingly, in the presence of NADPH, the sigmoid curve switched to a hyperbolic curve characterized by affinity labeling kinetics. This meant that NADPH significantly enhanced the inactivation of FAS by low concentrations of ursolic acid, and this inactivation took place through a two-step affinity labeling reaction: first ursolic acid reversibly bound to FAS and then inactivated it. Combined with the results that ursolic acid was an uncompetitive inhibitor of FAS over reaction against NADPH, it could be concluded that the binding of NADPH to the β-ketoacyl reductase domain probably caused a conformational change in FAS which promoted the binding of ursolic acid to acyl transferase. Similar to NADPH, ursolic acid itself was able to bind to β-ketoacyl reductase domain of FAS to increase its inactivation of acyl transferase and thus showed the positively cooperative effect. This speculation was further confirmed by the results that ursolic exerted positively cooperative effect at aconcentration around 20 to 30 μg/mL that was comparable to the IC50 value (27.1 μg/mL) for the inhibition of β-ketoacyl reductase by ursolic acid[41]. These results indicated that FAS shared some characteristics with allosteric enzymes to some extent, which surely would enhance the possibility of FAS as a key target for regulation of energy metabolism.
Chlorogenic acid, an ester of caffeic acid and quinic acid, is another commonly active constituent of fruits. It exhibited almost the same inhibition of the overall reaction and β-ketoacyl reduction of FAS with an IC50 of 33.6 μg/mL. Chlorogenic acid inhibited the β-ketoacyl reductase of FAS competitively with respect to NADPH, which suggested it possibly acted on the binding-site of NADPH on β-ketoacyl reductase. However, chlorogenic acid also affected the activity of enoyl reductase of FAS that also used NADPH as substrate. This indicated that the two binding-sites of NADPH are different from each other[42]. Although chlorogenic acid did not inhibit FAS very potently, it still was valuable to study due to its abundance in plants.
We found that stilbene glucoside, one member of stilbene family, in Chinese herb tuber fleeceflower root was a FAS inhibitor[43]. Another stilbene compound, resveratrol, rich in grape skin and seed, also was found to inhibit FAS. Recently, resveratrol has been gaining increasing attention, since it was reported to potentially prevent cardiovascular diseases and cancer and to delay aging[44]. The kinetic results showed that resveratrol reversibly inhibited FAS with an IC50 of 11.5 μg/mL, and mainly targeted the β-ketoacyl reductase domain of FAS. In addition, extracts from grape skin and seed showed very highly inhibitory activity on FAS while that from grape flesh only had marginal effects on FAS, which was consistent with their resveratrol content (Tian’s unpublished data).
The extracts from many plants that were able to prevent metabolic syndrome inhibited the activity of FAS. Is it just a coincidence? Moreover, the components that were active as FAS inhibitors differed in structure and chemical properties. The classical explanation for how they are healthful to humans is that they have antioxidant properties. However, there is so far no obvious evidence mechanistically linking antioxidation and energy metabolism. The fact that most of their active components can inhibit FAS may provide a more reasonable explanation. Also, all these results support the hypothesis that FAS is a critical regulator in energy metabolism.
Analysis for energy metabolism and roles of the substrates of FAS in energy metabolism
The role of FAS in the balance between energy provision and lipid production in vivo
Metabolic syndrome may be considered to be rooted in long term decompensation in energy metabolism. Energy metabolism is very complicated and affected by many factors. Generally, decompensation results from energy accumulation due to an imbalance between energy intake and production. Initially, it was thought that decompensation simply resulted from over-intake, and thereafter, diet (caloric restriction) became a popular way to control body weight. Unfortunately, simple intake-reduction is not very effective, and sometimes it causes health problems. It is often observed that people’s natural appetites are very different from each other. Some people eat a lot but do not become obese. In contrast, some gain or maintain a lot of weight even though they are on a restricted diet. One common phenomenon is that many young people eat a lot but are free from obesity, while middle aged easily become obese even with decreasing appetite. It has been suggested that food intake is not the only cause, even not the main cause in some conditions, of the decompensation of energy metabolism. The in vivo homeostasis in energy metabolism could be very critical. The balance between energy provision and fat production is very important for energy homeostasis, and thus deserves attention. Actually this balance is the homeostasis between energy storage and production. A possible problem of dieting is that the necessary energy provision for the body is not enough, and in this situation, the self-protective response of the body should be to either increase appetite or decrease energy production. This frequently results in a failure to control weight following initially successful weight-reduction on a restricted diet. If we can adjust the metabolic balance in energy, shift from energy storage to production, and thus keep the body energetic, we may increase the possibility for successful dietary weight reduction, because under this condition, reduced food intake will be accompanied by increasing energy production. FAS is a critical enzyme for synthesis of fatty acids, and these fatty acids and their derivatives are the main forms of energy storage. Therefore, the inhibition of FAS will suppress the energy storage and increase energy consumption by forcing its substrate, acetyl-CoA, to enter citric acid cycle. This regulation by FAS could be reasonable based on the analyses for the roles of three substrates of FAS, malonyl CoA, acetyl CoA and NADPH, in energy metabolism.
The role of malonyl CoA: why FAS is a more suitable regulator in energy metabolism than Acetyl CoA Carboxylase (ACC)
Many years ago, feeding was reported to significantly regulate the expression of FAS. Feeding with carbohydrate up-regulated the expression of FAS, while feeding with fat down-regulated it[45]. Afterward, it also was found that the converse regulation also existed. In 2000, Loftus et al.[5] reported that the inhibition of FAS reduced food intake of obese mice. The accumulated malonyl CoA from FAS inhibition was thought to be a very important mediator, and it suppressed the expression of neuropeptide Y in the hypothalamus to reduce the appetite of mice. Interestingly, the activities of mice were not affected by FAS inhibition, while the body weight decrease significantly. Therefore, it was proposed that FAS is highly associated with the regulation of appetite[5].
Food intake is the original source for in vivo energy and thus vital for the homeostasis of energy. Feedback between appetite and FAS is a normal physiological process in the body, and thus it does not produce side-effects. Therefore, the control of weight by regulating FAS should be different from that on simple diet that may be accompanied by health problems.
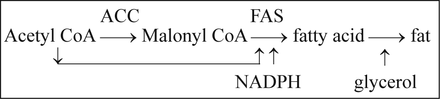
Let us take a look at the process of fat synthesis:
As the first enzyme in the process of fat synthesis, ACC was first paid the most attention by researchers. It was found that ACC indeed showed some effects on regulation of the fat synthesis, but the effects were very limited. Sometimes, ACC showed converse effects on the regulation of the whole energy metabolism. For example, FAS inhibition reduced food intake and body weight of mice, but it was reversed by the combination with ACC inhibition. It showed that ACC plays an opposing role in the regulation of fat synthesis compared with FAS.
The key point is malonyl CoA. Malony CoA is the substrate of FAS and product of ACC, and thus it accumulates with FAS inhibition, and decreases with ACC inhibition, which results in the reverse regulation of ACC. Malonyl CoA has been demonstrated to be a signal molecule in the regulation of food intake5 and fatty oxidation[46]. Many acyl-CoAs are important intermediates in energy metabolism, but malonyl CoA is special due to its negatively charged carboxyl group. This makes malonyl CoA suitable for a signal molecule. Therefore, FAS may be more suitable for regulation of energy metabolism than ACC.
The role of acetyl CoA in the energy metabolism
Acetyl CoA is another substrate of FAS, and it is also the substrate of ACC at the same time. Inhibition of FAS directly decreases incorporation of acetyl CoA into fatty acids, in the meantime, since malonyl CoA is the product of ACC, the accumulated malonyl CoA resulting from FAS inhibition suppresses the activity of ACC by end product inhibition, which further reduces the consumption of acetyl CoA. In a word, the inhibition of FAS blocks the incorporation of acetyl CoA into fat.
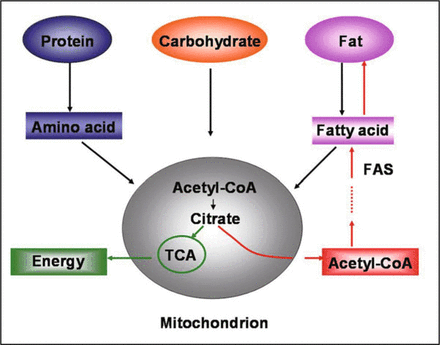
Although the whole system of energy metabolism is intricate, the central pathway of energy metabolism now is clear: after the initial processes of metabolism, fat, carbohydrate and some amino acids are finally converted to acetyl CoA. Acetyl CoA is the primer of citric acid cycle that is the major source for energy, at the same time it is the precurser of fat synthesis. Therefore, acetyl CoA is the undertaker of most of the ingested energy materials, and it serves as a very important regulator in the energy metabolism to decide the balance between energy storage and production, as shown in Fig.2.
Energy metabolism sketch. Green route shows energy producing; Red route shows fat producing.
By entering the citric acid cycle, acetyl CoA can promote the production of energy. FAS may be the best regulator to decide the fate of acetyl CoA: to energy storage (fat synthesis) or to energy production (enter citric acid circle).
The role of NADPH
FAS uses NADPH as a coenzyme to synthesize fatty acids, and therefore, NADPH may be considered a substrate of FAS. For synthesis of one mole of palmitate, FAS converts 14 moles of NADPH to NADP+. The ratio of NADPH and NADP+ reflects the redox status of body. Therefore, FAS is also very important to maintain redox balance.
NADPH is a basic high-energy molecular in vivo. The energy required for the synthesis of fatty acid by FAS mainly comes from NADPH and some from malonyl coenzyme A. NADPH is also used as an essential coenzyme and provides energy for many other biosyntheses. However, the synthesis of fatty acid is biggest consumer of NADPH. NADPH does not directly enter the oxidative phosphorylation pathway to produce ATP, but the restoration of NADPH requires energy. From the biochemical angle, one NADPH is equal to one NADH or three ATP for energy metrology. Therefore, the status of NADPH plays a very important role in the regulation of energy metabolism, and reduced consumption of NADPH by FAS should enhance energyproduction.
Taken all together, the inhibition of FAS results in the accumulation of its three substrates, and should result in the production of more energy. This can explain why the activity and metabolism of mice remained high even when these obese mice were injected with FAS inhibitors. Apparently, this effect cannot be produced by diet to control weight that is, of course, accompanied by the decreased metabolic activity. Therefore, restricted diet alone may often fail to reduce body weight. FAS can regulate energy metabolism via several pathways, and is a global regulator in energy metabolism.
The feasibility of FAS as a target of regulation in energy metabolism
The tissue specificity renders FAS as an available and safe target in mammal
FAS expression is not widely distributed in mammalian tissues: it is only expressed in a few tissues, mainly in liver and fatty tissue. Is this disadvantageous for FAS as a target in energy metabolism? FAS has not been considered a good target for regulation because of its absence in most of tissues. Actually this is not the case after carefully analyses. Why does FAS express highly in liver and fatty tissue? Do the liver and fat cells need so much fat for survival? Our results showed that FAS inhibitors fed to rats or mice reduced their appetite and body weight, and also suppressed their liver FAS activity. However, during the experiments, all these animals behaved normally, and their livers were normal after experiments. When treated with FAS inhibitors in vitro, fat accumulation in 3T3-L1 adipocytes was significantly reduced, but these cells were still alive and kept their normal morphology. This demonstrated that the high level of FAS in liver and fat is not necessary for their survival. So it is most likely that both liver and fatty tissues are responsible for the provision of fatty acids/fat for the whole body. This division of work refers to synthesis and storage of fat in fatty tissue, and fatty acids in liver, and these fat/fatty acids are provided not only for the liver and fatty tissue but for the whole body. This releases other tissues from the requirement of fatty acid synthesis, and thus from the expression of FAS. Consequently, the regulation of FAS activity should not affect tissues that do not express FAS.
All the above analyses show that proper regulation of FAS activity would only influence on the appetite and the homeostasis between energy and fat production, while leaving the normal function of the whole organism unaffected. This renders FAS as a safe target for the regulation of energy metabolism.
Once excessive fatty acids are synthesized in the liver, they will either be transferred to fatty tissue to be stored as fat that finally leads to obesity, or accumulate in liver to lead to fatty liver. Moderate regulation of FAS activity could attenuate or block the over-synthesis of fatty acids in liver, and in the meantime, it will not influence the normal physiology of the organism. Therefore, FAS is a potentially ideal target for the regulation of metabolism.
The regulation of FAS
As a multi-functional enzyme, the activity FAS can be easily regulated. It had been thought that the condensation reaction of FAS was the critical, rate-limiting step in fatty acid synthesis. It seems reasonable because this step determines the elongation of carbon-chain fatty acids, and all the early reported FAS inhibitors target the ketoacyl condensate of FAS. FAS II in prokaryotes and plants consists of seven separate enzymes and acyl carrier protein, and therefore, the ketoacyl condensase could be critical for the synthesis of fatty acids. However, in animals, all these enzymes are integrated to one peptide due to an evolutionary gene fusion. The whole reaction is catalyzed by a multi-functional enzyme, and no intermediate product is released from FAS. Thus each step in fatty acid synthesis must keep step with each other. This is to say, every step is critical and rate-limiting, and the inhibition of any reaction of mammalian FAS will lead to the blockage of fatty acid synthesis. FAS has seven different functional domains that can be targeted by inhibitors. Besides, the influence on the relative position of each domain in the FAS complex or on movement of ACP-phosphopantetheine with intermediates by compounds will also lead to the inhibition of fatty acid synthesis. Therefore, FAS can potentially have various types of inhibitors. Reported FAS inhibitors act on the KS, MAT, KR, or ER domains of FAS, and some targeted more than one domain. These inhibitors also differed in chemical structure.
Interestingly, the aforementioned ursolic acid, widely distributed in plants, inhibited FAS in a positive cooperative manner, and NADPH also exhibited a heterogeneous positive cooperative effect on the inhibition of FAS: after it bound to the KR domain of FAS, ursolic acid at low concentrations inactivated FAS much faster. Based on these kinetic results, FAS at least shared some characteristics with allosteric enzymes, which is obviously propitious for FAS to be subject to inhibition.
FAS, metabolic syndrome and cancer
Energy metabolism is a complicated system that contains lots of different processes, and thus its regulation inevitably involves many factors. So far, our understanding of the regulation of energy metabolism remains incomplete. This has led to relatively ineffective methods for the prevention and treatment of metabolic syndrome. Based on all the related results and analyses, we propose that FAS plays an important role in the regulation in energy metabolism.
It has been acknowledged that prevention is more important than treatment when dealing with the chronic diseases related to metabolic syndrome, such as cardio-cerebral vascular diseases, diabetes and cancer. As for treatment, more attention should be paid to the whole organism than to focus on concentrated treatment. As mentioned in the above, many FAS inhibitors are natural compounds in plants, among which most are non-toxic and well tolerated. Although the working concentrations of these natural FAS inhibitors do not reach the level required for the clinical drugs, these compounds can be taken at high dosages due to their edibility. Moreover, as foods, these natural FAS inhibitors may be more suitable for the prevention of and recuperation from diseases. Therefore, FAS could be an ideal target for the prevention of metabolic syndrome and associated diseases.
We previously measured the FAS activity of egg-laying chickens during their growth, and found that their liver FAS activity gradually increased as they grew up to middle age, accompanying by the accumulation of body fat[47]. In middle age obesity is a very common phenomenon in higher animals including humans, which should result from natural selection due to survival competition. Getting older and thus weaker, animals need more energy storage to compensate their decreased foraging ability. Now people, especially those in the developed countries, need not to fight for food to survive. Conversely, increasing fat accumulation in the body threatens their lives by inducing metabolic syndrome and associated diseases. Therefore, inhibition of fat accumulation has become a necessary choice for humans to prolong our life spans.
Over the past decades, oncogene revolution prevails in explaining the critical characteristics of cancer cells, although it is clear long time ago that cancers have many specific metabolic behaviors, compared with their normal counterparts. Just in recent years, interest in the topic of tumor metabolism raises and rapid becomes one of the most intense areas of cancer research, because the accumulating evidence shows that the signaling pathways driven by oncogenes finally converge at core metabolism[48]. The dissection of the underlying mechanism possibly provides further understanding and specific potential targets for the treatment of cancers. FAS is a very important enzyme involved in lipid synthesis metabolism, and ectopically expressed in many cancers[10]. This renders FAS as a potential target for cancer treatment, supported by the facts that the inhibition of FAS induces apoptosis in many cancer cells[10]. However, up to now, the mechanism still remains unknown. Initially, it is thought that FAS plays an indispensable role in providing fatty acids for the fast-growing cancer cells, but at present, this plausibly biochemical speculation is in question based on the unexpected results. Although the detailed molecular mechanism needs further exploring, it does not devaluate the potential of using FAS to target cancer cells. The transition of the disordered cell metabolism is possibly critical to tumorigenesis, and the hyperactive FAS might largely contribute to this progress. If so, the safe FAS inhibitors, as presented in the above, should also prevent carcinogenesis. Therefore, the dissection of molecular connection between FAS, metabolic disorder and cancers deserves the further study due to the potential application of FAS in the treatment and prevention of cancers.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received February 15, 2011.
- Accepted March 9, 2011.
- Copyright © 2011 by Tianjin Medical University Cancer Institute & Hospital and Springer