Mature T- and natural killer (NK)-cell lymphomas are heterogeneous groups of malignant lymphoid neoplasms arising from T and NK cells. The incidence of mature T- and NK-cell lymphomas is 2.1 per 100,000 people, according to a US report1. Due to the heterogeneity, rarity, and diagnostic complexity of these lymphomas, research focusing on treatment has been considerably hindered. Moreover, real-world data for patients with mature T- and NK-cell lymphomas are insufficient. Therefore, we conducted a retrospective study to determine the subtype distribution, treatment patterns, survival outcomes, and prognostic factors for these lymphomas.
Patients diagnosed with mature T- and NK-cell lymphomas between January 2010 and May 2020 at Tianjin Medical University Cancer Institute & Hospital (TMUCIH) were included in this study. Univariate and multivariate analyses were performed using Cox proportional hazards regression models. The progression-free survival (PFS) and overall survival (OS) rates were estimated using the Kaplan–Meier method. The last follow-up date was August 2022. Furthermore, we conducted comparisons and verifications based on a large database from the Surveillance, Epidemiology, and End Results (SEER) database2. The study flow chart and statistical analysis methods are detailed in the Supplementary materials.
Altogether, we enrolled 443 patients, 319 of whom had available survival data. The major results of the study are below.
Histologic subtype distribution
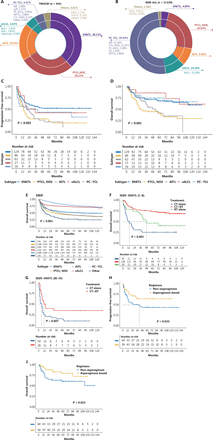
Among the 443 patients, the most common subtype was extranodal NK/T-cell lymphoma [ENKTL; n = 169 (38.15%)], followed by peripheral T-cell lymphoma, not otherwise specified [PTCL, NOS; n = 125 (28.22%)], angioimmunoblastic T-cell lymphoma [AITL; n = 86 (19.41%)], systemic anaplastic large cell lymphoma [sALCL; n = 25 (5.64%)], and primary cutaneous T-cell lymphoma [PC-TCL; n = 17 (4.97%)]. Moreover, enteropathy-associated T-cell lymphoma (EATL) and hepatosplenic T-cell lymphoma (HSTCL) accounted for only a small fraction of all patients (Table S1). Compared to the patients included in the SEER database (n = 17,639), the patients in our study had more ENKTLs and fewer PC-TCLs, especially patients with mycosis fungoides (MF; Figure 1A, 1B).
Histologic subtype distribution of patients from the TMUCIH cohort and SEER database (A, B); PFS (C) and OS (D) according to the pathologic subtype of patients from the TMUCIH cohort; OS (E) according to the pathologic subtype of patients from the SEER database; OS according to treatment in early (F) and late (G) ENKTL patients; PFS (H) and OS (I) of patients with ENKTL receiving chemotherapy regimens with or without asparaginase. LyP, lymphomatoid papulosis; PC-ALCL, primary cutaneous anaplastic large cell lymphoma; SPTCL, subcutaneous panniculitis-like T-cell lymphoma; CTL, NOS, cutaneous T-cell lymphomas, not otherwise specified; CT, chemotherapy; RT, radiotherapy.
Clinical features
Baseline clinical characteristics were available for 417 patients, as shown in Table S2. The median age was 54 years (range 4–83 years) and 36.21% of the patients were over 60 years of age. Among patients, 65.23% (n = 272) were male and 55.70% (n = 220) had stage III/IV lymphoma at the time of initial diagnosis.
A greater male prevalence existed for all 5 lymphoma subtypes (the male-to-female ratio ranged from 1.67–2.04). Patients with AITL (median age, 63 years; age range, 4–81 years), PC-TCL (median age, 58.5 years; age range, 6–67 years), and PTCL, NOS (median age, 58; age range, 5–80 years) were older than patients with ENKTL (median age, 43.5 years; age range, 8–78 years) or sALCL (median age, 46.5 years; age range, 4–79 years). The incidence of stage III–IV disease was lower among the ENKTL patients (23.78%) but higher among the AITL patients (93.90%; Table S2). The positivity rates of Epstein-Barr virus-encoded RNA (EBER) in patients with ENKTL, AITL, and PTCL-NOS were 100%, 66.1%, and 27.9%, respectively (P < 0.001).
Among the patients in our cohort with ENKTL, 89.63% (n = 147) had primary lesions located in upper aerodigestive tract sites. These patients were classified as upper aerodigestive tract NK/T-cell lymphoma (UNKTL), whereas 10.37% (n = 17) were classified as extra-upper aerodigestive tract NK/T-cell lymphoma (EUNKTL). We showed that EUNKTLs had a more aggressive clinical course and a more dismal survival outcome than UNKTLs. A comparison of EUNKTLs and UNKTLs, respectively, revealed the following: advanced stage (94.1% vs. 15.6%; P < 0.001); extranodal involvement > 1 (88.2% vs. 30.6%, P < 0.001); elevated lactic dehydrogenase (LDH; 64.7% vs. 42.5%, P = 0.081); distant lymph node involvement (41.2% vs. 5.4%, P < 0.001); 5-year PFS (22.7% vs. 54.4%, P < 0.001); and 5-year OS (19.6% vs. 71.0%, P = 0.001).
Survival outcome and prognostic factors
A total of 319 patients were included in the survival analysis. With a median follow-up of 56.4 months (range, 1.5–144.1 months), the median PFS and OS were 16.8 months (95% CI: 12.8–20.9 months) and 63.5 months (95% CI: 46.6–80.3 months), respectively, among patients with all subtypes other than ENKTL. The 1-, 3-, and 5-year PFS and OS were 60.4% and 82.7%, 33.1% and 58.7%, and 26.9% and 51.4%, respectively (Figure S3C, S3D). Survival outcomes according to pathologic subtype showed that the prognoses for patients with ENKTL, sALCL, and PC-TCL were better than patients with AITL, and PTCL, NOS (Figure 1C, 1D; Table S3), which were consistent with the findings of previous studies3. Additionally, a survival analysis of 7,768 patients with available survival data from the SEER database revealed similar survival outcomes, except that the ENKTL patients in our study had a longer OS (Figure 1E, Table S3). We concluded from the results of the SEER database that EATL and HSTCL are subtypes with an extremely poor prognosis and 3-year survival rates < 35% (Figure S3F).
We performed univariate and multivariate prognostic analyses of three lymphoma subtypes (NKTCL, PTCL-NOS, and AITL). According to multivariate analysis, only plasma EBV-DNA positivity was shown to be an independent prognostic factor for PFS (HR = 4.48, P = 0.015) and OS (HR = 13.93, P = 0.005) in patients with ENKTLs (Table S4). In PTCL, NOS, the prognostic impact of an advanced stage, elevated LDH, and higher IPI were shown via univariate analysis, but further verification could not be performed due to the limited variables available for multivariate analysis. For AITLs, anemia (HR = 3.08, P = 0.003) and a higher IPI (HR = 4.51, P = 0.001) were significant prognostic factors for PFS (Tables S5, S6).
Treatment
Radiotherapy
ENKTL cells are known to be radiosensitive and radiotherapy is currently one of the most important treatment modalities. We further validated the survival results of 125 patients (stage I–II, 82.4%; stage III–IV, 17.6%) who received different first-line treatment modalities. Only 4 patients in the early stage received radiotherapy alone and were therefore not included in the analysis. Combination of chemoradiotherapy was superior to monotherapy (Figure S7), as was the finding based on SEER data (Figure 1F, 1G). In early-stage patients, combination therapy achieved the highest 5-year OS rate (74.2%), followed by radiotherapy alone (46.5%), whereas chemotherapy alone had the lowest survival rate (25.3%). Moreover, combination therapy involving radiotherapy provided an advantage over chemotherapy alone for advanced-stage patients.
Chemotherapy or new drugs
ENKTL
Due to the high expression of multidrug resistance-related P-glycoprotein on ENKTL cells4, the survival of patients receiving traditional chemotherapy is dismal. Currently, asparaginase-based regimens have become essential for improving treatment efficacy and are associated with better survival rates (Figure 1H, 1I) and a greater ORR compared to non-asparaginase regimens (78.6% vs. 55.4%, P = 0.009; Table 1). The 5-year PFS and OS were 64.3% and 79.7% in the asparaginase-based group and 43.7% and 60.8% in the non-asparaginase group, respectively. SMILE (methotrexate, dexamethasone, ifosfamide, L-asparaginase, and etoposide) and P-GEMOX (pegaspargase, gemcitabine, and oxaliplatin) are common chemotherapy regimens in clinical settings. However, asparaginase-containing therapy failed to provide significant benefits in patients with relapsed/refractory (R/R) ENKTL (PFS2, P = 0.952; OS2, P = 0.979).
Treatment and response status in newly diagnosed ENKTL patients
Patients who received salvage treatment after the first progression or relapse only had a PFS2 and OS2 of 7.5 (95% CI: 2.6–12.4 months) and 22.3 months (95% CI: 1.7–43.0 months), respectively. PD1 antibodies were chosen for 6 of the patients, in whom the ORR was 50.0% [1 CR, 2 PR, 2 stable disease (SD), and 1 progressive disease (PD)].
Nodal PTCL
PTCL, NOS, AITL, and sALCL are nodal peripheral T-cell lymphomas with similar treatment strategies; therefore, the nodal peripheral T-cell lymphoma findings were analyzed together. Several regimens were commonly used as the first-line treatment choice. The ORR was 61.9% for CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone)-like therapy (n = 45), 65.9% for CHOPE (CHOP + etoposide)-like therapy (n = 45), 50.0% for HyperCVAD/MA (hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone/methotrexate and cytarabine; n = 9), 65.6% for chemotherapy with chidamide (n = 32), and 50.0% for other regimens (n = 34; Table S7); there were no significant differences in the ORR or survival rates between the groups. All anthracycline-based chemotherapy regimens, including intensified therapy, resulted in 5-year PFS and OS rates of 23.4% and 47.1% and the median PFS and OS were 15.5 (95% CI: 11.8–19.1 months) and 53.5 months (95% CI: 22.4–84.6 months), respectively. In addition, we determined that the addition of etoposide to CHOP improved the CRR and resulted in a trend toward an improved OS (P = 0.087) in the younger cohort (age < 60 years; Figure S8), but there was no advantage when patients of all ages were included.
Chidamide, a novel histone deacetylase inhibitor (HDACi), is currently approved for use in patients with R/R PTCL and has achieved favorable clinical benefit5. We evaluated the efficacy of chidamide combination therapy in newly diagnosed and R/R PTCL patients in our study. For first-line treatment with chidamide, 81.3% of patients received chidamide in combination with CHOP/CHOPE-like therapy, and 37.5% received additional maintenance therapy with chidamide monotherapy after achieving remission. With a median follow-up of 39.7 months, the median PFS was 18.3 months (95% CI: 0–46.0 months), the median OS was 36.5 months (95% CI: not available), the ORR was 65.6%, and the CRR was 21.9%. Chidamide therapy tended to improve survival, but the difference when compared to treatment without chidamide was not significant (Figure S9). When chidamide was used as salvage treatment, the ORR was 23.5% and the CRR was 11.8% in 18 R/R patients. The median PFS2 was 7.5 months (95% CI: 3.3–11.6 months) and the median OS2 was 19.4 months (95% CI: 12.6–26.2 months). After a period of remission, 77.8% of patients relapsed or progressed. Therefore, the efficacy of chidamide should be further validated in future prospective studies.
Various attempts have been made to treat patients with disease progression or relapse. In addition to second-line chemotherapy and chidamide combination therapy, some patients choose targeted therapy or participation in clinical trials involving agents, such as thalidomide, brentuximab vedotin, pralatrexate, mitoxantrone liposomes, PD1 inhibitors, JAK2 inhibitors, and EZH2 inhibitors. However, the outcomes were still unsatisfactory. The median PFS2 and OS2 were only 7.5 (95% CI: 5.3–9.8 months) and 17.3 months (95% CI: 8.3–26.3 months), respectively.
Hematopoietic stem cell transplantation
A total of 21 patients underwent hematopoietic stem cell transplantation, 20 of whom received autologous transplantation (autoHCT) and 1 of whom received allogeneic transplantation (alloHCT). Sixteen patients received autoHCT in the first-line setting and another 4 patients had a R/R. In autoHCT group, the median PFS and OS were not reached after transplantation with a median follow-up of 46.9 months. The PFS and OS rates were 60.0% and 85.0% 1 year and 55.0% and 65.1% 5 years post-transplantation, respectively. The CRR reached 70.0% and the ORR was 85.0%. As of the last follow-up date, 14 patients were still alive and 11 patients still had a response. Patients who received transplantation as first-line consolidation therapy exhibited improved survival compared with non-transplant patients (Figure S10). The PFS and OS rates for first-line transplant consolidation therapy were 68.8% and 81.3% 5 years post-transplantation, respectively.
Lymphoma is a common type of cancer in China6 and mature T- and NK-cell lymphomas account for 20%–30% of all lymphoid neoplasms7. Epidemiologic studies suggest that the distribution of lymphoma subtypes varies greatly by geographic location. Our study also revealed a higher proportion of ENKTLs among Chinese individuals.
Presently, the outcome of ENKTL has greatly improved with asparaginase-based chemotherapy and the combination of radiotherapy and chemotherapy. Radiotherapy reduces the probability of relapse or progression and improves the patient response to chemotherapy, and it remains irreplaceable in the era of non-anthracycline chemotherapy, especially for limited-stage patients8. For nodal PTCL, there are no standard treatment regimens available. Anthracycline-based chemotherapy can produce a reasonable initial ORR, but patients often have a short duration of response and a high rate of recurrence. Efforts are currently underway to explore effective treatments aimed at enhancing survival outcomes9. However, our study of chidamide did not reveal significant superiority over conventional chemotherapy even though a trend was observed. First, the results may have been affected by the small sample size or the short duration of follow-up. Second, we did not perform subgroup analyses for individual pathologic subtypes. In the future, HDAC inhibitors may be used more frequently for patients with the follicular helper T cell subtype, which exhibits a high frequency of epigenetic dysregulation10.
Overall, we have provided an overview of mature T- and NK-cell lymphomas, further demonstrating disease characteristics and treatment patterns. In the future, the focus of research should be on reducing relapses, improving long-term survival, and providing more personalized and precise treatment.
Supporting Information
Conflict of interest statement
No potential conflicts of interest are disclosed.
Author contributions
Conceived and designed the analysis: Huilai Zhang, Xianhuo Wang, Shiyong Zhou, Zheng Song.
Collected the data: Yue Fei, Shen Meng, Jingwei Yu, Xia Liu, Xue Han, Lanfang Li, Lihua Qiu, Zhengzi Qian, Shiyong Zhou.
Contributed data or analysis tools: Bin Meng, Wenchen Gong, Huilai Zhang, Xianhuo Wang, Shiyong Zhou, Xiubao Ren.
Performed the analysis: Junlei Jia, Xiaohui Wang, Zheng Song, Shen Meng.
Wrote the paper: Junlei Jia.
Data availability statement
The data generated in this study are available upon request from the corresponding author.
Footnotes
↵*These authors contributed equally to this work.
- Received December 22, 2023.
- Accepted February 8, 2024.
- Copyright: © 2024, Cancer Biology & Medicine
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY) 4.0, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.