Abstract
OBJECTIVE To investigate the clinical efficacy of three-dimensional conformal radiotherapy (3D-CRT) for locally advanced or postoperatively relapsed rectal cancer, and to examine the changes in cancer multi-biomarkers.
METHODS Sixty patients with locally advanced or postoperatively relapsed rectal cancer were randomly divided into two groups after 40 Gy external radiation, namely a late-course 3D-CRT group and a conventional radiotherapy group that served as the control. There were 30 patients in each group. For patients in the 3D-CRT group, multi-biomarkers were measured before and after radiotherapy and after relapse.
RESULTS Response rates in the 3D-CRT and the control groups were 86.7% (26/30) and 70% (21/30) respectively, without a significant difference (P>0.05). The 1-, 2- and 3-year survival rates were 80%, 53.3% and 36.7% in the 3D-CRT group; in the control group the rates were 56.7%, 40% and 13.3% respectively, with a significant difference (P=0.0213). CEA, CA19-9, CA242 and FER decreased after radiotherapy in the 3D-CRT group, P<0.01, indicating a significant difference. The values after relapse were higher than those without relapse, P<0.01, indicating a significant difference.
CONCLUSION Conventional radiotherapy with a 3D-CRT boost gives better therapeutic effect to patients with locally advanced or postoperatively locally relapsed rectal cancer. A multi-biomarker protein chip diagnosis system can be utilized as an effective tool to determine the therapeutic effect and prognosis.
keywords
INTRODUCTION
Rectal cancer is one of the most commonly observed malignancies. For postoperatively relapsed rectal cancer, the operability is rather low, making radiotherapy to be the primary treatment. However, conventional radiation can only provide a limited therapeutic effect, with a 5-year survival rate of less than 5%[1]. Aleman et al.[2], considered radiotherapy as one significant palliative tool to treat postoperatively relapsed or locally advanced rectal cancer. Three-dimensional-conformal radiotherapy (3D-CRT) is a novel radiotherapeutic technology emerging recently, which can converge the irradiation to a high-dose region, decrease the dose applied to normal tissues, and improve the local tumor control rate. This report describes the excellent therapeutic effect of late-course 3D-CRT and the changes in tumor biomarkers.
MATERIAL AND METHODS
Materials
Sixty patients with postoperatively relapsed or locally advanced rectal cancer, who had no possibility of surgical operation, were admitted in our hospital from December, 2003 to December, 2005. The patients were randomly divided into two groups as follows: a late-course 3D-CRT group and a conventional radiotherapy group as the control, with 30 patients in each group. Among all the patients, 39 were male and 21 female; 23 patients suffered from highly differentiated adenoma, 8 from highly-intermediately differentiated adenoma, 22 from intermediately differentiated adenoma, and 7 from intermediately-poorly differentiated adenoma. The patients’ ages ranged from 31 to 74, with a median age of 53; the Karnofsky Performance Status (KPS) was >70 for all the patients. Tumor diameters ranged from 1.5 to 8.7 cm (median, 4.9 cm).
Treatment methods
Treatment included two types in the 3D-CRT group: conventional anterior and posterior parallel field pelvic radiation and a 3D-CRT boost. The total dose of conventional radiotherapy was 40 Gy, with 2 Gy applied every time at a frequency of 5 times a week for 20 applications. Subsequently, patients received 3D-CRT as follows: the patients were stabilized on a vacuum cushion according to regulation, and then were scanned by CT. Images were transferred to a three-dimensional planning system. The gross tumor volume (GTV) was determined by cancer involvement on the CT image. The clinical target volume (CTV) was defined as the GTV plus adjacent sub-clinical loci, and planning target volume (PTV) was contoured with its margin extending 5~15 mm beyond the CTV. The median PTV volume was 517.8 cm3, ranging from 202.5 to 989.3 cm3. Organs and structures at risk, such as the bladder, colon and small intestine, were also contoured simultaneously. The field isocenter was defined as the geometric center of PTV, which was enclosed by a 90%~95% isodose line, and was conformed by field shape. The treatment plan was evaluated and optimized using a three-dimensional dose-distribution graph and dose-volume histogram (DVH). In accordance with the three-dimensional conformal plan, the linear accelerators (produced by VARIAN Inc.) and three-dimensional conformal treatment system provided multi-field isocenter coplane or non-coplane radiation, when the bladder was in a state of turgor. The total dose for the PTV was 25~30 Gy, with 2 Gy every time at a frequency of 5 times a week. After conventional anterior and posterior parallel field pelvic radiotherapy for 40 Gy in the control group, the total dose was escalated to 50~60 Gy with conventional parallel horizontal fields or an oblique field.
Measurement of tumor biomarkers
For all the patients in the 3D-CRT group, 3 ml of venous blood was obtained before breakfast, from 6 to 9 a.m.. Serum was separated and examined by a multi-tumor-biomarker protein-chip measurement system (C-12), which was produced by Huzhou Shukang Biological Technology Inc. Ltd., Zhejiang, China. The results were analyzed by a HD-2001A biochip measurement instrument, provided by the same company.
Evaluation of the therapeutic effect
The enhanced CT or MRI scan results were divided into complete remission (CR), partial remission (PR), stable disease (SD) and progressive disease (PD), among which CR and PR were defined as a response, according to the WHO criteria for therapeutic effects on solid tumors. The acute toxicity in normal tissue (the toxicity emerging in 90 days from the beginning of radiotherapy) was assessed by the RTOG criteria[3].
Reference scale of measured parameters
Carbohydrate antigen 19-9 (CA 19-9)<35 KU/L. Neuron specific esterase (NSE)<13 ng/ml. Carcinoembryonic antigen (CEA)<5 ng/ml. Carbohydrate antigen 242 (CA242)<20 KU/L. Ferritin (FER)<219 ng/ml. β-human chorionic gonadotropin (β-HCG)<3 ng/ml. Alpha-fetoprotein (AFP)<20 ng/ml. Free prostate specific antigen (f-PSA)<1.00 ng/ml. Prostate specific antigen (PSA)<5 ng/ml. Carbohydrate antigen 125(CA125)<35 KU/L. HGH<7.50 ng/ml. Carbohydrate antigen 153(CA153)<35 KU/L.
Follow-up
The patients were checked mainly by a physical examination, pelvic CT scan and blood C-12 before breakfast, every 3 months after therapy. Follow-up was conducted in our out-patient department. All of the 60 patients completed the therapy successfully. The follow-up rate reached 100% and the median duration was 25 months (ranging from 12 to 36 months). The survival time was dated from the beginning of the therapy.
Statistical analysis
Survival rate and local control rate were calculated using the Kaplan-Meier method with SPSS 10.0 software. Significance of difference was decided with the Logrank method, and other ratios were compared with the χ2 test.
RESULTS
Short-term results
The rates of symptom relief, such as local pain, sense of anal falling, frequent defecation, blood stool and swollen lower limbs, in the 3D-CRT group and the control group were 90% (27/30) and 80% (24/30) respectively, χ2=0.489 and P=0.4845; response rates in the conformal and the control group were 86.7% (26/30) and 70% (21/30) respectively, χ2=2.56 and P=0.1097.
Long-term results
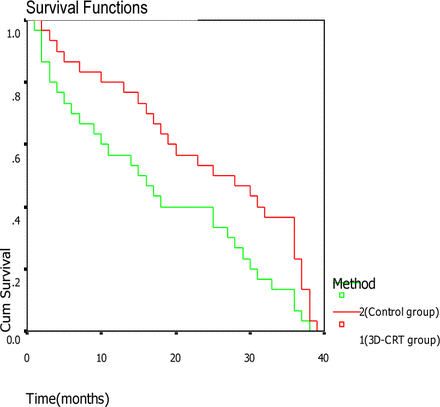
The 1-, 2- and 3-year survival rates were 80%, 53.3% and 36.7% in the 3D-CRT group. In the control group the rates were 56.7%, 40% and 13.3% respectively, χ2=5.30 and P=0.0213 (Fig.1). The 1-, 2- and 3-year local control rates were 86.7% (26/30), 80% (24/30), and 50% (15/30) in the 3D-CRT group and in the control group 73.3% (22/30), 53.3% (16/30) and 30% (9/30), with a median survival of 25 months vs. 15 months,χ2=4.07, P=0.0438.
Survival curve for patients receiving different treatments.
Adverse effects and complications
The short-term gastro-intestinal reaction, radiative rectitis and radiative cystitis were the main adverse effects, as noted in Table 1. Loperamide, antibiotics and gastro-intestinal bacterial adjustment alleviated these symptoms caused by the radiation.
Adverse effects and complications in the two groups [cases(%)].
Changes in tumor biomarkers in the 3D-CRT group
CEA, CA19-9, CA242 and FER decreased after radiotherapy in the 3D-CRT group, P<0.01, indicating a significant difference. The values after relapse were higher than those without relapse, P<0.01, also indicating a significant difference (Table 2).
Changes of 12 tumor biomarkers in the 3D-CRT group.
DISCUSSION
For rectal cancer, surgical resection alone results in a high recurrence rate. According to retrospective studies[1], the total recurrence rate of rectal cancer after surgery accounts for more than 40%, among which the post-treatment recurrence rate can reach as high as 45% to 65% for patients suffering from T3-4N1-2M0 disease. Death rates due to local relapse are about half of the 5-year mortality, and the other half results from distant metastasis. Except for a few cases with limited foci, the majority of the patients with postoperative local relapse have no chance to receive surgery. At present, no method of treatment has received a satisfactory consensus for relapsed or locally advanced patients who are not able to receive surgery. Radiotherapy is especially important for these patients[2,3]. However, high-dose radiation can lead to radiative enteritis, radiative cystitis, intestinal obstruction, and urethral injury, while a low-dose may not achieve a satisfactory therapeutic effect.
High-dose radiation can escalate the local control rate and tumor free survival rate. Rectal cancer is intermediately sensitive for radiotherapy, but for patients after surgery, radio-sensitivity decreases due to a damaged tumor bed, poor blood supply and an increase in hypoxic cells, requiring higher doses to control the tumor growth. The total dose of radical radiotherapy must be beyond 60 Gy; however, the tolerated dose for normal tissues, such as the small intestine and bladder, is only 50 to 60 Gy. The conventional radiotherapy extends the radiation field for normal tissue, limiting escalation of the dose to the tumor. 3D-CRT can localize a high dose of radiation to conform to the tumor target in three dimensions, thus protecting the adjacent normal tissues to the largest extent and reducing normal tissue complications. This accurate and reproducible technology allows elevation of the dose to the foci to more than 60 Gy, leading to increased local control, decreased distant metastasis, and consequently improved survival.
The short-term response rate of the 3D-CRT group in our study was 86.7%, with 1-, 2-, and 3-year local control rates of 86.7%, 80% and 50%, and 1-, 2- and 3-year survival rates as 80%, 53.3% and 36.7%, consistent with a report by Wu et al.[4] Compared with the control group, the 1-, 2-, and 3-year local control rates as well as the 1-, 2- and 3-year survival rates differed significantly (P<0.05), resulting in a better therapeutic effect in the 3D-CRT group. Because a CT scan at present is not able to detect sub-clinical lesions with a diameter of less than 1 mm (a sub-clinical lesion requires only 2/3 of the dose of radical radiation), we used conventional radiotherapy to eliminate sub-clinical tumors in the first 4 weeks, so as to avoid tumor oversights, and applied 3D-CRT in the late course. With regard to occurrence of adverse effects, such as gastro-intestinal reactions, radiative rectitis and radiative cystitis in the 3D-CRT group, they did not differ significantly with the control group (P>0.05). Incidence of toxicities in the 3D-CRT group was not lower than the control group, perhaps because of irradiation in the initial conventional radiotherapy for 4 weeks, and the higher total irradiation in the 3D-CRT group. Therefore, whether full-course conformal radiotherapy can decrease side effects should be considered further.
Baglan et al.[5] suggested that the DVH curve for the small intestine above 25 Gy was relatively flat with conventional radiotherapy, whereas it continuously declined with 3D-CRT, demonstrating progressively diminishing volume of the small intestine in a higher dose region. The steep change of the DVH curve at the field border has an obvious different biological effect from conventional radiotherapy. Myerson et al.[6,7] concluded that factors causing radiative enteritis after radiation for rectal cancer included: volume of the small intestine receiving >40 Gy was more than 150 ml and the gross tumor volume was more than 800 ml; PTV more than 800 ml was the factor causing radiative rectitis. Radiative enteritis in our research was mainly related to a large radiation volume, as all of the 12 cases with radiative enteritis were more than 600 ml.
In adjuvant diagnosis for rectal cancer, i.e. CEA, CA19-9 and CA242 are the most widely used tumor biomarkers. Sensitivity of these markers in late-stage rectal cancer is much higher than in early stage, and they show a significant difference before and after treatment. In addition, their levels increase dramatically when metastasis or relapse occurs. Detection methods for serum biomarkers include enzyme-linked immunoassays, radio-immunity tests and electro-chemical fluorescence. These methods measure only one parameter at one time, are costly and a large amount of serum is needed for multi-parameter detection. Solitary parameter detection has the disadvantage of low sensitivity, whereas multi-parameter measurements can solve this shortcoming. Analysis using protein chips can be conducted in miniature and is of high quality, sensitivity and high specificity[8]. Deng et al.[9] reported that serum CEA, CA19-9 in combination with CA242 for rectal cancer can reach 83.3% positivity. Our research found that, CEA, CA19-9, CA242 and FER are meaningful biomarkers, exhibiting remarkable significance before and after radiotherapy and after relapse, consistent with the findings of Sun et al.[10] Accordingly, tumor biomarkers can be utilized as important parameters for determining the therapeutic effect and prognosis in order to disclose problems promptly.
It can be concluded from this research that 3D-CRT provides a better therapeutic effect for inoperable patients with postoperatively locally relapsed or locally advanced rectal cancer compared to conventional radiotherapy. The adverse effects are tolerable, indicating that 3D-CRT provides clinical superiority. A multi-biomarker protein chip diagnosis system can be utilized as one important tool to determine the therapeutic effect and prognosis.
Footnotes
This work was supported by a grant from Scientific Foundation of the Health Department, Hunan Provence (No. B2004-038).
- Received July 4, 2007.
- Accepted November 20, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer