Abstract
OBJECTIVE To study the clinical value of hepatoma-specific alpha fetoprotein (HS-AFP) in the diagnosis and differential diagnosis of hepatocellular carcinoma (HCC).
METHODS A method of vertical slab polyacrylamide gel electrophoresis with discontinuous buffer system was developed to separate AFP subtypes. After separation, the AFP subtypes were transferred to nitrocellulose and reacted first with rabbit anti-human AFP and then with goat anti-rabbit IgG-HRP. Finally, AFP subtypes were visualized by reacting with 3,3'-diaminobenzidine tetrahydrochloride. A HS-AFP band was determined in 82 cases with HCC and 95 cases with benign liver diseases. The correlations between the positive rates of HS-AFP and serum AFP concentration, tumor size as well as portal vein metastasis were analyzed.
RESULTS Serum AFP in the cases with various liver diseases was separated into one to several bands. The fastest band on electrophoresis (F-AFP) was found in all patients, while the band at the cathodal site (HS-AFP) was detected predominantly in HCC but rarely in benign liver diseases. The positive rate of HS-AFP in HCC was 74.4%, which was significantly higher than that in benign liver diseases (9.1%, 7.3% and 10.0% in liver cirrhosis, chronic hepatitis and acute hepatitis respectively). HS-AFP was detected in 3 out of 9 HCC cases with AFP <50 μg/L, but in none of 22 cases of benign liver diseases with the same AFP concentration. HS-AFP correlated with serum AFP concentration and tumor size to some extent, but not with portal vein metastasis.
CONCLUSION HS-AFP increases the sensitivity of diagnosing HCC in patients with negative AFP, and is useful in distinguishing high AFP due to HCC from that caused by benign liver diseases.
keywords
Hepatocellular carcinoma (HCC) is a common malignant tumor worldwide and the number 2 cause of cancer mortality in China.[1] It often develops in cases of liver cirrhosis and chronic hepatitis.[1-3] Advanced imaging procedures including utrasonography, helical computed tomography and magnetic resonance imaging have facilitated the diagnosis of HCC. However, it is difficult to make the diagnosis in some HCC cases if typical imaging changes are not found, or if the tumor is very small. There are several useful serum markers for HCC, such as alpha-fetoprotein (AFP),[4-7] des gamma carboxypro thrombin,'[8-11] gamma-glutamyltransferase isoenzyme,[12], [13] alpha-L-fucosidase [14-16] and glycylproline dipeptidyl aminopeptidase isoenzyme [17] etc. Among them serum AFP is the most widely used. However, some benign liver diseases may also display elevated serum AFP levels. Therefore, it is very important to differentiate increased AFP due to HCC from that caused by benign liver diseases. It has been demonstrated that there is microheterogeneity in serum AFP.[18-22] Different AFP glycoforms show different affinity to lectins and have different isoelectric points. The HCC associated AFP isoform can be separated with lectin affinity electrophoresis or isoelectric focusing electrophoresis.[23-28] Both of these 2 methods are complicated and require expensive reagents. In the present study we have successfully detected an HCC associated AFP isoform with a newly-developed polyaciylamide gel electrophoresis combined with Western blots. By studying 82 cases with HCC and 95 cases with benign liver diseases, we found that an HCC associated AFP isoform detected by this method is very useful in the diagnosis and differential diagnosis of HCC.
Materials and Methods
Patients
Eighty two cases with HCC and 95 cases with benign liver diseases were enrolled in this study from May 2004 to January 2005 in the Affiliated Hospital of Nantong University, Nantong Tumor Hospital and the Third People’s Hospital of Nantong. The diagnosis of HCC was confirmed by pathological examination or needle biopsy in 30 cases. The other 52 cases were diagnosed by characteristic imaging changes and other clinical data. Patients with an equivocal diagnosis of HCC were excluded from this study. Liver cirrhosis and hepatitis were diagnosed according to laboratory results and imaging examinations. The differential diagnosis was made by needle biopsy in equivocal cases of liver cirrhosis and chronic hepatitis. All the cases with benign liver diseases had been followed up for at least 8 months without development of HCC. The sizes of the tumors were measured by CT or ultrasonography.
Separation and determination of HCC associated AFP isoform
Vertical slab polyacrylamide gel electrophoresis with a discontinuous buffer system was developed to separate AFP subtypes. The concentrations of stack gel and separating gel were 5.0% and 8.8% respectively. Trisborate (0.038 M, pH 7.8) and Tris-HCl (0.06M, pH7. 5) were used as cathodal and anodal buffers respectively. Serum (15 p,l) was loaded at the cathodal side and electrophoresis was performed at 60 V for 1 h and then at 100 V for another 3.5 h. The AFP bands separated were then transferred to nitrocellulose with the current of 1 mA/cm2 for 3 h. The transferred nitrocellulose was then blocked with 5% nonfat milk PBS at 37 °C for 1 h and incubated with 1:80 rabbit anti-human AFP (DAKO Company, Denmark) at 4 °C overnight. After that, the nitrocellulose was incubated with 1:80 goat anti rabbit IgG HRP (Hua-Mei Bioengineering Company, China) at room temperature for 2 h. Finally separated AFP bands were visualized by reacting with 3,3'diaminobenzidine tetrahydrochloride.
Quantitation of AFP
AFP was analyzed with a chemoluminescence test.
Statistical analysis
The Chi-square test was used and P<0.05 was considered as statistically significant.
Results
Separation of AFP subtypes with electrophoresis
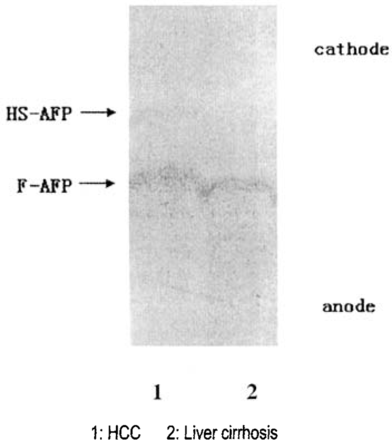
Serum AFP in the cases with various liver diseases was separated into one to several bands. The fastest band on electrophoresis at the anode was called fast AFP band (F-AFP), which was positive in all patients. F-AFP was the only AFP band in most benign liver diseases. At the cathodal side, another AFP band often occured in cases with HCC, but rarely in other patients, so we called it hepatoma specific AFP (HS-AFP). Occasionally, there might be some other AFP bands between F-AFP and HS-AFP or at the cathodal side of HS-AFP, but all of them were very weak and not specific to diseases (Fig.1).
Serum AFP subtypes separated by polyacrylamide electrophoresis.
HS-AFP and AFP concentrations in various livei diseases
Most patients with benign liver diseases selected in the present study had elevated AFP and some of them AFF concentrations higher than 200 μg/L. There was nc difference in the sensitivity of diagnosing HCC between HS-AFP and AFP. But positive rates of HSAFF in liver cirrhosis and hepatitis were significantly lowei than that of AFP (Table 1).
HS-AFP and AFP in various liver diseases
Relationship between serum AFP and HS-AFP
Serum AFP concentrations and positive rate of HS-AFP were compared and it was shown that the positive rate of HS-AFP in cases with AFP<50 μg/L was significantly lower than that in cases with AFP>50 μg/L, whether in HCC or in benign liver diseases. However, there was no statistical difference of positive rates of HS-AFP between the cases with AFP 50-500 μg/L and those with AFP>500 μg/L (Table 2). In the group of patients with AFP<50 μg/L, positive HS-AFP was detected in 3 out of 9 cases with HCC but in none of 22 cases with benign liver diseases.
Relationship of AFP concentrations and positive rate of HS-AFP
Relationship between tumor sizes and positive rate of HS-AFP
The cases with HCC were divided into 2 groups based on the tumor sizes: one group with the diameter less than 5 cm and the other larger than or equal to 5 cm. The positive rates of HS-AFP in the cases with larger and smaller tumors were 81.2% (56/69) and 38.4% (5/13) respectively. There was a statistical difference of positive rates of HS-AFP between the 2 groups (P<0.01).
Relationship between HS-AFP and portal vein thrombus
HCC was divided into 2 groups: one with, and the other without portal vein thrombus. The positive rate of HS-AFP in HCC with portal vein thrombus was 88.9% (24/27), and that in HCC without portal vein thrombus was 67.2% (37/55), there being no statistical difference between the 2 groups.
DISCUSSION
The approach for HCC diagnosis includes imaging techniques for tumor localization and tumor markers for determining its malignant nature. Serum AFP is the most widely used tumor marker for HCC. However, benign liver diseases such as liver cirrhosis and chronic hepatitis sometimes may also be associated with elevated serum AFP.[29-30] Moreover, more than 80% of HCCs arise in patients with preexisting liver cirrhosis. [2,3] Therefore, it is necessary to differentiate HCC from benign liver diseases in the cases with elevated serum AFP concentrations.
It has been known that AFP shows microheterogeneity because of structural variance in its composition of sugar chains.[18,21,22] The sugar chains in the AFP isoform produced by HCC have higher affinity to lectins than that produced by benign liver diseases. Therefore, HCC-associated AFP isoform can be isolated by lectin affinity chromatography or affinity electrophoresis. [23-26] Lens culinaris agglutinin-reactive alpha-fetoprotein (AFP-L3) is useful in the early diagnosis of HCC,[31-33] the diagnosis of tumor recurrence[34]and distant metastasis,[35] the differential diagnosis between HCC and benign liver diseases,[23] and prognosis. [36-37] HCC associated AFP can be separated by isoelectric focusing electrophoresis. [27,28,38] Because of high cost and complicated procedures of lectin affinity electrophoresis and isoelectric focusing electrophoresis, HCC associated AFP has not been widely used in clinical practice.
In the present study we have developed polyacrylamide gel electrophoresis with a discontinuous buffer system and successfully separated HS-AFP, which was clearly detected by Western blots. Compared with detection of AFP-L3, this method is simple. HS-AFP is qualitative and the band can easily be judged with the naked eye, whereas AFP-L3 is quantitative and a densitometer is required for the assay. Moreover, this method is much cheaper than lectin affinity electrophoresis and isoelectric focusing electrophoresis because of no expensive reagents in the assay.
In this study, we identified 2 main AFP bands. The anodal band was found in all cases, whereas the cathodal band (HS-AFP) was predominantly in cases with HCC and rarely in benign liver diseases. Clinical studies have shown that HS-AFP is a more specific tumor marker than serum AFP in the diagnosis of HCC. Three out of 9 cases with HCC with negative AFP (<50 μg/L) displayed the HS-AFP band, while none of the 22 cases with benign liver diseases with negative AFP had HS-AFP, suggesting that HS-AFP may increase diagnostic sensitivity to HCC in the patients with negative AFP.
Most cases with HCC have underlying cirrhosis or chronic hepatitis. [1-3] Elevated serum AFP may be caused by both HCC and the underlying benign liver diseases. It is particularly difficult to distinguish HCC from benign liver diseases in cases with serum AFP concentrations between 50μg/L to 500μg/L. In this study, the positive rate of HS-AFP was 75% in HCC but only 10.9% in benign liver diseases with serum AFP concentrations of 50-500μ/L. These results indicate that HS-AFP is of great value to the differentiation between HCC and benign liver diseases. Because HCC often develops in the cases with cirrhosis and chronic hepatitis, whether the false positivity of HS-AFP in benign liver diseases represents a tendency for hepato carcinogenesis needs to be clarified by further studies. The structure of HS-AFP has not been clarified, and whether HS-AFP is the same as or similar to AFP L3 needs to be elucidated.
HS-AFP is supposed to originate from cancer tissues because the frequency of HS-AFP increases as the tumor size increases. However, HS-AFP was also found in 38.4% cases with the tumor size less than 5 cm. Moreover, there was no correlation between the positive rate of HS-AFP and portal vein metastasis, indicating that HS-AFP is helpful for early diagnosis of HCC.
Taken together, HS-AFP is a sensitive and specific marker for the diagnosis of HCC. It is useful for differentiation between HCC and benign liver diseases with elevated serum AFP concentrations. The method for HS-AFP assay is cheap and easy so that it is suitable for clinical application.
Footnotes
This work was supported by grants from Jiangsu Science and Technology Department (No. BS2004528) and Jiangsu Health Department (No. H200521).
- Received May 9, 2006.
- Accepted June 15, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer