Abstract
OBJECTIVE To explore the relationship between CAR and the development of human lung cancer, as well as to provide the basis for the clinical treatment of lung cancer using an adenovirus vector-based gene therapy.
METHODS CAR expression was assessed immunohistochemically in tumoral, paraneoplastic and normal samples from 112 lung cancer patients. At the same time, the mRNA and protein expression of CAR in 32 cases were determined by RT-PCR and Western blot. The relationship between CAR expression and clinicopathologic parameters was statistically analyzed.
RESULTS There was no expression of CAR in normal lung tissue but a little in paraneoplastic tissue. The positive rate was 43% in squamous cell carcinoma, and 70% in adenocarcinoma. Both were much significantly higher than that in paraneoplastic tissue. The CAR expression level in adenocarcinoma was higher than that in squamous cell cancer. mRNA expression by RT-PCR and protein expression by Western blot were consistent with immunohistochemistry results.
CONCLUSION CAR is overexpressed in human lung cancer, especially in adenocarcinoma. This data offer the reliable basis for adenovirus-mediated gene therapy of lung cancer; more important, CAR may take part in the formation or development of lung cancer; this may be exploitable for the development of antibody-directed therapy in human lung cancer.
keywords
- coxsackie virus and adenovirus receptor protein (CAR)
- lung cancer
- immunohistochemistry
- RT-PCR
- Western blot
- gene therapy
Introduction
Coxsackievirus and adenovirus receptor (CAR)[1,2] is a cell-cell adhesion protein and high-affinity viral receptor in normal epithelial cells[3]. It is a transmembrane receptor of the immunoglobulin superfamily which shows tissue specificity and species specificity[4], and has some relationships with age[5]. CAR has primarily been studied in its role as the initial cell surface attachment receptor for Coxsackie B (CVB) and adenovirus (Ad). This cell surface receptor plays a crucial role in CVB and Ad entry into host cells[1]. CAR mediates homotypic intercellular interactions, while in polarized endothelial cells CAR is closely associated with tight junctions, where it contributes to the barrier of the paracellular flow of solutes and macromolecules[6]. A strong correlation of CAR levels with the viral sensitivity of several cell types has been reported[7-9]. In fact, CAR has been shown to be a docking site for Ad, thus acting as a key receptor for the enhancement of the virus-to-host affinity and the initiation of the virus internalization to the host cell[10,11]. On cells lacking CAR, virus uptake takes place with lower efficiency[10,12] due to the existence of a secondary pathway leading to the viral internalization[10,13].
The very promising use of Ad vectors in gene therapy, since Ads are relatively safe, highly infectious, and capable of delivering therapeutic genes to different cell types[13,14], and still faces a critical prerequisite, which is no other than the identification of highly efficient and accurate systems for delivering the therapeutic genes into target cells[15]. In this regard, CAR expression could be a surrogate marker for monitoring and/or predicting the outcome of gene therapy, while by increasing CAR levels, resistant cells could become more sensitive to Ad infection[16]. Adenovirus-mediated wild-type p53 gene replacement is an attractive therapeutic strategy for various human cancers, and clinical trials of this new therapy are currently being conducted in many countries[17].
However, only a limited number of studies concerning CAR expression have been conducted on clinical tissue. In this aspect, Persson et al.[18,19] presented an immunohistochemical study in human normal brain and human brain tumors, suggesting that neuroblastomas and medulloblastomas could be suitable for adenovirus-mediated gene therapy. Moreover, recent studies have suggested a pathophysiological role for CAR in urogenital cancer and glioma cells, rendering CAR as a membrane receptor which conveys its signal into the nucleus and results in cell proliferation suppression[5,20-21]. However, some research[22-25] has shown that CAR is in favor of non small cell lung cancer in vitro or that high levels of CAR expression are detected in lung cancer. It is very difficult for people to know CAR’s role in gene therapy and tumor development well from the contradictory results. These findings raise the question of whether CAR expression could be related to tumor proliferative capacity or differentiation amongst the different tumor cell types.
For a better understanding of the effect of CAR expression in lung cancer carcinogenesis and development, on the grounds of cell and in vitro research. In this study, we analyzed CAR expression in lung normal tissue, paraneoplastic tissue and carcinoma tissue by immunohistochemistry, RT–PCR and Western blot, to supply a promising option for lung cancer diagnosis and gene therapy.
Materials and Methods
Clinical data
A total of 112 cases with lung cancer from January 2006 to December 2006 were selected, including 81 male and 31 female patients. The age of the patients ranged from 40 to 78 years, with an average age of 62.25 years. According to 2004 WHO Lung Cancer Classification, there were 65 squamous cancers and 57 adenocarcinomas. The specimens from the patients were approved by the local ethics committee.
Tissue samples
The study protocol was approved by the ethics committees of our institutions. Samples of normal lung tissue, paraneoplastic tissue and cancer tissues were obtained from 32 patients (22 men and 10 women) receiving surgical treatment. Each tissue specimen was divided into 2 pieces; 1 sample was immediately frozen in liquid nitrogen at the time of surgery and stored at -80°C until RNA extraction was performed. The other sample was processed for pathologic examination. Because preliminary examination revealed that necrotic tumor decreased the expression of CAR, tumors that had 50% necrosis diagnosed microscopically by pathologists were excluded from this study.
Immunohistochemistry
Samples were fixed in formalin and embedded in paraffin. Paraffin blocks were cut on polylysin-coated microscopy slides for immunohistochemistry. The sections were deparaffinized in xylene, thrice for 10 min, then dehydrated in a descending series of ethanol (100%, 96%, 70%), followed by washing in TBS (0.05 mmol/L Tris-buffer physiological saline, pH 7.4-7.6), thrice for 5 min. Antigen retrieval was achieved by heating the samples without boiling in 10 mmol/L sodium citrate buffer, pH 6.0 (200 mL) in a microwave oven. The sections were then incubated with previously characterized primary polyclonal antibody CAR 72 (Onyx Pharmaceuticals, Richmond, CA, USA) diluted at 1:7000 at 37°C for 30 min. After washing in PBS for 10 min, the sections were incubated with the EnVision reagent (Dako, Denmark) at 37°C for 30 min. Following washing in TBS for 10 min, the sections were incubated with streptavidin-peroxidase (Maixin. Bio., Beijing China) in humid boxes at room temperature for 30 min. The final staining was done in diaminobenzidine tetrahydrochloride (DAB, Maixin. Bio.) solution (49 ml the TBS-buffer, 34 mg imidazole, 17 μl 30% hydrogen peroxide and 1 ml 30% DAB), for 5 min. The slides were washed with distilled water, 70% ethanol for 1 min, then in distilled water. The nuclei were stained with Mayer’s hematoxylin for 30 s. Extra stain was washed with tap water. The slides were then transferred through an ascending ethanol series and xylene before mounting.
The percentages of positively stained cells were obtained by counting at least 1000 tumor cells in each case by 2 independent observers blinded to the clinical data with complete observer agreement. Specimens were considered “positive” for CAR when more than 5% of the tumor cells were stained, while they were characterized to represent “high” CAR expression when the percentage of positively stained cells exceeded the mean percentage value. The cellular pattern of distribution of CAR immunostaining was characterized as membraneous and cytoplasmic.
To ensure the specificity and the intensity comparison of CAR staining, archived normal prostate tissues (with known CAR expression in prostate epithelium), and lymph nodes with reactive hyperplasia (with no known expression of CAR in lymphocytes) were used as positive and negative controls, respectively.
RT-PCR
Total RNA was extracted from tissue samples by the guanidinium thiocyanate-phenol chloroform extraction method. RNA (2.0 μg) was converted to cDNA by reverse transcription, using avain myeloblastosis virus reverse transcriptase (TaKaRo Biotechnology (Dalian Co., Ltd.) and oligo (dT)15 primers (Promega, Madison, WI). For RT-PCR, one-twentieth of the cDNA was used to amplify the entire coding region of the CAR gene. Three primers used were as follows: CAR1 forward, 5’-GCA GGA GCC ATT ATA GGA ACT TTG-3’; and CAR1 reverse 5’-GGA CCC CAG GGA TGA ATG AT-3’. CAR2 forward, 5’-CTG GAC TCT CCT GCG GCG TC-3’; and CAR2 reverse 5’-AGC GGC CCG GCT GCT CCT G-3’. CAR3 forward, 5’-ACT GGA ACT GAT TAT GGC TCA-3’; and CAR3 reverse 5’-ACC CCT GCC CTC TCC GAC-3’. The lengths of the expected products were 195 bp, 226 bp and 363 bp, respectively. The PCR protocol consisted of initial denaturation for 3 min at 94°C, followed by 35 cycles of denaturation for 30 s, at 94°C, annealing for 30 s, at 57°C and extension for 30 s, at 72°C; and a final extension for 10 min, at 72°C. A control PCR with primers that amplify a 114-bp fragment of the β-actin, forward 5’-ACC CCC ACT GAA AAA GAT GA-3’; reverse 5’ -ATC TTC AAA CCT CCA TGA TG-3’ was performed in parallel to verify similar amounts of cDNA in each sample. The PCR products were analyzed by electrophoresis on a 1.8% agarose gel containing ethidium bromide and photographed under UV light. The semiquantitive value of each PCR band equal to the ratio of the CAR value to the β-actin value was analyzed by Quantity One 1-D Analysis Software Version 4.6.0.
Western blot
For immunoblotting analysis, cells were lysed for 1 h at 4°C in lysis buffer containing 50 mmol/L Tris-Hcl (pH 7.5), 150 mmol/L NaCl, 1% Nonidet-P40 (NP40), 1% triton-X, 1% sodium deoxycholate, 1mM EDTA, 1 mM PMSF, 1μg/ml each aprotinin, leupeptin, pepstatin (Roche, Germany) per 10 ml lysis buffer. After the removal of cell nuclei by centrifugation, protein concentrations were measured by the BCA protein assay. Equal amounts of protein (15 μg) from each cell lysate were diluted with SDS loading buffer, heated for 5 min, at 95 °C and electrophoresed on a 10% SDS-polyacrylamide gel. The separated proteins were then electrotransferred to a nitrocellulose membrane, which was blocked with 5% non-fat dried milk in Tris buffered saline-Tween (TBST) buffer to block nonspecific binding sites. The blot was then incubated with polyclonal rabbit anti-CAR primary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA; 1/1000 dilution in TBST and 0.01 g/ml BSA) with gentle shaking for 1 h, at room temperature. After extensive washing, polyclonal anti-rabbit horseradish peroxidase-conjugated secondary antibody (1/1000 dilution in TBST and 0.01 g/ml BSA) was applied for 1 h, at room temperature with gentle shaking. Bands were visualized using an ECL kit (Amersham) according to the manufacturer’s instructions. β-actin was used as control. We used ImageQuant TL v2005 software to analyze the grey scale of the CAR protein bands. The relative expression of CAR protein was the ratio of CAR expression to β-actin expression.
Statistical analysis
The Chi-square test was used to assess the association of CAR positivity with clinicopathologic variables. A 2-tailed P < 0.05 was considered statistically significant. Statistical analysis was performed using the software package SPSS for Windows (version 11.0; SPSS Inc., Chicago, IL, USA).
Results
Imuunohistochemistry
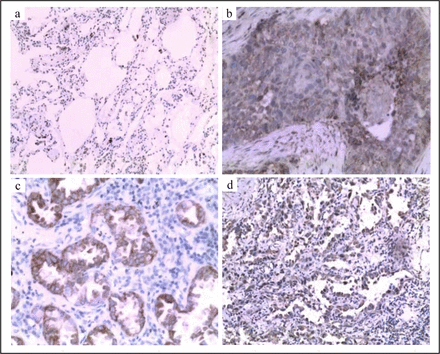
CAR stained the cell membrane of epithelial cells and tumor cells but not that of interstitial cells in all 112 cases (Fig. 1). First, we checked the expression of CAR in squamous cancer. High levels of CAR expression were detected in 28 of 65 squamous cell carcinoma specimens and in 5 of paraneoplastic epithelial tissue. The positive rate in squamous cancer (43%) was significantly higher than that in paraneoplastic epithelial tissue (8%) (P < 0.05) (Table 1). We also studied the expression of CAR in adenocarcinoma. There were 33 cases of positive CAR among the 47 adenocarcinomas, a positivity rate of 70%; 15 cases were positive in paraneoplastic epithelial tissue, a positivity rate of 32% (Table 1). The elevated level of CAR in adenocarcinoma was significantly much higher than that in the paraneoplastic epithelial tissue (P < 0.05).
Immunohistochemical staining of non-malignant lung tissue and lung cancer tissue with polyclonal antibody CAR. CAR staining was not detectable in paraneoplastic lung tissue (a). Representative CAR staining in squamous cell carcinoma lung cancers (b), lung adenocarcinoma (c) and bronchioalveolar adenocarcinoma (d). All data were documented under a magnification of × 200.
Different expression of CAR between tumor tissues and paraneoplastic tissues.
Then we also analyzed the correlation of CAR protein expression with different clinicopathologic parameters. Compared with CAR expression in squamous cancer, the positive expression rate in adenocarcinoma was significantly much higher than that in squamous cancer (P < 0.05) (Table 2). There was no statistical significance among other clinicopathologic parameters (P > 0.05), including gender, age, smoking status, tumor size, and with or without lymph node metastasis (Table 2).
Association between CAR expression and clinicopathologic characteristics of the 112 patients with lung cancer.
Detection of CAR mRNA by RT-PCR
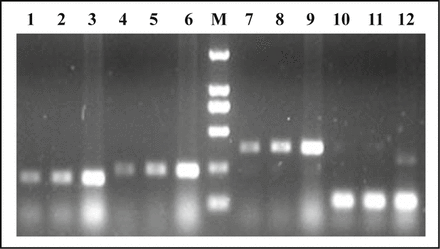
We first examined the expression of CAR by RT-PCR. Integrity was evaluated by running RNA on a 1.8% agarose gel; the quantity of RNA was measured spectrophotometrically. cDNA was then synthesized, and CAR specific sequences were amplified using CAR-specific primers (CAR1, CAR2, CAR3). As shown in Fig. 2, CAR primers generated fragments with the expected lengths of 195 bp, 226 bp and 363 bp. The expression value of CAR mRNA in normal lung tissue, paraneoplastic tissue and carcinoma tissue were 1.000 ± 0.012, 1.048 ± 0.035, 1.282 ± 0.072, respectively. Carcinoma tissue was statistically significant higher than normal lung tissue and paraneoplastic tissue, and the difference was significant (P = 0.022, P = 0.007).
Agarose gel electrophoresis of CAR RT-PCR products. No. 1-3 lanes used primer 1 (195 bp), No. 4-6 lanes used primer 2 (226 bp), No. 7-9 lanes used primer 3 (363 bp) and No. 10-12 lanes are Actin (114 bp). No. 1,4,7,10 lanes are lung normal lung tissue, No. 2, 5, 8, 11 lanes are paraneoplastic tissue and No. 3, 6, 9, 12 lanes are carcinoma.
Detection of CAR protein by Western blot
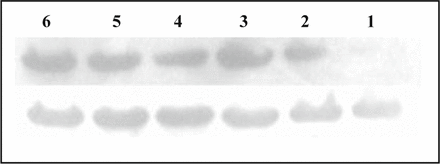
As shown in Fig. 3, a band of about 46 kd was detected in normal lung tissue, paraneoplastic tissue and carcinoma tissue, with the expression value 0.902 ± 0.038, 0.944 ± 0.042, 1.08 ± 0.052, which are shown in Fig. 3. Carcinoma tissue was significant higher than normal lung tissue and paraneoplastic tissue (P = 0.09, P = 0.027). And there was a statistically significant positive correlation between expression of CAR mRNA and protein (r = 0.448, P = 0.026).
The expression of CAR protein in normal lung tissue, paraneoplastic tissue and carcinoma by Western blot assay: the upper line shows the expression of CAR protein and the lower one shows actin. No. 1, 4 lanes are normal tissue; No. 2, 5 lanes are paraneoplastic tissue and No. 3, 6 lanes are carcinoma.
Discussion
Recently, many researchers have characterized CAR as a global tumor suppressor which demonstrates that CAR serves as a tumor suppressor in glioma cells both in vitro and in vivo[21]. This provides a physiological explanation for the downregulation of CAR observed in malignant glioma cells[26]. Downregulation of CAR has similarly been observed in other tumor types, including ovarian cancer[27,28], melanoma[29], head and neck cancer[27,29-32], malignant glioma[21], musculoskeletal tumors[33], immature osteoblasts[34], human rhabdomyosarcoma[35] and bladder cancer[5,36]. Similarly, when compared with normal prostate, CAR expression decreased in prostate carcinoma specimens of all Gleason scores[37]. Yamashita et al. reported that CAR expression significantly decreased the lung metastatic potential of B16 melanoma cells in lung after IV injection and in the migration in murine model[38]. Here, we systematically analyzed CAR expression in a large number of lung cancer specimens with RT-PCR, immunohistochemistry and Western blot analysis. Our study is the first one to report the examination of the clinical significance of CAR expression in patients with lung cancer. We found that there was a little expression of CAR mRNA and protein in normal lung tissue or paraneoplastic epithelial tissue. However, a high level expression of CAR, not only the mRNA but the protein, was found in most of lung cancer samples, especially in lung adenocarcinoma. It suggests that there may be some relationship between lung cancer and the expression of CAR. Based on these results, we proposed a hypothesis that the increase in CAR activity may well be one of the characteristics of lung tumors and that CAR may play an important role in the formation or development of certain lung cancers, especially lung adenocarcinoma, though it may play a suppressor role in other kinds of cancers.
All reports about CAR expression and human lung cancer which we searched using MEDLINE support our hypothesis: Wang et al. reported that high levels of CAR expression were detected in a majority of squamous cell lung cancers, but only in about 1/3 of lung adenocarcinomas; it was not reportedly detected in non-malignant lung cells[23]. Qin et al. reported that CAR expression in vitro favorably predicted a comparable pattern of expression in transplanted NSCLC xenografts in vivo; and xenografts generated from NSCLC cells showed increased CAR expression[24]. They further demonstrated that silencing surface CAR expression abrogated xenograft tumorigenesis in vivo and colony formation in vitro and invoked the novel possibility that CAR expression was needed for the efficient formation of tumors by a subset of lung cancer cells. They also believed that CAR expression may play a prominent role in the pathogenesis of certain lung cancers; however, they used representative cell lines, not primary lung cancer materials[25]. Brüning et al.[22] analyzed the role of differential CAR expression during tumorigenesis and in cell survival of adenocarcinomas using a murine mammary cancer model and different cancer cell lines. They found that in comparison of preneoplastic precursor lesions with established adenocarcinomas, CAR expression was enhanced 2- to 5-fold in all 6 tissues which had survived and transformed into invasive adenocarcinomas. The elevated expression of CAR in human lung cancer, especially in lung adenocarcinoma, as described here, the enhanced CAR expression in adenocarcinoma outgrowths and the involvement of CAR in adenocarcinoma xenograft formation all suggest a potential function of CAR in the early development of at least adenocarcinomas[25]. This data may warrant further research on the role and mechanisms of CAR in the formation and development of human lung cancer. If CAR is a promoting factor of human lung cancer, we might develop antibody-directed therapy as epidermal growth factor (EGF) receptor did in human lung cancer.
On the other hand, if CAR is not a promoting factor of human lung cancer, the elevated expression of CAR in lung cancer will be more favorable to the gene therapy of lung cancer since the level of CAR expression may be one of the most important determinants of the efficacy of gene therapy and late resistance to Ad-p53[32,39]. Our data reveal that there is little expression of CAR in normal lung tissue and paraneoplastic tissue, but high positive rates in human lung cancer: 43% in squamous cancer with a relatively low CAR expression in low-grade cancers compared to high-grade, and 70% in adenocarcinoma. Therefore, adenocarcinoma may be more suitable to gene therapy, especially bronchioalveolar carcinomas.
In summary, we demonstrate that the mRNA and protein of CAR are overexpressed in human lung cancer, especially in adenocarcinoma. We believe that CAR may play an important role in the formation or development of human lung cancers, and may be exploitable for the development of antibody-directed therapy in human lung adenocarcinoma. Even though CAR is not a promoting factor of lung cancer, our data can offer the reliable basis for the gene therapy of lung cancer, especially adenocarcinoma.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Acknowledgement
The investigators would like to thank Dr. Xiao-qing LI and Dr. Yi PAN for their assistance with this project.
- Received January 4, 2010.
- Accepted February 12, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer