Abstract
OBJECTIVE To investigate the effects of percutaneous cryoablation on uterine fibroids using computed tomographic (CT) guidance after uterine artery embolization.
METHODS Twelve patients who failed to respond to uterine artery embolization were treated using percutaneous cryoablation. All patients had undergone previous uterine artery embolization an average of 1.2 years (0.7~1.6 years) ago. Two cases had abnormal bleeding, and the other 10 suffered from pressure and/or pelvic pain. Myoma diameters were 4 cm to 16.5 cm. By using CT guidance, 2.0, 2.4, 3.0 or 3.8 mm cryoprobes were placed into the fibroid, and two 15~20 min freezing processes were performed. Hemostasis was achieved only by pressing the incisions for several minutes. MR-imaging was performed before the procedure to measure the size and number of fibroid tumors, and follow-up MR-imaging determined the reduction of the lesions.
RESULTS All patients were discharged within 48 h of treatment. Almost no hemorrhage was found in all of the cases. No bladder or bowel injury or significant postsurgery pain was reported. Ice spheres were readily visualized at CT. Beam-hardening artifact from the metal probes was present, but did not interfere with the procedure. Myomas regressed up to 76.3% after 12 months of treatment, and the primary symptoms improved in all treated women.
CONCLUSION CT-monitored percutaneous cryoablation is an effective and minimally invasive therapy for symptom relief and fibroid shrinkage after the failure of uterine artery embolization.
keywords
INTRODUCTION
Uterine artery embolization (UAE) is a procedure which is becoming more common. It is a minimally invasive alternative to hysterectomy or myomectomy for women with symptomatic uterine fibroids. The goal of performing UAE is to produce an ischemic infarction of fibroids while maintaining endometrial and myometrial perfusion. Postprocedural studies have documented a significant decrease in uterine and fibroid volumes with a concomitant decrease in bleeding and bulk-related symptoms[1]. However, failure is sometimes encountered especially for subserosal fibroids [2].
We are now searching for other minimally invasive methods to overcome this failure. Cryoablation has been widely used and accepted because it produces ice sphere of consistent size and shape, causing cell death within the cryolesion[3]. In the late 1980s, Adolf Gallinat was the first to treat fibroids with a new procedure known as myolysis [4]. However, because of the danger of serious bleeding when the probes were removed, the technology was mostly used intra-operatively especially in organs with a rich blood supply like the uterus.
The purpose of this study was to evaluate the safety and feasibility of percutaneous cryoablation with computed tomographic (CT) guidance for selected women who did not have a significant decrease in fibroid volume and symptom relief after UAE. We investigated whether serious bleeding could be avoided by this procedure, and evaluated the effectiveness of the treatment for the relief of symptoms and reduction of the volume of the myomas under CT guidance.
MATERIALS AND METHODS
Patients
A protocol was approved by the Human Investigation Committee of the Cancer Hospital of Tianjin Medical University. An informed consent form was signed by all patients. Patients included were those who had undergone UAE therapies that were unsuccessful. The inclusion criteria were: (a) the volume of the uterus or myoma decreased less than 10% at six months following UAE; (b)they still have fibroid-associated symptoms, like abnormal bleeding, pressure or urinary frequency or pelvic pain. The exclusion criteria were: (a) difficulty in accessing the fibroids percutaneously because of possible danger to the bowel and bladder; (b) desire for future pregnancy. Twelve patients (37.5%) with 23 fibriods were treated between March 2003 and February 2006. Eight of the cases met both inclusion criteria, two met inclusion criteria (a) and the other two met inclusion criteria (b).
Bilateral UAE had been performed by appropriately trained interventional radiologists using standard techniques. After catheterization of the respective uterine arteries, polyvinyl alcohol particles (150~450 μm or 500~700 μm in diameter) and then iodine lipiodol were injected into the circulation to embolize both uterine arteries. Embolization was considered complete when there was virtually no demonstrable flow in the distal uterine arteries.
Cryoablation under CT Guidance
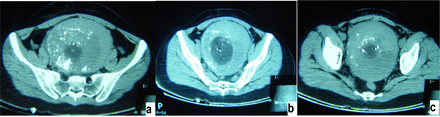
A central venous line was placed prior to the operation and a Foley catheter was inserted into the bladder for continuous drainage. The patients were placed supine and head first into the CT gantry. All procedures were monitored by using a CT scanner (HeSpeed NX/1, GE Medical Systems). The location and size of the fibroids were noted in two dimensions on CT images at the level of the tumor’s largest appearance in the transverse plane. All patients received only local anesthesia. According to the size of the fibroids, (2.0, 2.4, 3.0 or 3.8 mm, outer diameter) cryoprobes were inserted percutaneously into the substance of the targeted fibroid tumor through small (1~2 cm) skin incisions. One to two cryoprobes with different diameters were inserted into the same fibroid and one to threee fibroids were treated at the same time. Limited CT scanning was performed to confirm proper positioning of the cryoprobes. Adjustments of the cryoprobes were made by CT guidance if necessary. As each probe was optimally inserted, the cryosystem was immediately activated. The cryosurgical unit for all procedures was an argon gas-based system (Cryocare, Endocare). We performed two 15~20 min freezing cycles with 1~3 min of thawing under CT monitoring. The procedure required approximately 35~50 min, and the temperature reached -138°C to -175°C at the tip until the frozen myoma appeared as an ice sphere and encompassed the entire myoma. CT scans were performed immediately after an active thaw allowing removal of the cryoprobes less than 2 min after completion of the second freeze (Fig.1). CT scans were also performed 2 and 7 days after the procedure to detect any possible damage of the surrounding organs and observe the short-term effects.
CT scans performed immediately after cryoablation. The markedly hypoattenuating appearance of the ice spheres with smooth, clearly demarcated borders represent the shrinkage of the fibroid. a: top slide of the myoma. b: slide of the myoma with the largest cryoablation diameter. c: the under slide of the myoma.
Follow-up
MR-imaging was conducted 3, 6 and 12 months following treatment to note the differences in fibroid size. Uterine volume estimations were made in each case, by taking uterine measurements in three dimensions. The volume of each treated fibroid tumor was calculated using the following formula: volume = 4/3πr3, where r equals the average radius of the fibroid tumor in three dimensions. The elimination of symptoms were evaluated clinically by verbal description and subjectively described by the patient.
Statistical analysis
The data are presented as the mean±SD. Analyses were performed using the SPSS statistical package (SPSS, Inc, Chicago, Ill). The Wilcoxon rank test was used for continuous data and the Student t test for repetitive measurements of normal and nonnormal distributed data. Statistical significance was set at P<0.05.
RESULTS
In our study, 12 patients underwent cryoablation after uterine artery embolization. The mean age of the women was 44 years(range 38~54 years). Of the 12 patients evaluated, 2 (16.6%) had menorrhagia and 10 (83.3%) had pressure, urinary frequency or pelvic pain.
The average time range from UAE to cryoablation was 1.2 years (0.7~1.6 years) for 4 (33.3%) cases. it was 6 to 12 months and for 8 (66.7%) cases it was more than 1 year.
A total of 23 myomas were treated in the 12 patients: 4 (17.3%) myomas had a diameter of 4~6 cm,5 (21.7%) had a diameter of 7~10 cm, 8 (34.8%) had a diameter of 11~13 cm, and 6 (26.1%) had a diameter of 14~16.5 cm. As for the location of the myomas, 2 (8.7%) were submucosal, 7 (30.4%) were intramural and 14 (60.9%) were subserosal.
All patients were successfully treated with a mean operating time of 42 min (range, 35~55 min), depending on the myoma diameter and number. There was no bleeding during the cryotherapy procedures, no significant postsurgery pain and no indication of bladder or bowel injuries. Among the total patients following the tratment, 7 (58.3%) had yellow vaginal discharges whicn continued for 3 to 6 months. All patients were released in good physical condition within 48 h.
All 12 patients had significant improvement of symptoms, some as early as 1-week after the treatment. Five (41.7%) patients had marked improvement in urinary frequency and/or pressure. The two patients who had menorrhagia had a sharp decrease in menstrual bleeding 1 and 2 months later respectively (Table 1).
Changes in symptoms after cryoablation.
Follow-up MRI showed effects of cryomyolysis and the changes in fibroid tumor volume were calculated. The mean reduction of myoma volume was 32.8% (±7.2%) after 3 months, 51.6% (±8.7%) after 6 months and 76.3% (±12.5%) after 12 months (Table 2,Fig.2).
Reduction of myoma volumes on follow-up examination at 3, 6 and 12 months after cryomyolysis.
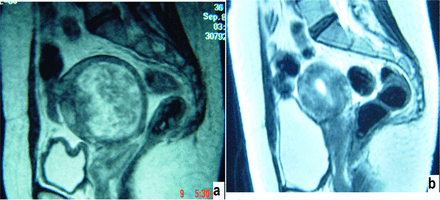
Results of cryoablation. a: Before the treatment, the largest myoma diameter was 9.2 cm. b: After cryoablation, the largest diameter of the myoma shrunk to 1.9 cm.
DISCUSSION
Treatments of uterine leiomyomas are always needed for those with fibroid related symptoms such as menorrhagia, anaemia, pelvic pain, compression, infertility or miscarriage. UAE is an intervention that is especially suited for women who wish to preserve their uterus. This procedure does provide women with a valid alternative treatment pathway for fibroids. Yet, UAE can lead to complications and failure. There have been only few reports in the literature related to treatment of women who failed to respond to UAE, but still wanted to preserve their uterus. Most have had to accept a hysterectomy.
Percutaneous cryoablation has been successfully used to treat the lesions of the liver, kidneys, prostate, thorax and other kinds of cancers[5-7]. However, owing to the concern that large probes can cause bleeding when removed, experience is limited in percutaneous cryoablation of uterine fibroids.
Several studies have shown that laparoscopic cryomyolysis is an effective and safe technique for symptom relief from leiomyomas[8,9]. A clinical study[10] described 61 patients who underwent laparoscopic cryomyolysis resulting in a significant decrease of myoma volume of 60.3% (±20.7), and complete symptom relief in 83.6% of the patients after a 12-month follow-up. But as with other laparoscopic techniques, this kind of procedure requires special equipment and high cost.
In our study, we evaluated the safety and feasibility of percutaneous cryoablation of fibroids after UAE with computed tomographic (CT) guidance. Twelve patients whose uterine fibroid volume failed to decrease and/or with associated symptoms at least 6 months after uterine artery embolization were treated. Cryomyolysis resulted in a significant reduction in myoma size of up to 76.3% with specific symptom relief in all of the patients at 12 months. No bleeding or collateral damage to organs occurred proximal to the uterus.
We conclude that it is safe to conduct percutaneous cryoablation of fibroids after UAE. In our study, after the extraction of the probes, hemostasis was achieved by only pressing the incisions for several minutes rather than by inserting absorbable cellulose fabric as used in other studies. Furthermore, there was no abdominal bleeding, perhaps because after UAE, the blood supply to the uterus had sharply decreased since small embolizing agents may have deposited into the terminal blood vessles of the uterine fibroids[11], For all of the 12 patients, small polyvinyl alcohol particles (150~450 μm or 500~700 μm in diameter) had been used for the UAE.
Our results shows that percutaneous cryoablation after UAE produces a greater myoma volume reduction than that achieved by laparoscopic cryomyolysis[10]. This may suggest that embolization and cryomyolysis have a synergetic effect. As irreversible cell injury occurs only at temperatures below -20°C the effectiveness of the cryoablation, however, appears to be greatly influenced by the degree of blood flow to the fibroid itself and the surrounding uterine parenchyma. Thus, there is a close inverse correlation between the cryoablation lesion size and blood flow. After UAE, the blood flow to the fibroid decreased so the effectiveness of cryoablation increased which resulted in greater fibroid shrinkage.
As shown in this study, beam-hardening artifact from the metal probes was present but did not interfere with the procedure. Although there has been a report concerning cryoablation of fibroids under MRI guidance[5], special MRI equipment(open-configuration MR imaging system) should be used. The ice sphere is well depicted with CT, which adds to the confidence that the lesion has been adequately treated. Another great advantage is that the embolising agent(liquid iodine) in the myoma is easily detected upon CT, which can aid in location of the probes, whereas it can not be seen under MR imaging.
However, in our study we used MR-imaging after the procedure as a main follow-up assessment, because MR-imaging of the uterus enabled a precise quantitative determination of the response to therapy[6]. Furthermore, MR can depict whether there is a reduction in vascularity of a fibroleiomyomata after UAE, and thus assess the vascular supply[7]. Also, MR-imaging can indicate a hemorrhagic infarction and thereby assess the effectiveness of the therapy[12]. Although ultrasound(US) has been used in the guidance of percutaneous cryoablation in some studies[11,13], CT appears to have several advantages. The entire ice sphere, including the deep margin, is well depicted with CT. In contrast, US depicts only the anterior edge of the ice sphere[14,15]. These limitations may contribute to the large number of local recurrences seen with some ablation techniques that are guided solely by percutaneous US rather than by intraoperative US[11].
Our preliminary results suggest that percutaneous cryoablation under CT guidance is a minimally invasive intervention which may offer a realistic alternative to surgery for women with symptomatic uterine fibroid tumors who failed to respond to UAE treatment. This therapy produces no bleeding, little pain, and dramatic results can be achieved with minimal intervention. Longer-term results should be observed to rate complications and limits to this therapy.
- Received March 20, 2007.
- Accepted June 1, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer