Abstract
OBJECTIVE To investigate the efficacy and side effects of high intensity focused ultrasound(HIFU) in the treatment of malignant solid tumors.
METHODS Thirty patients who refused surgery and/or were refractory to chemotherapy were treated by HIFU alone, with the efficacy and side effects monitored as follows: observation of vital organ signs; functional assay of important organs; imaging examinations including: digital subtraction angiography (DSA), CT, MRI, single photon emission computed tomography (SPECT), large core needle biopsy, complications and metastasis.
RESULTS After HIFU therapy, the vital signs remained stable and the functions of the heart, lung, kidney and liver were also normal. DSA images showed that small or larger arteries were not damaged. After a follow-up of 10–38 months(mean 23.1 months), 26 patients(87%) were alive. The volume of the tumor underwent complete regression in 10 patients. Shrinkage of the tumor volume ≥50% was observed in 13 patients. Eight of 13 patients were examined by large core needle biopsy, all showing necrosis and /or fibrosis though 3 patients(10%) had local recurrence. Two of these were retreated again by HIFU and the locally recurrent tumors were controlled. New metastases developed in 5 patients after HIFU. Two patients suffered from peripheral nerve injuriy and they have recovered during the follow-up. One patient developed skin injury.
CONCLUSION High intensity focused ultrasound is effective and safe in the treatment of malignant solid tumors.
keywords
High intensity focused ultrasound (HIFU) is a novel noninvasive technique developed recently for the treatment of malignant solid tumors. Numerous animal experiments have shown that treating malignant tumors with HIFU was effective [1–3]. From January 1998 to June 2000, thirty patients with malignant tumors, who refused surgery or chemotherapy, and/or who were refractory or could not tolerate chemotherapy, were treated by HIFU. The therapeutic effects and side effects of HIFU are reported.
MATERIALS AND METHODS
In this study, 20 male and 10 female patients, ages 14–72 (mean 48.5) years, were treated with HIFU. The tumor histology and stage are described in Table 1. Among them, 14 patients had a local recurrence after surgical operation, 7 with metastases and 9 with primary tumors. All cases were diagnosed by pathological examination.
Tumor histology and stage of 30 patients
After receiving durative epidural anesthesia or general anesthesia, the patients were immobilized on a treatment bed and treated with a Haifu focused ultrasound tumor therapeutic system (developed and produced by Chongqing Haifu Technology Co., Ltd.). Based on the principles used for tumor surgery, the region to be treated, and that covering the tumor mass and some surrounding normal tissue, was identified. Frequencies of 0.8–1.6 MHz, focal lengths of 90–135 mm and treatment time of 3,460–12,865 s were used. During the procedure, real-time ultrasonography was used to monitor the treatment process and adjust the therapeutic dose. The criterion for stopping a treatment session was the obvious echo enhancement of targeted tissue on ultrasonic imaging.
Before and two weeks after HIFU therapy, computed tomography (CT) or magnetic resonance imaging (MRI), and 99mTc–MIBI or 99mTc–MDP single photon emission computerized tomography (SPECT) were performed to verify the treatment range of the HIFU and to determine whether the treated region included the expected target. A re–treatment was necessary to cover the entire tumor mass, if the previous treatment failed to ablate the whole tumor mass. CT or MRI imaging showed that intensification in the treatment region disappeared and there was a sharp and regular intensified ring surrounding the target (Fig. 1, 2). 99mTc–MIBI or 99mTc–MDP SPECT examinations showed that there was no radionuclide uptake area, forming a cold radioactive region (Fig.3).
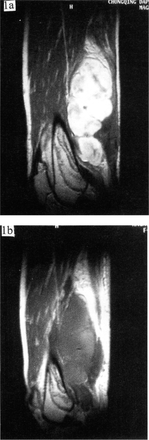
Enhanced MRI changes before and after HIFU; 1a The tumor mass was obviously enhanced before treatment;1b Two weeks after HIFU, the tumor mass shrank with a loss of enhancement.
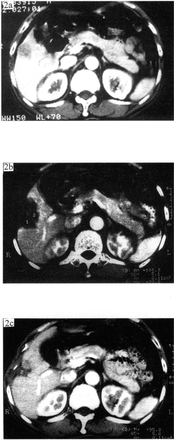
CT image changes before and after HIFU;2a Before treatment; 2b Two weeks after HIFU, CT showed the low-density area without intensification had overlaid the tumor mass; 2c Six months after HIFU, the treated region shrank markedly.
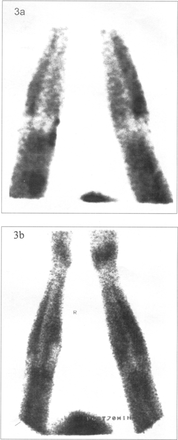
99mTc–MIBI before and after HIFU; 3a There was radionuclide uptake area in the tumor mass before treatment; 3b The radionuclide uptake disappeared showing a cold radioactive area.
In the follow–up period, color Doppler ultrasound examinations and chest X ray imaging were performed every 3 months. CT or MR images, and 99mTc–MIBI or 99mTc–MDP SPECT examinations were performed every 6 months. The examination items which were followed closely included tumor size changes, local recurrence and metastasis, pain relief and effects of HIFU to vital organs and blood vessels in the vicinity that was treated. The tumor size was calculated by the product of the maximum cross diameter and vertical diameter of the tumor mass. Changes of tumor blood vessels were evaluated by monitoring blood flow in the extremities with color Doppler flow imaging (CDFI) and by digital subtraction angiography (DSA) before treatment and 3–10 months after HIFU. Within three days after HIFU treatment of breast cancers and tumors in the chest and back, electrocardiography was continuously performed to observe changes of breathing and hemoglobin saturation. The patients received a myocardium zymogram and chest examination to check the effects of HIFU on heart and lung function. Before treatment and on the 3rd, 7th and 14th day after HIFU, the patients were given liver and kidney function examinations, and the functional changes analyzed. All measurement data underwent t testing.
RESULTS
A total of 43 sessions of HIFU were given to 30 patients, among which 15 patients received 1 session, 11 received 2, and 4 received 3 (mean 1.4 sessions). In 29 cases, the tumor mass was completely ablated, but one with a liposarcoma failed to be entirely diminished after 3 sessions of HIFU. In fifteen patients with extremity tumors, no blood flow changes were found during treatment. Eight of these patients had DSA examinations 3–10 months after HIFU, indicating that tumor staining had disappeared and that arteries larger than 2 mm in diameter had remained intact compared to their condition before treatment (Fig.4). Electrocardiography, breath, myocardium zymogram and chest X ray examinations showed no abnormity. The changes of liver and kidney function of all patients were not remarkable (P>0.05).
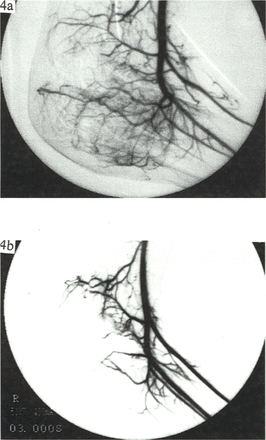
Changes of DSA before and after HIFU; 4a Numerous tumor vessels were seen before HIFU; 4b Abnormal tumor vessels disappeared.
Before HIFU, 8 patients had suffered cancerous pain, one of them with severe pain, 4 with moderate pain and 3 with mild pain. One week after HIFU, their pain disappeared completely.
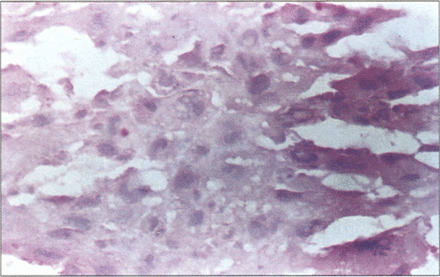
A follow-up study of 10–38 months (mean 23.1 months) showed that 4 patients had died; among them 3 died of lung metastasis with 2 having lung metastasis before the HIFU procedure. One had a thoracic spine metastasis with extensive paraplegia and died of a lung infection. Twenty–six patients survived with a rate of 87%. Among them, complete tumor–mass disappearance was found in 10; over 50% shrinkage of their tumor mass was found in 13 patients, with the imaging examination showing no sign of local recurrence. Large core needle biopsy of 8 patients with a shrunken tumor mass revealed necrosis of tumor cells and/or fibroid tissue (Fig. 5). In 3 cases the tumor mass shrank, but local recurrence was proven by imaging. The recurrent time was 6–13 months after HIFU therapy and the recurrent rate was 10%. Re-treatment was given to 2 of the three patients and after 11–14 months follow-up, the tumor was found locally well controlled. After HIFU therapy, new metastases were found in 5 cases, among them, 2 with lung metastasis, 2 with vertebral column metastasis and one with rib metastasis.
One year after HIFU, tumor tissue treated was pathologically proven to be necrosed and only the rough outline of necrosed cells could be seen. HE×400.
In 2 of the 30 patients, injuries were found of the common peroneal nerve and ulnar nerve. During the 12–30 months follow-up, complete functional recovery of the common peroneal nerve was achieved and most functional recovery of the ulnar nerve was achieved. One patient with malignant neurilemoma was found to have local cutaneous necrosis after HIFU, which was healed by external drug application three months later.
DISCUSSION
Mechanism and efficacy
In HIFU technology, extracorporeally low power ultrasound beams, which may be focused on deep–targeted tissue in the human body, are utilized. The focused high intensity ultrasonic beams interact with and destroy the target tissue with transient hyperthermia and cavitation, resulting in coagulative necrosis of the target without damaging the tissue in the vicinity Hyperthermia refers to temperature rising within the targeted region reaching 60–100°C in a few seconds [4,5] HIFU may cause contraction and occlusion of minor blood vessels and injury to the epithelium, causing the formation of thrombi which prevent the vessels from supplying blood to the tumor, and thereby prevent metastasis growth of the tumor. It may also stimulate the anti–cancer immunity of the body and inhibit the growth of residual tumor tissue [7–9]. Most patients in this group were at a late stage of tumor progression. After a mean follow-up period of 23.1 months, most treated tumors were completely absorbed or underwent shrinkage, and only three had recurred, showing the local tumor-control effect of HIFU. As a candidate modality for the treatment of cancer patients who have no chance for receiving conventional therapeutic measures, further studies of the therapeutic effects of HIFU and clinical protocols for various types of tumor are needed.
Indications and contraindications
Theoretically, HIFU could be used to treat most types of malignant tumors of the parenchyma[6,10]. Since HIFU is now in a early period of clinical application, it is recommended that patients be treated with malignant tumors that can be visualized with B mode ultrasound. Candidates for HIFU are those who can not endure surgery, who reject surgery, who have a recurrence after surgery or have residual tumor tissue after surgery. On the contrary, for those patients who can not endure anesthesia, or where there is air–containing tissue or bone within the acoustic pathway, or where the superficial tissue of the tumor has disrupted, or if there is an expectation that the residual normal tissue of a vital organs can not remain viable, it is inadvisable to utilize HIFU therapy. Liver cancer behind the ribs or a tumor at the top end of the kidney could be treated after removing part of the ribs. As for various types of tumors, the indications of HIFU need to be further discussed and studied.
Safety and complications
By observing the influence of HIFU therapy to vital structures adjacent to the targeted and systemic vital organs, we can draw the conclusion that HIFU is safe. There were two nerve injuries in the experimental group, likely relevant to the difficulty of monitoring ultrasound to visualize the nerve. Nerve may have a higher sensitivity to HIFU therapy, although the mechanism remains unknown. One patient with a malignant neurilemoma was found to have a recurrence in the incision after surgery; skin injury, disruption and secondary infection occurred after HIFU therapy, which were cured by anti–infection drug application. In case the subcutaneous tissue and skin are involved, it must be very cautious in using HIFU therapy for treatment. It is a critical safety issue as to whether HIFU therapy augments tumor metastasis or not. A few scholars think HIFU does, but most differ in opinion since they believe tumor metastasis after HIFU relates to incomplete ultrasonic coverage [3,4,5]. In our study, 5 patients were found to have new metastasis after the tumor foci were completely scanned by HIFU. Since there was no obvious augmentation of metastatic lesions, it was concluded that HIFU does not enhance tumor metastasis; however, more randomized controlled trials(RCT) are needed to validate this conclusion.
Recent studies have shown that surgical trauma may foster the growth and metastasis of tumor and the degree of metastasis is positively related to the degree of traum degree [14,15]. There is a trend to explore new noninvasive tumor treatment methods. HIFU is a technique employing extracorporeally thermal ablation of the tumor tissue, a procedure which has a number of advantages. For example, firstly, it is a noninvasive 3-D conformal treatment causing minimal damage to the surrounding normal tissue; secondly, it can be applied to tumors of different size and shape (diameters in our study ranging 2–16cm). Thirdly, it result in no harm to blood vessels over 2 mm in diameter, offering chances for the treatment of a tumor located in the neighborhood of larger blood vessels. Fourthly, it is safe, reliable and has few complications.
Footnotes
This study was supported by the Key National “Ninth-five” Scientific and Technological Breakthrough Program (Grant No. 96-905-02-01), and the National Natural Science Foundation of China (Grant No. 39770841).
- Received January 30, 2004.
- Accepted March 29, 2004.
- Copyright © 2004 by Tianjin Medical University Cancer Institute & Hospital and Springer