Abstract
Wnt signaling has been shown to engage a multifunctional pathway that is involved in the regulation of a wide variety of normal and pathologic processes, including embryogenesis, differentiation and tumorigenesis. Recent studies have demonstrated that Wnt5a expression is frequently seen in various human cancers. In contrast to the transforming members of the Wnt family, shown to be upregulated in many cancers, the role of Wnt5a is still controversial in its expression in different tumors. There is increasing evidence that Wnt5a has tumor suppressor function in some malignancies, and in addition, it elicits promigratory and proinvasive effects via the planar cell polarity pathway, which suggests that Wnt5a might be an effective marker for the progression and prognosis of tumors. Obviously, the outcome of Wnt5a signaling is dependent on a multitude of variables, ranging from receptors, downstream effectors and inhibitors, to external influences coming from the tumor microenvironment. This review will focus on the role of Wnt5a signaling and, as a consequence, provide an outline describing the expression and functions of Wnt5a in cancer progression.
keywords
Introduction
The Wnt family of signaling molecules regulates numerous processes in animal development and has increasingly been implicated in tissue homeostasis and is involved in a variety of diseases[1,2]. Wnt proteins are secreted from cells and act on target cells through a pathway that, compared to other signaling pathways, is unusually complex and subject to extensive feedback control. Wnt5a is a member of the Wnt family of proteins, which are 38 - 45 kDa secreted cysteine-rich proteins with hydrophilic signal peptides. Wnt5a lacks transforming activity[3,4], and it does not typically signal via the canonical β-catenin pathway. Recent data provide strong evidence that Wnt5a also plays an essential role in cancer, contributing not so much to its initiation but more to malignant progression, despite the lack of comparatively well-defined clinical models. However, the exact nature of this role, either as a tumor suppressor or a promoter, is still controversial. This review will focus on the Wnt5a signal transduction pathway in tumor development.
Mechanisms of the Wnt5a signaling pathway
Wnt proteins constitute a large family of cysteine-rich secreted molecules[5]. To date, at least 19 Wnt members are present in mammals. The members exhibit unique expression patterns and distinct functions in development. The members of the Wnt protein family can be divided into distinct types based on their ability to induce transformation of the mouse mammary epithelial cell line C57MG[3,6]. The high transforming members include Wnt1, Wnt3a, and Wnt7a. The intermediate transforming members include Wnt2, Wnt5b, and Wnt7b and the non-transforming members are Wnt4, Wnt5a, Wnt6, and Wnt11. Wnt5a has been classified as a so-called noncanonical ligand, which uses alternative signaling pathways in contrast to the canonical Wnt1-type ligands. Signal transmission through either of these pathways has been considered mutually exclusive. However, recent data suggest a much more complicated transduction pattern.
The canonical pathway is activated by simultaneous binding of Wnt1-type ligands to receptors of the Frizzled (Fz)-family[7-9]. Activation of the receptor leads to the phosphorylation of the dishevelled protein which, through its association with axin, prevents glycogen synthase kinase 3β (GSK3β) from phosphorylating critical substrates [10,11]. The GSK3β substrates include the negative regulators, axin and APC, as well as β-catenin itself[12]. Unphosphorylated β-catenin escapes recognition by β-TRCP, a component of an E3 ubiquitin ligase, and translocates to the nucleus where it engages transcription factors such as TCF and LEF[13], which are involved in cancer development. Additional components in the pathway include casein kinases I and II, both of which have been proposed to phosphorylate dishevelled[14]. The serine/threonine phosphatase PP2A associates with axin and APC, although its functional role in the pathway remains obscure[15].
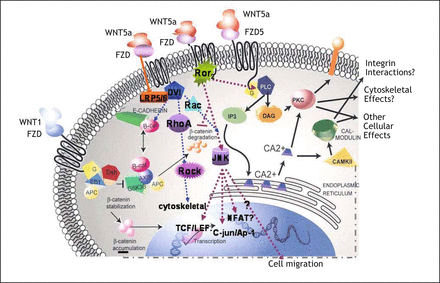
Noncanonical signaling from Wnt5a activates the planar cell polarity (PCP) and the Wnt/Ca2+ pathway. The noncanonical Wnt/Ca2+ pathway is triggered by Wnt4, Wnt5a, and Wnt11, and it induces intracellular Ca2+ release. This stimulates protein kinase C (PKC), a nuclear factor which is associated with T cells (NFAT) or stimulates calmodulin-dependent kinase II (CamKII)[16]. Other noncanonical Wnt signals activate small Rho-GTPases and regulate cytoskeletal architecture and cellular polarity. Channeling of Wnt5a-signals into the PCP pathway is mediated by the PDZ and DEP regions of Dvl and results in activation of the small Rho-GTPases RhoA and Rac[17]. They, in turn, induce the Rho-associated kinase (ROCK), leading to reorganization of the cytoskeleton, as well as activation of the Jun-N-terminal kinase (JNK). The latter phosphorylates c-jun and transcriptionally activates AP-1. Wnt5a-triggered PCP signaling mediates epithelial cell polarity in Drosophila development and convergent extension movements during vertebrate gastrulation[18,19]. There is growing evidence that noncanonical Wnt5a signaling can interact with canonical pathway in several ways. First, Wnt5a can bind to the receptor tyrosine kinase Ror2[20,21], which induces Rho GTPase-independent activation of JNK and also inhibits canonical signaling[22]. Second, Wnt5a performs phosphorylation of c-jun by JNK to act synergistically with TCF/LEF on the promoter of the canonical target gene c-jun[23]. Third, Wnt5a has even been shown to be able to directly activate canonical signaling by simultaneous overexpression of Wnt5a and Fz5 in Xenopus[24]. These apparently contradictory findings suggest that the function of Wnt5a is modified by the availability of receptors and other mediators of the signaling cascade and by the presence of natural inhibitors or by substances in the tumor stroma (Fig. 1).
Expression of Wnt5a in various cancer tissues
Aberrant Wnt5a mRNA and/or protein expression has been demonstrated in a variety of cancers of epithelial and mesenchymal origin. However, data on the expression of Wnt5a in cancer tissue is contradictory. Based on the results from previous research, we summarized the expressions of Wnt5a in different cancer tissues.
Melanoma
In the previous study on Wnt5a RNA expression in tumors, Iozzo et al.[25] identified that, on a gross level, many tumors showed increased Wnt5a expression relative to their normal tissue of origin and that melanomas showed increased Wnt5a expression relative to skin and to its correlation to outcome. Bittner et al.[26] detected Wnt5a gene expression in tumor tissues and cell lines by using microarrays and revealed that Wnt5a gene expression increased significantly. They showed that Wnt5a gene expression was one of the most robust markers of aggressive behavior in cutaneous melanomas, which was supported by the detection of a direct correlation between immunohistologic Wnt5a positivity and high tumor grade. Weeraratna et al.[27] examined via tumor microarrays that Wnt5a expression in human melanoma biopsies directly correlated to increasing tumor grade. These observations support a role for Wnt5a in human melanoma progression. Furthermore, the authors found that a dramatic increase in activated PKC indirectly correlated with Wnt5a expression and PKC activation, and that there was an increase in melanoma cell invasion. Blocking this pathway using antibodies to Frizzled-5, the receptor for Wnt5a inhibited PKC activity and cellular invasion. In this study, the authors also showed that the increased expression of Wnt5a in malignant melanoma is localized, occurring in cells at the site of active invasion and in cells showing morphologic features associated with aggressive tumor behavior. This stimulus has also been shown to increase the motility of melanoma cells in melanoma in vivo[28]. In the recent study by Bachmann et al.[29], the authors found the contrary conclusion that the expression of Wnt5a and Frizzled receptors were decreased in the nucleus of melanoma cells.
Tumors of the digestive system
In a recent study, Kurayoshi[30] revealed that abnormal expression of Wnt5a was observed in 71 of 237 gastric cancer cases by means of immunohistochemistry. They found that positivity of Wnt5a expression correlated with advanced stages and with poor prognosis of gastric cancer. Wnt5a had the abilities to stimulate cell migration and invasion in gastric cancer cells. Cell migration, membrane ruffling, and turnover of paxillin were suppressed in Wnt5a knockdown cells. Furthermore, anti-Wnt5a antibody suppressed gastric cancer cell migration. Liu et al.[31] indicated that the expression of Wnt5a protein and RNA were lower in HCC tissue in comparison to normal tissue. Expression correlated with the aberrant performance of β-catenin and E-cadherin. Ripka et al.[32] demonstrated that the expression of Wnt5a RNA and protein in pancreatic cancer was higher in malignant tissue and cell lines when compared to that in normal tissues. Wnt5a was identified as a downstream target of the homeobox transcription factor CUTL1 in pancreas carcinoma cell lines, where it mediated CUTL1-induced enhanced migration and invasion. Dejmek et al.[33] found that patients who had the lower Wnt5a expressions had shortened survival in stage Dukes B colon cancer. The same correlation between Wnt5a expression and advanced tumor stage with higher rate of lymph node involvement was found in colon cancer[34].
Breast cancer
In an early study, Olson[4] showed that Wnt5a was associated with decreased proliferation in mouse mammary cells. The inverse correlation between expression of the endogenous Wnt genes and cell proliferation suggests that Wnt4 and Wnt5a may be involved in restricting the proliferation of C57mg cells. The authors demonstrated that Wnt4 and Wnt5a expression in mammary cells was responsive to growth regulatory signals and that the down-modulation of gene expression correlated with cell proliferation. However, Lejeune et al.[35] established cell lines from benign or normal tissue for further evaluation of the role of Wnt5a and to assess its regulation. This group demonstrated that the upregulation of Wnt5a in both benign and malignant proliferative diseases of the breast suggested an important role for Wnt genes in breast pathology. Based on those contradictory results, Jönsson et al.[36] detected Wnt5a in cancer tissue from 96 patients with primary invasive breast carcinoma using immunohistochemical techniques. The results showed that loss of Wnt5a increased the risk of early relapse and death due to recurrent ductal breast cancer, and these findings supported the notion that this protein retains tumor suppressor function by virtue of its effects on cell adhesion and motility. Regarding the evaluation of metastasis in ductal breast cancer, the group also observed 83 patients during follow up and found that 78% of patients who presented with loss of expression of Wnt5a had tumor recurrence. The survival rate in the Wnt5a negative group was significantly lower compared with that in the Wnt5a positive group. The authors’ findings strongly suggest that Wnt5a performs an inhibitory function in breast cancer progression and that Wnt5a may be a good marker for the prediction of prognosis in cancer patients.
Thyroid carcinoma
Kremenevskaja et al.[37] tested the effects of a member of the noncanonical Wnt signaling pathway, Wnt5a, in primary thyroid carcinomas and in thyroid carcinoma cell lines. They found that Wnt5a mRNA expression was clearly increased in thyroid carcinomas compared to normal tissue. Immunohistochemically, a bell-shaped response was observed with low to undetectable levels in normal tissue and in anaplastic tumors whereas differentiated thyroid carcinomas showed strong positive immunostaining for Wnt5a. Transfection of Wnt5a into the thyroid tumor cell line FTC-133 was able to reduce proliferation, migration, invasiveness and clonogenicity in these cells. These effects of Wnt5a were associated with membranous β-catenin translocation and c-myc oncogene suppression and were mediated through an increase in intracellular Ca2+ release, which via CaMKII pathways promoted β-catenin phosphorylation. Specific inhibition of β-catenin phosphorylation by W-7, a calmodulin inhibitor, or by KN-93, a CaMKII inhibitor, supports these findings whereas PKC inhibitors are not effective. The authors thus proposed the hypothesis that Wnt5a served as an antagonist to the canonical Wnt signaling pathway with tumor suppressor activity in differentiated thyroid carcinomas.
Others
Wnt5a positivity in non-small cell lung cancers was a significant predictor of shortened survival upon multivariate analysis[38]. Consistent with the concept of Wnt5a as a tumor suppressor, it has been found downregulated in comparison to the respective normal tissue in endometrial cancer and in high-risk neuroblastomas[39,40]. In contrast to human peripheral blood B- and myeloid cells, where Wnt5a transcripts were readily detectable, transcripts were absent or greatly reduced in the respective acute leukemias[25]. Wnt5a was found to be overexpressed in comparison to the respective normal tissues in cancers of the lung, prostate, and nasopharynx[41,42]. Further, overexpression of Wnt5a has been found in endometrial cancer[43].
Functions and effects of Wnt5a on cancer progression
The functional studies on Wnt5a are complex. The Wnt signaling pathway is involved in various events in tumor progression. However, the role of Wnt5a in tumorigenesis remains ambiguous. In several cellular models including those using hematopoietic tissue, breast and uroepithelial cancers, it has been shown to inhibit tumor cell proliferation, yet, there is evidence from several other studies indicating that increased Wnt5a expression is indeed important for cancer progression.
The Wnt5a signaling pathway performs different functions depending on its method of production and action. Wnts are produced not only by malignant cells but also via the presence of receptors, downstream effectors and inhibitors. Wnt proteins also act through interactions between different cell types and through modulation of Wnt5a gradients by matrix components that influence the complex signaling network and determine the actual signaling output (Fig. 1).
Wnt5a signaling directly affects cancer cell motility and invasion. Most of the studies on Wnt5a focus on the protein’s functions that influence the migration of cancer cells. In the human mammary cell line HB2, Wnt 5a inhibited hepatocyte growth factor-stimulated migration and enhanced cell-to-collagen binding via the discoidin domain receptor[44]. Similarly, the migratory capacity of the highly invasive colon cancer cell line SW480 was inhibited by addition of Wnt5a[45]. A synthetic hexapeptide, based on the sequence of Wnt5a, mimicked this effect in a Wnt/Ca2+-dependent manner[46]. Transfection of Wnt5a in thyroid carcinoma cell lines led to increased intracellular Ca2+-release and activation of CamKII[37]. Wnt5a enhanced cell migration in mouse embryonic fibroblasts via Ror2-induced filopodia formation. Knockdown of Wnt5a inhibited the migration of HeLaS3 cells, while its addition stimulated adhesion-dependent migration and invasion of NIH3T3. This was associated with an increased number of lamellipodia and with activation of focal adhesion kinase and accelerated turnover of paxillin in these structures. Wnt5a was identified as a downstream target of the homeobox transcription factor CUTL1 in pancreatic carcinoma cell lines, where it mediated CUTL1-induced enhanced migration and invasion[32]. In melanomas, Wnt5a stimulated motility of the cancer cells via activation of PKC[27].
Tumor microenvironment is associated with the effects of Wnt5a on cancer absolutely. In addition to the availability of receptors and downstream effectors on/in the tumor cells themselves, Wnt5a is influenced by the surrounding stromal compartment, containing benign cells (fibroblasts, lymphocytes, macrophages) and extracellular matrix (ECM). Wnt5a proteins are not only produced by malignant cells, but also by the benign cellular components of the tumor[47]. Wnt5a produced by tumor cells can act in either an autocrine or paracrine modality on adjacent malignant and benign cells. Wnt5a can also be produced by stromal cells, which can be further modulated by chemical links to components of the ECM. Secreted physiological inhibitors, the presence of other agonists on Wnt ligands and promigratory molecules from the stromal compartment, e.g. cytokines, additionally interfere with signaling and functional effects. It is a dynamic signaling network which can either give rise to or modulate the activity of signaling molecules[48]. Much of the secreted Wnt5a is bound to components of the ECM[49]. In addition, Wnt5a has been considered as a potential immune modulatory agent. Wnt5a is upregulated in macrophages in granulomatous lesions due to infection with mycobacterium tuberculosis[50], and it was found to be associated with the secretion of chemokines in the tumor microenvironment. Pukrop et al.[51] reported that Wnt5a was upregulated in macrophages upon co-culture with breast cancer cells and that Wnt5a induced macrophages to produce MMP-7 and TNF-γ. Wnt5a is essential for macrophage-induced invasion, because it regulates tumor cell migration as well as proteolytic activity of macrophages. The importance of the tumor microenvironment, where extracellular matrix, growth factors and inflammatory factors play a key role in tumor invasion, cannot be overlooked. A complex network, which is engaged by the Wnt5a pathway and other signaling pathways, may be involved in the regulation of tumor-microenvironment crosstalk.
Conclusion
In summary, strong evidence indicates that Wnt 5a is involved in cancer progression. Individually, Wnt5a protein expression varies in different cancers, which could provide a new and attractive target for specific anticancer strategies. Many findings have pointed to the promigratory and proinvasive role of Wnt5a, and its expression and function either as a tumor suppressor or promoter obviously depend on the individual intra- and intercellular content. Further studies are needed to investigate the specific role of Wnt5a in different cancers. The actions of Wnt5a described above need to be clarified in more detail, and the development of models to predict the individual signaling outcomes is essential. In the realm of clinical practice, these models would allow us to find new potential diagnostic and therapeutic targets for the precise diagnosis and individualized treatment for aggressive cancer.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received December 2, 2009.
- Accepted January 12, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer