keywords
Introduction
Chronic myelomonocytic leukemia, (CMML) is a clinically rare chronic myeloid leukemia, with an incidence rate of about 1-2/100,000/year, and the age of the predominant cases is over 60 years. The median age of onset is 65-70 years, and the ratio of the incidence between male and female is about 1.5 : 3.1. Specific etiological factors of the disease are not clear, but may be associated with the exposure to ionizing radiation, occupational and environmental carcinogens, or toxins. The FAB (French-American-British) Cooperative Group defined CMML as a subtype of myelodysplastic syndrome (MDS)[1]. Since it has both characters of bone marrow abnormal proliferation and bone marrow dysplasia, recently, the FAB Cooperative Group has classified CMML into myelodysplastic/myeloproliferative diseases (MDS/MPD)[2,3], based on the classification of neoplastic diseases of the hematopoietic and lymphoid tissue. CMML is a malignant-clone disease, which involves bone marrow hematopoietic stem cells, but no specific chromosomal abnormalities are involved. The most common chromosomal abnormalities are +8, -7/7q-, and the structural abnormality is 12p. The chromosomal abnormality found in t (3; 9) (p21; p13) is very rare in hematologic malignancy. So far, only 1 case with acute lymphoblastic leukemia (ALL) has been reported both in China and abroad[4]. A CMML patient with genetic abnormality at t (3; 9) (p21; p13) was admitted and treated in our hospital. The case is reported as follows with a associated literature review.
Case Report
A 75-year-old male was admitted to our hospital for a reducible mass in his left inguinal groove for 2 months. Doctors firstly made diagnosis as left oblique inguinal hernia and prepared to give an operation to the patient. Physical examinations showed mild anemia, no yellow stain of skin and mucus membranes. Superficial lymph nodes could not be touched, and sternum tenderness was negative. There were no abnormalities in heart and lung function, and there was no tenderness and rebound tenderness in the abdomen. Liver could not be palpated along the costal margin, and the spleen enlarged about 2 cm under the lower costal. No edema of the limb extremities was found. X-ray and ECG didn’t show any abnormalities of the target organs, and B-mode ultrasonography of abdomen showed splenomegaly. Blood routine test (BRT) showed white blood cell (WBC) count 17.8 × 109/L, hemoglobin (Hb) concentration 72 g/L, platelet (PLT) 699 × 109/L. Based on the findings of the examinations, such as BRT abnormality and splenomegaly, the patient was given a surgery. The patient was also suggested to receive bone marrow puncture for further diagnosis.
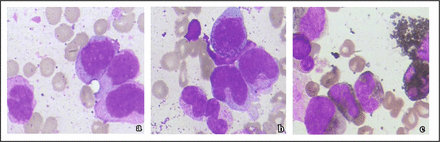
Bone marrow was aspirated about 0.5 mL from posterior superior iliac spine of the patient. The smears were simultaneously stained with Wright-Giemsa method. We observed the findings: obviously active proliferation of the bone marrow, myeloblast accounting for 4.4%, developmental disorder of nucleus and cytoplasm in parts of promyelocytes, pelger deformity, and vacuolus degeneration. The ratio of the eosinophils in the peripheral blood increased to 14.4%. Monocytes accounted for 22.8%, with 8.4% original monocytes and promonocytes. Erythroid proliferation was active, including megaloblastoid change, nomoblasts division, sister nucleus of erythrocytes, 2 and 3 nuclear of erythrocytes. Seventeen platelet-forming megakaryocytes were found around the whole smear. Small megakaryocyte, megakaryocytes with multiple round nucleolus and large platelets could also be seen. Cytochemistry myeloperoxidase (MPO) and alkaline phosphatase (NAP) staining: POX (peroxidase) showed weakly positive; the positive rate of serum NAP was 44%, with labeling index of 144 points. According to FAB classification and morphology, the leukemia patient was considered as CMML-II-type (Fig. 1).
a, The leukemic cells in peripheral blood; b, The leukemic cells in bone marrow; c, POX staining shows weak positive stain in the leukemic cells.
Examination of molecular biology
As CMML was a chronic myeloproliferative disease, BCR-ABL fusion gene and JAK2 gene mutation should be checked. Nested RT-PCR was used for detecting the transcript of BCR-ABL fused gene. Results showed: no BCR-ABL fusion gene transcripts in the sample, which verified the diagnosis of CMML. Real-Time 7500 PCR (ABI) was used to check whether the JAK2 gene had mutation or not, and the detection kit was purchased from Huaguan Shanghai Biochip Co. Ltd. Results showed wild-type JAK2 gene (+), mutant JAK2 gene (-), therefore, the JAK2 gene mutation didn’t occur.
Conventional karyotype analysis
Direct method and 24-h culture for karyotype analysis were used for conventional chromosome preparation, and RHG-banding was used to analyze the chromosome, in which 20 cells of metaphase were analyzed. For image acquisition, the Cycovision Karyotype Analysis System of AI company in the USA was applied, and the identification and description of karyotype abnormalities were based on the International System for Human Cytogenetic Nomenclature (ISCN 2005). The karyotype of the patient was 46, xy, t (3; 9) (p21; p13) (Fig. 2).
a, A metaphase cell stained with Giemsa-banding before analysis; b, Full karyogram of the bone marrow cells: 46, xy, t (3; 9) (p21; p13); c, Partial karyogram of t (3; 9) (p21; p13) in the patient.
Since the diagnosis of CMML, the patient only accepted supportive therapy, such as blood transfusion. Till July, 2009, the patient suddenly presented fever and dyspnea, and was admitted to hospital for the second time. Routine blood test showed that WBC increased to 71.0 × 109/L. Mildly enlarged spleen was detected. The patient refused to have bone marrow aspiration due to his early diagnosis of CMML. The patient accepted chemotherapy, in which hydroxyurea was employed. Two weeks after the chemotherapy, the condition of the patient was obviously improved, with the blood routine test returning to the initial level of preliminary diagnosis. The patient was still under follow up.
Discussion
CMML is a kind of malignant disease of bone marrow clonal hematopoietic stem cell, with both features of myelodysplastic syndrome (MDS) and chronic myeloproliferative disease (MPD). It was firstly classified in MDS in 1982 by French-American-British (FAB) classification system. However, CMML seems out of place from other 4 entities due to the hematologic and morphologic features of CMML varying from predominant myelodysplasis to main myelosis. In 2001, the WHO classification moved CMML from MDS to a new category of mixed MDS/MPD, and also defined CMML I and CMML II according to medullary and peripheral blast counts[1,2]. This new category of CMML made in 2001 WHO was not substantively changed when revised in 2008 WHO[3].
In recent years, more and more genetics researches have focused on CMML. According to current statistic data, about 20%-30% of CMML patients have genetic abnormalities, including abnormalities in chromosome number or structure, for instance +1, +8, del (20q), -7/7q-, del (11q), del (16q), t (5; 12) (q33; p13), t (5; 7) (q33; q11.2), t (11; 16) (q23; p13), t (1; 13) (p36; q21), t (7; 11) (p15; p15), t (8; 9) (p11; q34), et al.[4] and some of which can be rarely seen in myeloproliferative disorder or myelodysplastic syndromes. In addition, some new genetic abnormality has been reported in recent years, such as der (14) t (1; 14) (q12; p11)[5], t (5; 21) (q13; q22)[6], and der (9) t (1;9) (q11;q34)[7]. This CMML case showed a sole abnormality of t (3; 9) (p21; p13), which has never been reported in the literature. This translocation of chromosomes is a very rare genetic abnormality in hematologic malignancies, which was currently found in a case with ALL[8].
The breakpoint of t (3; 9) (p21; p13) generates at 3p21 and 9p13. As far as we know, a variety of tumor suppressor genes have been detected in the region of 3p21, among which DNA mismatch repair genes (MLH1), LIMD1 gene and leucyl tRNA synthetase gene (LARS2) can be commonly seen. The 3 genes are closely related to the occurrence and development of esophageal squamous cell carcinoma, colorectal cancer, lung cancer, and nasopharyngeal carcinoma, respectively[9-11]. In the condition of 3p21 breakage, one or more tumor suppressor genes could be lost, which induces the occurrence of different tumors. The breakage of 3p21 region has been reported in chronic myeloid leukemia with the abnormality of t (3; 9; 22) (p21; q34; q11)[12]. Otherwise, it has been reported that familial melanoma predisposing gene (MLM) is located in the region of 9p13 as a kind of oncogene, which is closely related to the occurrence and development of laryngeal squamous cell carcinoma[13]. The breakage at 9p13 region in ALL patients has been found and reported, and most presently, it has been found and reported in malignant hematological patients[14].
According to the analysis above, the abnormality of t (3; 9) (p21; p13) occurring in this case may lead to losing tumor suppressor genes located in the upper part of short arm of chromosome 3, or lead to increasing the amplification of oncogene located in the short arm of chromosome 9, and all these probably result in the occurrence of CMML in this case. The abnormality of t (3; 9) (p21; p13) could occur not only in ALL patients, but also in CMML patients. Unfortunately, our study is lack of data regarding other cytogenetic abnormalities, such as alteration in the RAS pathway that have been frequently found in CMML[15]. Such information may be helpful in further elucidating the molecular biology of the disease.
This is the first CMML case with t (3; 9) (p21; p13) as a sole abnormality. Although more cases are needed to investigate the molecular pathogenesis of this rare transformation in hematologic malignancy, we suggest that t (3;9) (p21; p13) could be a recurrent chromosomal aberration in myeloid neoplasms, particularly in monocytic-lineage leukemia. Further studies are needed to evaluate the prognosis, survival, and treatment response of the CMML cases with t (3; 9) (p21; p13) abnormality.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received August 29, 2010.
- Accepted October 8, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer