Abstract
OBJECTIVE To evaluate the therapeutic efficacy of treating glioblastoma with interstitial irradiation using radioactive seeds.
METHODS First, the tumor was removed as completely as possible under an operating microscope. Then 125I seeds which had been selected were implanted in the tumor bed followed by routine radiotherapy after the operation. The time of progression (TTP) and overall survival (OS) of the patients were recorded, and statistically compared with patients who had received only surgery and radiotherapy for glioblastoma during the same period.
RESULTS Nineteen patients, who were diagnosed with glioblastoma for the first time, with a mean age of 51.2 years, were involved in this study. Radioactive seeds (8~20) were implanted into the tumor bed after tumorectomy with a mean surrounding therapeutic dose of 31.6 Gy, followed by an external irradiation dose of 42~54 Gy. The average TTP in the group with implanted seeds was 42.1±2.4 w (95% confidence interval, 37.2~46.5 w), and 27.1±2.7 w (95% confidence interval, 24.3~31.8 w) in the control group. The average OS was 66.3±3.2 w (95% confidence interval, 61.9~70.4 w) and 47.7±2.6 w (95% confidence interval, 44.4~50.5 w), respectively. Statistical analysis showed there was a significant difference between two groups.
CONCLUSION The survival time of patients with glioblastoma can be prolonged through radioactive seed implantation at the time of operation. This therapeutic method can rapidly reduce the tumor volume with a minimum number of seeds to avoid excessive radiation exposure. Because of some shortcomings of this study, such as a small number of patients, and because implantation was performed under the naked eye so that the seed alignment could not be verified accurately, the experience of combining implantation of seeds with external irradiation needs to be investigated further.
keywords
INTRODUCTION
Glioblastoma, also called glioblastoma multiforme, WHO grade IV, is considered to be one of the most common malignant intracranial human tumors. It makes up about 25% of intracranial adult tumors. The effect of combined therapy (surgery, radio-and chemo-therapy) is unsatisfactory, so patients with glioblastoma usually have a short survival due to its early recurrance. To investigate a more effective therapeutic method, 19 patients suffering from glioblastoma received implantation of 125I seeds in the tumor bed after the surgical resection was completed. The research was conducted in the Neurosurgery Department of the Fourth Hospital of Hebei Medical University from June 2002 to December 2005. All patients received follow-up.
MATERIALS AND METHODS
Clinical data
General data
Nineteen patients including 11 males and 8 females were involved in this study after their first diagnosis of glioblastoma. The range of ages was 35~65 years, with a mean of 51.2 years. The Karnofsky Performance Scale (KPS) of all patients was 70 or more.
Location and size of tumors
All the tumors in the 19 cases were above the tentorium of the cerebellum. Eight of the 19 were located in the frontal lobe, 6 in temperal lobe, 3 in the temperal parietal lobe and 2 in the parietal occiptal lobe. Based on the CT scan and MRIs, the tumor diameters were 2.5~4.2 cm, with a mean value of 3.6 cm. From images in all the cases there was no indication of infiltration of the meninges or ependyma.
Therapeutic methods
Surgical approach
The surgical approach selected was based on the location of the tumor. Most of the tumors were char-acterized by a part being located under the cortex, with cystis degeneration and necrosis and an unclear boundary from the normal brain tissue. Glioblastoma diagnoses were established by a rapid histopathology examination during the operation. The tumors located in a nonfunctional region were removed completely, and edematous tissue around the tumor also was partly resected. Some tumors near an important brain area were separated and resected from the peripheral gliosis zone so as to protect neural function as far as possible. All operations were performed under an operating microscope.
Selection of radioactive seeds
125I seeds, which were selected for permanent implantation, had the following radioactive activity: half life of 60.2 days, γ-ray energy of 0.028 MeV, tissue penetration range of 1.7 cm and half value thickness of 0.025 mm lead. All 125I seeds involved in this study were provided by the ATB Biomedical Engineering & Technology Co. Ltd. The activity of the seeds was 0.3~0.4 mCi.
Implantation of 125I seeds
The number of seeds required was calculated using the formula (length+width+height of the tumor bed)/3×5/seed activity. Despite irregular shapes of the tumor beds, the distribution of seeds was conducted strictly following the Paris principal. The seeds were arranged in a straight line with an equal interval (1.5~2.0 cm), and parallel to each other. After hemostasis in the tumor bed, the seeds were implanted into the superficial layer of the brain tissue, having their long axis vertical to the surface of the tumor bed. At last the surface of the tumor bed was covered by a surgical or thin collagen sponge fixed with OB biogel. Postoperative verification: CT scanning was performed from 24 to 48 h after operation to verify the location of the seeds and to determine whether or not acute cerebral edema eveloped. It was completed once again before administering radiotherapy at the 4th week after operation. Meanwhile the radiation dose was confirmed through reconstructed images.
External radiotherapy
All patients had gone through routine radiotherapy which usually started from the 4th week after operation. The dosage of external radiotherapy was determined based on the nature of the seeds implanted. At the same time, all patients received dexamethasone and some routine medicines for haemostasis, infections and epilepsy. The dose of these medicines were be adjusted in reference to the actual condition.
Follow-up
The follow-up period for all patients was 6 months to 2 years during which the time to progression (TTP) and overall survival (OS) of patients were recorded. TTP was the time from operation to tumor progression which was definited as when new tumor formed or the tumor volume exceeded 25% of the initial volume before chemotherapy. OS was recorded as the time from the begining of chemotherapy to death. The Kaplan-Meier survival curve was then producted. The control group was comprised of 24 patients who had received only surgery and external radiotherapy after 2000 in the Neurosurgery Department of the Fourth Hospital of Hebei Medical University. There was a significant difference in the survival curves between the two groups using the logarithm rank test.
Statistical analysis
There were no differences in sex, age, KPS score and tumor distribution between the two groups. Side effects and changes in imaging after radiotherapy were also observed during the follow-up period. Analysis of the data was completed with SPSS 12.0 software (SPSS company, America).
RESUTLS
Data from implantation of seeds and external radiotherapy
Implanation of 8~20 125I seeds after operation resulted in a therapeutical dose in the tumor bed perimeter of 24.5~38.7 Gy, with a mean dose of 31.6 Gy. The total dose of external radiotherapy was 42~54 Gy, divided routinely over 20~25 days. The exposure field was limited to a small range.
Therapeutic efficacy
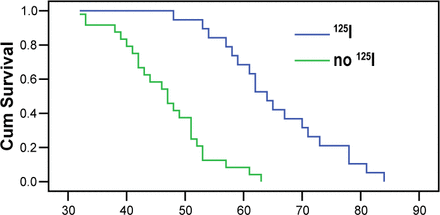
The average TTP for the group receiving the implants was 42.1±2.4 w (95% confidence interval, 37.2~46.5 w), and for the control group 27.1±2.7 w (95% confidence interval, 24.3~31.8 w) as shown in Fig.1. Fig.2 shows that the average OS was 66.3±3.2 w (95% confidence interval, 61.9~70.4 w) and 47.7±2.6 w (95% confidence interval, 44.4~50.5 w), respectively for the treated and control groups. Statistical analysis indicated that there was a significant difference between the two groups (P<0.01).
Time to progression (w).
Overall Survival (w).
Side effects of radiotherapy
Through regular CT scaning, all patients with implantated seeds were monitored for tumor assessment, the position of the seeds, cerebral edema, signs of recurrence and cerebral radiation necrosis. In the first week after operation, the cerebral edema was relieved by different amounts in 11 patients, regressed more slowly in 7 patients and these showed no evidence of consciousness disturbance or motor disorders. Severe edema occurred in one patient whose relatives refused decompression surgery for the patient. This patient’s symptoms improved gradually until the 18th day after operation through more dehydration and hormone therapy, and stabilized fully after one month. Cerebral radiation necrosis was found in none of the patients. Epilepsy appeared in 2 patients who had received anti-epileptic treatment, but was then controlled by drag adjustment. The incidence of epilepsy showed no significant difference between the two groups.
CT scan revealed that the position of seeds was relatively stable except for aggregation to some extent. There was no migration of the seeds into the cerebral ventricle or deep tissue.
DISCUSSION
The treatment for glioblatoma always represents a significant challenge to neurosurgeons. Over the long term, many techniques have been tried to hamper the growth of the tumor, such as immunotherapy, antiangiopoiesis, gene therapy and so on. But clinical practice has shown that therapeutic efficacy can not be improved by any single method. Short-term tumor recurrence is the leading cause of short survival times and deterioration of the quality of life. Recurrences are the result of remnant tumor cells after operation[1]. Some factors that may lead to tumor remnants are summarized as follows: 1) with severe infiltration of the surrounding tissue and an unclear boundary, the tumor can not be completely resected. 2) sub-clinical lesions and less failure of the blood-brain barrier (BBB) on the margin of tumor, and the remant tumor cells may have poor radiosensitivity. 3) remnant cells on the tumor margin can not be treated effectually by applied chemotherapeutics. So the main problem is how to maximatily kill the tumor cells and prevent a recurrence of the tumor. It is worth trying any method that might reduce the tumor residue.
Radiotherapy has been regarded as an indispensable therapeutic process after glioma resection. But due to the limits of tolence of normal brain tissue, the conventional external radiotherapy dosage can not utilized. Continuous interstitial irradiation may be able to specifically resolve this problem by improving the local dose applied to the remnant tumor, reducing the injury to normal tissue, inhibiting mitosis and repopulation of tumor cells, and enhanceing tumor cell radiosensitivity[2]. Although interstitial radiotherapy has been used to treat tumors for more than 100 years, it has been applied against intracranial tumor in China for only a few years. The form of radionuclide most frequently used for intracranial glioma is 125I seeds[3,4] which posseses advantages of low average energy, quick energy attenuation beyond an effective distance, long half-life time, and an ease in safe guarding and preserving the radioactivity. Some studies have shown that 125I can be tolerated by brain tissue, and can produce a satisfactory therapeutic efficacy[5,6]. The 125I seeds implanted permanently into tissue usually have been of low activity with a low dose rate (LDR) so as to reduce injury to the surrounding normal tissue, prolong the time of continuous exposure and more effectively control the growth of remnant tumor cells. But Koot et al.[7] treated primary glioblastomas cases with permanent implantation of 125I (DR, 2.5~2.9 cGy/h) or transient interposition of 192Ir (DR, 4.6 cGy/h), and compared the median survival time of the two groups. They suggested that there was no significant difference in the therapeutic effect between the two groups. Moreover the dose rate had no manifest relationship with the prognosis of the primary glioblastoma patients.
Now the method of seed implantation is mainly conducted by template implantation during an opera-tion under direct vision. The procedure has advantages of even distribution of the seeds and causes less injury to normal brain tissue, but it does not reduce the volume of the tumor, nor relieve the secondary lesion due to edema around the tumor and brain tissue displacement. The latter can decrease the high intracranial pressure and significantly relieve tissue edema because of the tumor resection. Studies have suggested that[8] the extent of tumor resection can greatly influence the prognosis of patients with a high grade glioma, especialy a glioblastoma. Our results support this concept. In addition, the reduction of the tumor size also allowed a decrease in the number of seeds and exposure rate to the surrounding tissue. Because of the lower cost and ease of application, the authors suggest that the implantation of seeds during an operation should be more widely used and selected preferentially when interstitial irradiation is required for the treatment of high grade glioma, especially glioblastoma.
At the present time, radioactive seed implantation during an operation still has had some apparent shortcomings. The plan of implantation can not be accurately divised before surgery due to the irregular tumor bed; also an even distribution of seeds can not also be ensured only based on placement with the naked eye. Surgeons just deviated from the key claims of the Paris principles. After removing the tumor as completely as possible, the tumor bed often was near the ventricular wall and/or important domains. In order to avoid aggravation of neural symptoms, it may be necessary to reduce the density of seeds, and even abandon seed implantation. In our study, the closer the seeds were to crucial domains, the more slowly the symptoms improved. This condition appeared on 4 patients with tumor recurrence within 6 months after operation. More attention should be used in selecting indications.
An important way to improve the therapeutic effect of radioactive seed implantation is to combine it with external radiotherapy[9], but there is no guide for their application. Necrosis of brain tissue due to radiation overdosage should be avoided as much as possible. It usually appears at the sixth month after external radiotherapy, resulting in brain tissue edema which can not, using only MR or CT scans, be discriminated accurately from a tumor recurrence. Of course PET, especialy 18F-FDG PET, can recognize non-tumorous tissue that has a lower relative metabolic rate. However it is often refused by many patients because of its cost. The dose of radiation from seeds must be determined before external radiotherapy is applied. Because of the influence of tissue edema, verification of the 125I dose should be conducted at the fourth week after implantation of the seeds.
In this study, the implantation of radioactive seeds certainly prolonged the patients survival time, reduced the tumor recurrence rate but failed to completely cure the patients. Before any new therapeutic method is well established, indications shoud be identified. Age, KPS score, tumor size and location are important factors worthy of consideration[10,11]. A broader consideration of indications might prevent observation of a curative effect, might make it difficult to interpret the results, and thereby hinder generalization of the technology.
- Received July 2, 2007.
- Accepted November 12, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer