Abstract
OBJECTIVE To investigate the expression of RECK mRNA and protein in esophageal squamous cell carcinoma (ESCC) and to examine its relationship with the clinicopathologic features.
METHODS The expression of RECK mRNA and protein in 62 cases of ESCC, 31 of paraneoplastic atypical hyperplasia (PAH) and 62 normal esophageal mucous membrane specimens was examined, using RT-PCR and immunohistochemistry.
RESULTS During canceration of the ESCC, the mRNA of RECK increased sequentially from ESCC tissue to PAH and normal mucous membranes. Values were 1.052±0.078, 1.274±0.235 and 1.306±0.121, respectively, with a significant difference among different groups (F=49.936, P<0.05). There was a statistically significant difference in the relative amount of the RECK mRNA among the ESCC tissues at various levels of differentiation, depth of infiltration, and different types of lymph node metastasis (F=5.081, F=26.084, U=24.011, P<0.05). In the ESCC tissue and PAH, the positive rates of RECK protein expressions were lower compared to the normal mucosa tissue, i.e. 59.7% (37/62), 71.0% (22/31) and 85.5% (53/62), respectively. There was a significant difference among the inter-group comparisons (χ2=10.331, P<0.01). In ESCC, there was a close correlation between the RECK protein expression and the degree of cancer differentiation, and the depth of invasion and the types of ESCC lymph node metastasis (P<0.05).
CONCLUSION The decrease in expression of both RECK mRNA and protein in ESCC suggest that these low expressions may relate to ESCC development. Examination of RECK mRNA and protein expression may develop into one of the molecular indices for early ESCC diagnosis and prognosis.
keywords
Introduction
In 1998, Japanese scholars[1] discovered a novel tumor-suppressor gene, RECK, that coded for a reversion-inducing cysteine-rich protein with Kasal motifs. It had an unique function of inhibiting the expression and activity of the matrix metalloproteinases (MMPs), and was shown to be a matrix metallo-proteinase inhibitor. Available findings indicated that there was a negative correlation between the RECK gene expression and tumor invasion in hepatic, pancreatic, breast and lung cancers, and usually the prognosis for patients with a higher RECK gene expression was significantly better compared to those with a lower expression[2-4]. In our study we used RT-PCR and immunohistochemical methods to examine RECK expression in ESCC, paraneoplastic atypical hyperplasia (PAH) and normal esophageal mucous membranes. The relationship of RECK expression to the canceration of the esophageal mucosal epithelioma was examined to establish molecular criteria for early ESCC diagnosis and prognosis.
Materials and Methods
General data
Excisional samples of 62 cases of esophageal cancer (EC) were obtained from the Anyang Cancer Hospital, Anyang, Henan province, in a high-risk EC area from February to March, 2006. Among the patients, 36 were males and 26 females, with an age range from 38 to 75 years and an average of 60.6±9.5. No preoperative medical records of chemotherapy, radiotherapy or immunotherapy were found in any of the cases.
Main reagents and primers
A RNA Extraction Kit and Trizol were bought from the Sigma Co. Ltd., and the One Step RNA PCR Kit from the Dalian TaKaRa Bioengineering Co., Ltd. The primers were synthetized by the Beijing AuGCT Co., Ltd., with a primer sequence as follows: RECK upstream primer 5’-TGG AGT CAC TGT ACA CCC TC-3’, downstream primer 5’-CGG ACA TCC GCT AAA CAG GT-3’. β-actin was used for internal reference, with an internal reference primer sequence, i.e. the upstream primer, 5’-CAT CCT GCG TCT GGA CCT-3’, and the downstream, 5’-TCA GGA GGA GCA ATG ATC TTG-3’. The lengths of the anticipated amplified fragment were respectively 209 and 480 bp.
Total RNA extraction from the samples
About 0.1 g of tissue was treated with 1 ml precooled Trizol reagent to extract the RNA. The mixture was ground in a glass homogenizer, and the procedure followed using the instructions provided with the Trizol RNA Extraction Kit. Then the RNA was analyzed spectrophotometrically to determine the RNA concentration and purity, after which the RNA was dissolved in 50 μl DEPC treatment fluid.
RT-PCR measurement of RECK expression
The total reverse transcription system was 25 μl using the operating procedure based on the description in the Kit. Centrifugation was performed for 30 s after mixing. The homogenized reaction was placed into the PCR instrument and the RT-PCR conditions employed as follows: RT reaction was conducted for 30 min at 50°C, and then for 2 min at 94°C to inactivate the RTase; the PCR parameters were 94°C for 30 s, 57°C for 90 s and 72°C for 30 s, with a total 35 cycles. A 10-min extension was run at 72°C. The amplifi cation products (5 μl) were taken out to conduct agarose gel electrophoresis, photographing and analysis to determine the density of the target gene band relative to the band of the β-actin internal reference.
RECK immunohistochemical staining
The mouse-antihuman RECK monoclonal antibody was bought from the Santa Cruz Co., and the SP immunohistochemical Kit from the Beijing Zhongshan Golden Bridge Bio Tech Development Co. The SP method was conducted using a RECK monoclonal antibody working concentration of 1:100, followed by DAB staining and a hematoxylin afterstain. The staining procedure was performed strictly based on the Kit description, with PBS substituting for the first antibody as a negative control, and known positive sections as positive controls.
Assessment of the immunohistochemical staining results
The RECK protein produced a dark-yellow granular staining, located at the cytoplasm. Five visual fields were randomly selected under high power (the number of cells observed in each field was no less than 200), and assessment of the results was conducted according to a percentage of the positive cells and shade of the coloration[5]. A 9-score system was employed as follows: based on the proportion of positive cells, ≤10% meant a score of 1, 10% to 50% a score of 2 and >50% a score of 3. Based on the shade of the staining, a score was assigned: a negative was 0, a light-yellow staining was 1, median yellow was 2, dark-yellow staining was 3. Then a total score was calculated as follows: i.e. a positive-cell score × staining-shade score. A total score of <3 was considered negative, a total score of ≥3 and <6 was positive, and that of ≥6 a stronger positive.
Statistical analysis
The statistical software SPSS11.0 was used to conduct the χ2 test, analysis of variance and rank-sum tests. Results were considered significant if α=0.05.
Results
Expression of the RECK mRNA in the ESCC, PAH and normal mucous tissues
Fig.1 shows the RT-PCR results for the RECK gene. The fragments from the RECK target gene appeared at the band of 209 bp, and β-actin at the 480 bp band. The relative amount of RECKmRNA increased in cancer, paraneoplastic atypical hyperplasia and normal mucosa one by one, with a significant difference in the comparison between the groups (F=49.936, P<0.05). The relative amount of RECKmRNA related to the histological grade, depth of infiltration and lymph node metastasis (F values were respectively 5.081, 26.084 and 24.011, P average value<0.05), see Table 1.
Expression of RECK mRNA in the EC tissue, paraneoplastic atypical-hyperplasia mucous membrane and normal mucous membrane. M: Marker; 1, normal mucous membrane of the esophagus; 2, esophageal mucous membrane with atypical hyperplasia; 3, cancer tissue.
Relationship between the expression of RECK protein and mRNA and the clinical biological behaviour of ESCC.
RECK protein expression in the ESCC tissue, PAH and normal mucous membrane
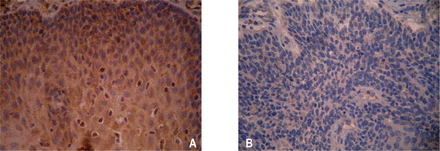
RECK positive staining was found in the cytoplasm with the positive signal identified as a dark-yellow granular substance (Fig.2). The RECK protein expressing rate increased in cancer, paraneoplastic atypical hyperplasia and normal mucosa one by one, with a significant difference in the comparison between the groups (χ2=10.331, P<0.01). RECK protein expression related to the histological grade, depth of infiltration and lymph node metastasis (χ2 was 10.422, 8.550 and 4.751, respectively, P average value <0.05, Table 1).
RECK protein expression in the normal mucous membrane of the esophagus. (A) and ESCC tissue (B) (SP ×400).
Discussion
EC is one of the most frequent malignant tumors. Therefore studies on occurrence and development of EC, as well as on the mechanism of infiltration and metastasis are of particular importance. The oncogenesis and development of EC takes a multiphase and multifactorial course[6], involving changes in various gene expression, but the details are still unclear. In our study, we employed RT-PCR and immunohistochemical technology to investigate the relationship of the RECK protein and mRNA expression with the development and clinical biological behaviour of ESCC.
The RECK gene was found, by Takahashi et al.[1], in the NIH3T3 cell line, with a v-Ki-Ras gene transfection. It is located at the 9p13-p12 chromosome, with a total length of about 87 Kb. There was an over expression of RECK gene in normal tissues, but the expression of RECK gene was significantly lowered, or even was not found in tumor cell lines and the cells transfected by oncogenes such as Ras, etc. Many oncogenes, such as Ras and fos, etc., can down-regulate the expression of RECK genes[7], indicating the RECK gene may possibly be a negative-accommodation target site as a common effect of the oncogenes.
There is high expression of the RECK gene in various normal histocytes of the human body. Immuno-histochemical assays and RT-PCR detection indicated that in tumors displaying strong invasion, such as the cancer of the lung and pancreas, the RECK mRNA and protein expression rate was significantly lowered, or even was not found[8,9]. Results of our study showed that the RECK gene was expressed in normal esophageal tissue, esophageal PAH and cancerorous tissue. However, the RECK expression was lower in the cancer tissues, with a statistically significant difference, indicating that the tumor with a low expression of the RECK gene has a greater potential for invasion.
Our findings show that the RECK gene is expressed in normal esophageal tissue, PAH and cancer tissue, but the expression is significantly lower in the EC tissue. In ESCC, there is a very close correlation between the changes in RECK mRNA and protein expression and the invasion and metastatic potential. The expression of the RECK gene may also be regarded as an index for prognostic judgment. Studies of RECK expression will further our understanding of the biological behaviour of EC, and may provide a new means for early diagnosis and treatment of EC.
Footnotes
This work was supported by a grant from the Key-Faculty Construction Fund for Project 211 of the 10th Five-Year Plan of the Ministry of Education [Jiaozhongban (2002) No.2].
- Received November 2, 2007.
- Accepted January 9, 2008.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer