Abstract
OBJECTIVE To establish a murine uterine cervical cancer cell line and to define its biological characters.
METHODS Transplanted tumor tissue was used for in vitro primary culture of U14 cervical carcinoma cells. After 20 passages, we examined its morphology, chromosomes, tumorigenicity and produced a growth curve. CK was detected by immunohistochemistry, the cell cycle determined by flow cytometry and the metastatic potential assessed in 615 and C57BL/c mice. We also transfected the cells with the pEGFP-N1 plasmid.
RESULTS A newly established murine cell line was passaged 50 times over a period of 10 months. The cells grow as a partially suspended culture, and are immunohistochemically CK(+). The cell line is characterized by a hypotetraploid karyotype, a chromosomal number of 64-68 and a doubling time of 21.8 h. Exponential growth occurs by the third and forth day of culture. Cell cycle analysis showed G1 34%, G2 26%, and 40% in the S phase. The tumorigenicity was 100% upon implantation. No mycoplasma contamination was detected. A monoclonal continuous U14-GFP cell strain which was 100% GFP (+) was also produced.
CONCLUSION We successfully established a new murine cervical U14 carcinoma cell line and an U14-GFP monoclonal strain. These cell lines are ideal for combined in vivo and in vitro tumor research.
keywords
Introduction
The U14 mouse tumor (No.14 of cervical carcinoma) is a squamous cell carcinoma. It was an ectopically induced carcinoma by treating the uterine cervix with 20-methylcholanthrene. The tumor was established and maintained by the Laboratory of Experimental Oncology, Department of Pathology (Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences & Peking Union Medical College), in 1958[1]. At an early stage it morphologically looked like a carcinosarcoma, but in the 1980s’ it was identified as an undifferentiated carcinoma. When U14 cells were transplanted into mice, the incidence of lymphatic metastasis and pulmonary metastasis was respectively 95% and 80%, and the average survival time of the mice with the tumor was 27 days. U14 cells are widely used in studies of tumor invasion, metastasis, recurrence and drug screening. Establishment of a cultured tumor cell line, which can form a tumor in vivo, would be helpful for the study of tumorbiology on a cellular and molecular level[2-5].
In this report, we describe the establishment of a new U14 cell line and characterized its properties. These cells can provide an ideal model for combined tumor research in vivo and in vitro. The cells were also transfected with the GFP molecular marker to enable viewing it in vivo.
Materials and Methods
Tumor tissue source and culture of tumor cells
The U14 tumor was established and maintained in stock in our labotatory[1]. After the U14 cells were thawed from liquid nitrogen, they were transplanted intraperitoneally into 615 inbred mice, 18~22 g, 4~6 weeks old. The mice were R Grade animals [Certification: SCXK (JING) 2004-0001], from the Institute of Laboratory Animals of the Chinese Academy of Medical Sciences. On day 13, 1 ml of ascites fluid was drawn from the mice using a sterile technique. The extract then was diluted with D-Hank’s solution and centrifuged for 5 min at 1000 rpm. After washing the cells 3 times with D-Hank’s solution, the suspension was plated into culture flasks with DMEM growth medium (10% fetal bovine serum, 0.1 U/L penicillin G sodium, 0.1 U/L streptomycin sulfate) followed by culturing in 5% CO2 at 37°C. The media were half replaced after 2 days, and the cells were passaged every 3 days.
Observation of morphology
The monolayer cells were fixed on coverslips with 95% (V/V) ethanol. The morphology of the live cells and H&E stained cells were examined under an inverted phase contrast microscope (Olympus CK30) or light microscope (Nikon SE).
Growth properties
The cultured cells at passage 24 and passage 26 were plated at a density of 3×105 cells/flask and 1.2×105 cells/flask into 24 T25 flasks. From the next day on, cells were harvested from 3 cultures per day, counted on a CASYTT cell viability counting meter and the data used to diagram a growth curve.
Colony forming efficiency
Cells of the 26th passage were harvested and seeded into a 6-well plate, using 200 cells per well. Then the plates were incubated in 37°C under 5% CO2 in a saturated atmosphere. Growth medium was changed every other day. After 10 days, the cell colonies were counted to calculate a colony forming efficiency using the following equation:
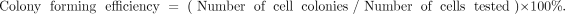
Immunohistochemistry
A polyclonal cytokeratin antibody (rabbit anti-human cytokeratin, Zhongshan Biotechnical Corp., Beijing) was used for CK staining. After fixation with 95% ethanol, the monolayer cells were incubated with the CK antibody at 4°C overnight, after which the color was developed with an envision kit. The stained slides were observed microscopically.
Analysis of the cell cycle
Cells at the 51st passage were harvested, centrifuged, washed with PBS and adjusted to a density of 2×106/ml. After the cells were fixed in cold ethanol, they were incubated with PI for 30 min and analyzed immediately by flow cytometry (SONY Counlter).
Chromosome analysis
The established tumor cells at the 12th passage were subjected to chromosomal analysis. The cells were treated with 0.6 μg/ml colchicine for 4 h. After the medium was removed, the cells were digested with 0.05% trypsin and centrifuged at 1000 rpm for 5 min and suspended in hypotonic 0.075% KCl for 30 min. The cells then were fixed in freshly prepared, icecold acetic methanol for 20 min. The suspension was dropped onto a cold glass slide from around 40 cm, followed by staining with Giemsa for 5 min[6]. After washing the cells with distilled water and air drying, the chromosomal number was counted under an oil immersion (Olympus).
In vivo transplantation
A suspension of 7.5×106 U14 cells from the 49th passage and 4.7×106 cells from the 51st passage were injected subcutaneously into the axilla of 10 inbred 615 mice and 10 C57BL/c mice respectively. The latent period of tumor mass formation was observed and once one of the tumor-bearing mice had died, all the animals were sacrificed euthanized and the tumorigenicity calculated.
Detection of mycoplasma
Cells from the 19th passage and 24th passage were propagated in DMEM without antibiotics for 4 days, after which the supernatants were examined for mycoplasma infection by PCR and culture methods[7].
Transfection of the U14 cells with pEG-FP-N1
U-14 cells in an exponential growth phase were plated without antibiotics into 24-well plates (Corning, New York, USA) the day before transfection. The cells were transfected with pEGFP-N1 (the vector was kindly provided by Prof. Xuemei Xu, from the Institute of Basic Medical Sciences of Chinese Academy of Medical Science and Peking Union Medical College, Beijing), when they reached 90% confluency. The transfection was performed according to the instructions of the Lipofectamine 2000 supplier (Invitrogen). After incubation of the cells for 24 h at 37°C under 5% CO2, the efficiency of transfection was assessed under a fluorescence microscope (Leica DMI 4000B)[8]. After 48 h, the cells were transfered to selective media containing G418 sulfate (800 μg/ml). Then the GFP-positive cells were subcloned into a 96-well plate (Corning, New York, USA) by a limited dilution, and a individual cell clone which was GFP (+) was propagated and designated U14-GFP.
Viviperception of nude mouse inoculated with U14-GFP cells
U14 cells (1×106) were injected i.v. into nude mice. On the 6th day after transplantation, the animals were anaesthetized using tribromoethanol and the expression of GFP in vivo was observed by the Viviperception Chemiluminescence and Fluorescence Imaging System (provided by Institute of Laboratory Animals of Chinese Academy of Medical Sciences, Beijing).
Resutls
Cell culture and morphological analysis
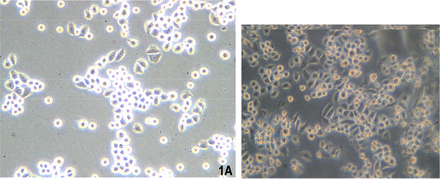
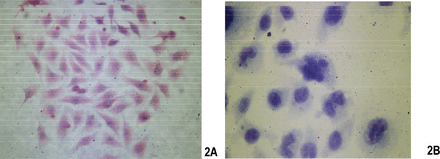
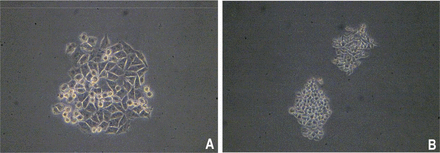
Two days after culture in vitro, some cells became adherent, but the majority remained suspended (Fig.1). The cells grew quickly and the media were half replaced after 2 days. The cells were passaged every 3 days. From the 20th passage, the percentage of adherent cells increased and the suspended cells decreased. The cells were passaged 50 times over a 10 month period. The U14 cells are epithelioid, display overlapping growth and mainly appear polygonal and spindle shaped under a contrast phase microscope. The cells are lightly alkalophilic, have a large nucleus and display numerous mitosis with H&E and Gimsa staining (Fig.2).
Images of U14 cells under a phase contrast microscope. (A) the 6th passage (Olympus 10×10); (B) the 26th passage (Olympus 10×10).
The staining images after fixation. (A) H&E staining (Olympus BH-2, 10×10); (B) Giemsa staining (Olympus BH-2, 10×10).
Growth kinetics
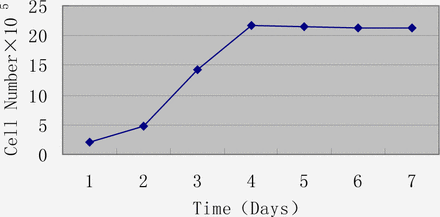
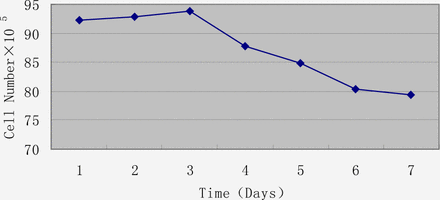
When 1.2×105 cells were incubated in T25 flasks, the latent period was short. On the 3rd day, they entered a logarithmic growth phase, reaching a plateau on day 4 (Fig.3). After 5 days, many cells had died due to exhaustion of nutrition (Fig.4).
Growth curve of the 14th passage of U14 cells.
The rate of viability of U14 cells by a CASY®TT cell counter and analyser system.
Colony forming efficiency
Colony forming was positively related to cell malignancy. The U14 cells formed 33, 25, 31 and 34 colonies in our study with an average of 30.8. Colony forming efficiency was 15.4% (Fig.5).
U14 cells form colonies (A) (Olympus CK30, 10×10); (B) (Olympus CK30, 10×4).
Immunohistochemistry
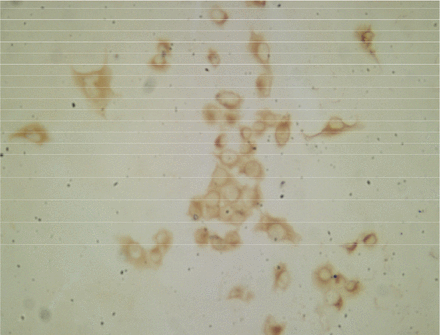
Analysis by immunohistochemical staining using polyclonal antibodies showed positive staining of the cytoplasm for cytokeratins (Fig.6), verifying the epithelial origin of the cells.
U14 cells with positive staining for epidermal cytokeratins (Olympus BH-2, 10×10).
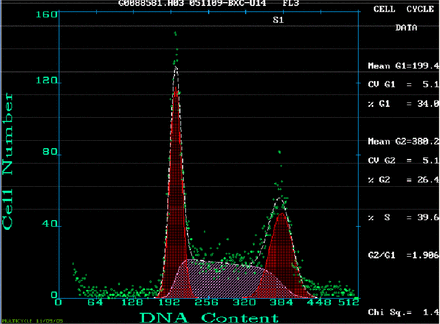
Cell cycle analysis
The cell cycle is a good index to reflect the proliferating status of the cell population. The percentages of the U14 cells in G1, G2 and S were 34%, 26% and 40% respectively (Fig.7). These results showed that a high proportion of the U14 cells were proliferating.
The cell cycle distribution of logarithmic phase U14 cells.
Chromosome analysis
A total of 100 spreads were counted, finding that the chromosome number was 64~86 and the karyotype was hypotetraploid (Fig.8).
The karyotype of the U14 cell was hypotetraploid.
In vivo transplantation
Tumors grew in all the mice transplanted with U14 cells showing a latent period of 7~10 days (Fig.9). Nodular skin-invasive tumors with necrotic centers formed. The 615 mice and C57BL/c mice died subsequently, after 28 days and 34 days of transplantation respectively.
The tumorigenicity of C57BL/c mice transplanted with U14 cells was 100%.
Detection of mycoplasma
No mycoplasma contamination was detected.
pEGFP-N1 Transfection
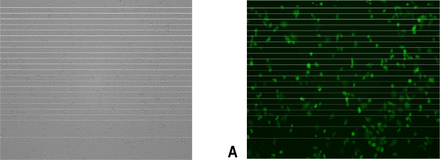
The efficiency of pEGFP-N1 transfection at 24 h was 60% based on fluorescence microscopic examination. A clonal strain, which expressed GFP at a level 100% (designated U14-GFP), was scaled up and cryopreserved (Fig.10).
U14-GFP cells express GFP 100% (A) light microscope (Leica 10×10), (B) fluorescence microscope (Leica 10×10).
Viviperception of transplanted U14-GFP cells
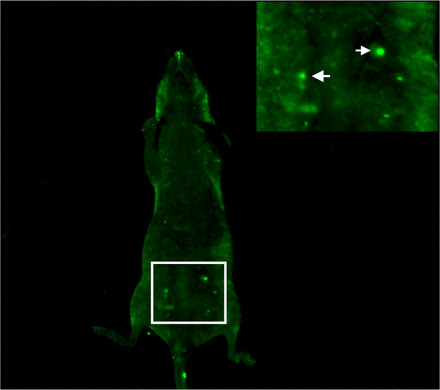
No fluorescence could be detected immediately after intraperitoneal injection of 1×106 U14-GFP cells. On the 6th day, ascites developed in the nude mice with significant green fluorescence cell clumps in the abdominal cavity (Fig.11).
In vivo imagines of a nude mouse injected i.p. with U14-GFP cells. The arrows indicate the aggregated U14-GFP cells.
Discussion
U14 cells are widely used for studies of tumor invasion, metastasis, recurrence and drug screening[9]. Metastases develop in both lymph nodes and lungs. Our studies showed that the U14 tumor cell line was positive for cytokeratin staining, demonstrating its epidermal origin. U14 cells are adherent epithelioid cells with polygonal or triangle morphology, and show an overlapping culture in flasks. The cells have a large nucleus, and display active patho-karyokinesis. There are also multinucleated giant cells. The cells propagate quickly, reaching a plateau on day 4 and die without media supplementation. These highly malignant cells have a tumorgenicity of 100%, a hypotetraploid karyotype, and a chromosome number of 64~68. U14 cells have been cultured in vitro for 50 passages over a period of 10 months, indicating that it is a stable tumor-cell line. No mycoplasma contamination was detected in the cultured cells. The cells have a clear background and definite properties, and they have been maintained and kept in stock of the Cell Culture Center of the Institute of Basic Medical Sciences, PUMC. In addition, we also have established a stable pEGFP-N1 transfected monoclonal cell strain-U14-GFP, which can serve as an ideal labeled tumor model in vivo[10]. On the whole, the U14 murine uterine cervical cancer cell line, which propagates quickly and has a 100% tumorigenicity, can be a useful tool for experimental oncologic research.
- Received August 16, 2007.
- Accepted December 20, 2007.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer