Abstract
Objective: Cancer cell radioresistance is a stumbling block in radiation therapy. The activity in the nuclear factor kappa B (NFκB) pathway correlates with anti-apoptotic mechanisms and increased radioresistance. The IKK complex plays a major role in NFκB activation upon numerous signals. In this study, we examined the interaction between ionizing radiation (IR) and different members of the IKK-NFκB pathway, as well as upstream activators, RAF1, ERK, and AKT1.
Methods: The effect of 4 Gy of IR on the expression of the RAF1-ERK-IKK-NFκB pathway was examined in A549 and H1299 lung cancer cell lines using Western blot analysis and confocal microscopy. We examined changes in radiation sensitivity using gene silencing or pharmacological inhibitors of ERK and IKKβ.
Results: IKKα, IKKγ, and IκBα increased upon exposure to IR, thereby affecting nuclear levels of NFκB (phospho-p65). ERK inhibition or siRNA-mediated down-regulation of RAF1 suppressed the post-irradiation survival of the examined lung cancer cell lines. A similar effect was detected on survival upon silencing IKKα/IKKγ or inhibiting IKKβ.
Conclusions: Exposure of lung cancer cells to IR results in NFκB activation via IKK. The genetic or pharmacological blockage of the RAF1-ERK-IKK-NFκB pathway sensitizes cells to therapeutic doses of radiation. Therefore, the IKK pathway is a promising target for therapeutic intervention in combination with radiotherapy.
keywords
Introduction
Nuclear factor kappa B (NFκB) transcription factors are dimers that comprise RELA (p65), RELB, c-REL, NF-κB1 (p105/p50), and NF-κB2 (p100/p52), which are considered fundamental regulators in a plethora of cellular processes and physiological functions, such as inflammation and cell growth, differentiation, migration, and development1-3. NFκB dimers have a well-conserved role in immune and inflammatory responses2. Moreover, the activation of NFκB transcription factors has been dysregulated in numerous autoimmune diseases, chronic inflammations, and cancers due to the significance of these NFκB dimers in regulating the gene expression of downstream targets1,4-6.
Extracellular stimuli, such as cytokines, viruses, and other stressful signals, stimulate receptors and initiate a signaling cascade2, which converges to activate the main NFκB pathway upon inflammation, immunity, radiation, and other stresses. In unstimulated cells, NFκB dimers are bound to IκBα, which maintains NFκB in the cytoplasm, thus preventing its translocation to the nucleus and its transcriptional activity.
The activation of the pathway leading to NFκB nuclear translocation starts with the phosphorylation of the IKK kinases by proinflammatory stimuli, such as in TNFα receptor activation. The IKKα and IKKβ kinases are the catalytic subunits of the IKK complex, along with IKKγ (NEMO), which is a non-catalytic, regulatory, and scaffold subunit7-10. Activated IKKα and IKKβ kinases phosphorylate the IκBα kinase at two serine residues, thereby ubiquitinating and degrading IκBα in a proteasome-dependent manner. The IκBα-NFκB heterodimer is therefore disbanded, and NFκB proteins enter the cell nucleus and regulate the transcription of a vast range of genes2,11.
The IKKs may be activated through multiple pathways apart from TNFα via the upstream IKK kinases (IKKKs), such as TAK1, mitogen-activated protein/ERK kinase kinase 3, and trans-autophosphorylation1,12,13. Apart from phosphorylating numerous transcription factors, such as Ets-1, c-Jun, c-Myc, and CREB114, the Ras/Raf/MEK/ERK pathway is also involved in IKK regulation. Moreover, the activated Akt pathway also converges to IKK phosphorylation15, thereby suggesting that IKK is a critical point in the regulation of NFκB activity by multiple crucial molecular pathways.
A constitutive expression of NFκB correlates with increased radioresistance and reduced apoptosis of cancer cells, which is a stumbling block in radiation therapy16-20. We studied the effect of ionizing radiation (IR) on the IKK-NFκB pathway in two lung cancer cell lines: A549, an adenocarcinoma human alveolar basal epithelial cell line, and H1299, a human non-small cell lung carcinoma cell line. As the IKK pathway is activated upon multiple signals, thereby activating NFκB, we examined the effect of inhibiting the Raf, ERK, and IKK complex upon cellular radiosensitivity using either siRNA or pharmacological small-molecule inhibitors.
Materials and methods
Cell lines, culture conditions, and irradiation
A549 and H1299 non-small cell lung cancer cells21,22 and normal lung epithelial cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% FBS, 2 mM of L-glutamine, 100 U/mL of penicillin, and 100 μg/mL of streptomycin (Invitrogen) at 37 °C with 5% CO2 in a humidified incubator. Cell number was determined in duplicates using a haemocytometer. Irradiation was performed using a cobalt-60 gamma-ray irradiator (Theratron).
The A549 and H1299 cell lines were purchased from ATCC (ATCC® CCL-185TM and ATCC® CRL-5803TM, respectively), and all experiments were performed within 12 months. Details on the cell lines can be found at https://www.atcc.org/Products/All/CCL-185.aspx?slp=1#generalinformation and http://www.atcc.org/Products/All/CRL-5803.aspx#characteristics. The cell lines were authenticated by ATCC, and details and methods of characterization are available at http://www.atcc.org/Products/Cells_and_Microorganisms/Testing_and_Characterization/STR_Profiling_Analysis.aspx.
Both cell lines constitutively activated the NFκB pathway, as shown by the higher levels of pp65 expression in the nuclei of cells under standard conditions compared with normal human lung protein extracts (Figure S1).
Proliferation assays
The viability of the A549 and H1299 cell lines was evaluated using the Alamar Blue® cell viability reagent (DAL1100, Invitrogen) and then measured at 540 nm excitation and 590 nm emission wavelength in a FLUOstar® Omega microplate reader (BMG Labtech). This assay quantifies the number of cells with metabolically active mitochondria based on the reduction of resazurin by mitochondrial enzymes, as previously evaluated in several cell lines in post-irradiation survival experiments23. An example of the concordance between this method and the traditional colony formation assay has been published previously24. All viability experiments were performed six times, and the mean values are plotted in the figures.
RNA interference
Predesigned pools of four siRNA oligonucleotides (Shanghai GenePharma Co., Ltd.) were used for silencing the expression of RAF1 (RAF1-homo-943 5′-GCACCAAAGU ACCUACUAUTT-3′, RAF1-homo-1913 5′-GCAGGUUGAA CAACCUACUTT-3′, RAF1-homo-643 5′-GCUUGCAUGA CUGCCUUAUTT-3′, RAF1-homo-491 5′-CUGCAUCUCU CCUACAAUATT-3′), IKKα (CHUK-homo-1077 5′-GAGC GUGAAACUGGAAUAATT-3′, CHUK-homo-739 5′-CCU GGCAUGAGAAGAUUAATT-3′, CHUK-homo-1891 5′-G CUGUAAGCAGAAGAUUAUTT-3′, CHUK-homo-677 5′-GACCAUGGUAUUUGAAUGUTT-3′) and IKKγ (IKBKG-homo-1638 5′-CCCUGCAGAUACAUGUCAUTT-3′, IKBKG-homo-437 5′-GCCAACUGUGUGAGAUGGUTT-3′, IKBKG-homo895 5′-GAGGCUGCCACUAAGGAAUTT-3′, IKBKG-homo-1109 5′- UGGCCUAUCACCAGCUCUUTT-3′).
The A549 and H1299 cells were transfected at 50%–60% confluence with 50, 40, and 35 nM siRNA against RAF1, IKKα, and IKKγ, respectively, or an equal molar of mismatched scrambled siRNA (siscr). The growth medium containing the transfection complexes was replaced with a fresh complete medium 6 h after transfection, and the cells were incubated for 48 h in total. Cell viability was measured using Alamar Blue® assay. Western blot analysis was performed for each experiment to ensure functional and specific silencing. Transfections with siRNAs were performed using Metafectene Pro (Biontex Laboratories GmbH) in serum-free DMEM following the manufacturer’s instructions.
Inhibitors
ERK (FR180204) and IKKβ (IMD-0354) inhibitors were obtained from SelleckChem. Twenty-four hours after plating 250 A549 or H1299 cells, 75 uM of ERK or 20 uM of IKKβ inhibitors was added to the cell medium for another 24 h. The inhibitors were then removed, and the cells were treated with single irradiation doses of 0, 2, 4, 6, or 8 Gy IR and incubated in fresh, inhibitor-free medium for another 7 days, when cell proliferation was evaluated with Alamar Blue®.
Western blot analysis
Nuclear and cytoplasmic or whole-cell lysates from both cell lines were used for immunoblotting. Total cell lysates were centrifuged at 1000 g for 10 min for nuclear–cytoplasmic fractionation. The resulting supernatant was the cytoplasmic fraction, and the remaining pellet was resuspended in a lysis buffer, which contained the nuclear fraction. The lysates were prepared after the incubation of the cells in an ice-cold lysis buffer (0.25 M sucrose, 10 mM tris pH 7.4) with a complete mini protease inhibitor cocktail (Roche Diagnostics, GmbH) and phosphatase inhibitor cocktail (Cell Signaling Technology, Inc.) for 30 min, which was followed by manual scraping and homogenization using a blue pellet pestle. Protein concentrations were determined using a BCA protein assay kit (Pierce) with bovine serum albumin as standard. Samples (20–30 μg) were analyzed by 10%–12% SDS–PAGE according to standard procedures and transferred onto PVDF membranes. Membranes were probed with the appropriate primary antibodies: rabbit anti-RAF1 (ab137435, 1:1000), mouse anti-IKKα [3G12H9] (ab105923, 1:1000), rabbit anti-IKKγ (ab137363, 1:1500), and rabbit anti-HSP90 (ab13495, 1:1000), which were obtained from Abcam; rabbit anti-AKT1 S1 (phospho-Thr246) (A0453, 1:1000), rabbit IKK-alpha (phospho-serine176)/IKK-beta (phospho-serine177) (p12-1120, 1:1000), and rabbit NFκB p65 (phospho-Thr254 (A7169, 1:1000), which were obtained from Assay Biotech; and anti-nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha (NFκBIA) (Ser32), (Ser36) (ABIN197305, 1:1000) antibody from Antibodies Online. Anti-mouse or anti-rabbit secondary antibodies conjugated with horseradish peroxidase (1:3000; PO447, DAKO, and 1:3000, 170-6515, Biorad) were used, and enhanced chemiluminescence was detected using ECL Western blot substrates (Thermo Fisher Scientific, Inc., IL, USA). Equal protein loading was verified by reprobing each membrane with an antibody against beta-actin (1:5000, Abcam; cat. no. ab75186) or against lamin B1 for nuclear fractions (ab133741, 1:3000).
The blot images were captured using a Chemidoc® MP imaging system (Biorad), and band densitometry was analyzed with the accompanying Image Lab software.
Immunofluorescence
For immunofluorescence staining, both cell lines were grown on No 1.5 glass coverslips, fixed in 3.7% formaldehyde/PBS pH 7.4 for 20 min at 37 °C, and then permeabilized in PBS/0.1% v/v Triton X-100 pH 7.4 for 5 min at room temperature. Cells were blocked in PBS/5% w/v BSA pH 7.4 and stained for 1 h at room temperature with various combinations of rabbit anti-RAF1 (ab137435, 1:100), mouse anti-IKKα [3G12H9] (ab105923, 1:100), rabbit anti-IKKγ (ab137363, 1:100), and rabbit anti-Hsp90 (ab13495, 1:100), which were obtained from Abcam; rabbit anti-Akt1 S1 (Phospho-Thr246) (A0453, 1:100), rabbit IKK-alpha (phospho-serine176)/IKK-beta (phospho-serine177) (p12-1120, 1:100), and rabbit NFκB B p65 (phospho-Thr254) (A7169, 1:100), which were obtained from Assay Biotech; and NFκBIA (Ser32), (Ser36) (ABIN197305, 1:100) antibody, which was from Antibodies Online. Cells were washed in PBS pH 7.4, incubated with the appropriate CF 488 and 564 secondary antibodies (1:500, Biotium) at room temperature, and DNA was counterstained with Hoechst 33342 (1 μg/mL; Sigma-Aldrich). After the final washes, the coverslips were mounted in a Mowiol 4-88 based mounting medium. Imaging was performed on a customized Andor Revolution Spinning Disk confocal system built around a stand (IX81; Olympus) with a 60 and 100× lens and a digital camera (Andor Ixon+885) (CIBIT Facility, MBG-DUTH). Image acquisition was performed using Andor IQ 2 software. Optical sections were recorded every 0.3 μm. All confocal microscopy images presented in this work are two-dimensional maximum intensity projections of z-stack images (ImageJ 1.47v National Institute of Health, USA).
Statistical analysis
Statistical analysis and graphical presentation were performed using the statistical package, GraphPad Prism Version 5.01a (GraphPad Software, Inc., USA). Paired and unpaired two-tailed t-tests were used for comparisons as appropriate. A P-value of <0.05 was used for determining statistical significance.
Results
IR affects the IKK-NFκB pathway protein expression and upstream activators
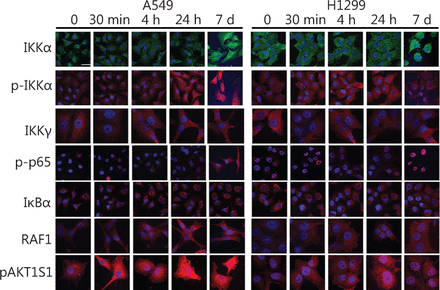
First, we investigated whether and how IR affects family members of the IKK-NFκB complex. The A549 and H1299 lung cancer cell lines were irradiated with 4 Gy IR and analyzed using immunoblotting and confocal microscopy. The protein levels of IKKα and its phosphorylated form pIKKα started increasing within the first 4 h after-4 Gy irradiation of the A549 and H1299 lung cancer cells (IKKα: 1.73 fold/P<0.05 in A549 and 2.45 fold/P<0.05 in H1299; pIKKα: 2.65 fold/P<0.05 for pIKKα in A549 and 1.35 fold/P<0.05 in H1299), and IKKα expression remained elevated for 7 days. However, pIKKα levels returned to normal on day 7 in the H1299 cell line (Figures 1 and 2). Although the IKKγ protein declined 30 min after irradiation in both cell lines, its levels returned to normal or even above normal at 4 h after irradiation (1.59 fold/P<0.005 in A549 and 1.24 fold/P<0.005 in H1299) and were stable for the following 7 days. The IκBα protein levels did not change in the H1299 cell line but increased in the A549 cell line (1.17 fold/P=0.17 in H1299 and 1.76 fold/P<0.005 in A549). The phosphorylated form of the NFκB p65 protein component (pp65) noticeably increased in the nuclear fraction 30 min after irradiation in both cell lines (1.25 fold/P<0.05 in A549 and 1.45 fold/P<0.05 in H1299), but its levels returned to normal or below normal several hours later. However, elevated pp65 levels were detected again after 7 days (Figures 1 and 2). Unlike nuclear levels, cytoplasmic levels of NFκB seemed to decrease at 30 min after-IR but were replenished at later time points and showed a second peak at 7 days after-IR (Figures 1 and 2). Moreover, the pp65 nuclear levels seemed to be more constitutively expressed in tumor cells than those in normal human lung tissue extracts (Figure S1).
IR affects protein levels of the IKK members, upstream activators, and phosphorylation of the downstream p65 NFκB in A549 and H1299 lung cancer cell lines. A549 and H1299 cells were treated with 4 Gy IR, and cytoplasmic (IKKα, pIKKα, IKKγ, IκBα, pp65 NFκB, RAF1, and pAKT1S1) or nuclear fractions (pp65 NFκB) of cell lysates were collected 30 min, 4 and 24 h, and 7 days post-irradiation and analyzed with Western blot. Non-irradiated samples were used as an untreated control (denoted as 0), and β-actin and lamin B1 were used as loading controls for cytoplasmic and nuclear fractions, respectively.
IR affects protein levels of IKK members, upstream activators, and phosphorylation of the downstream p65 NFκB in A549 and H1299 lung cancer cell lines. Cells were collected for the confocal microscopy immunofluorescence analysis at the same time points as for the Western blot analysis (Figure 1) and fixed and stained with the relevant antibodies. Representative images demonstrate antibody staining at the indicated time points. Hoechst 33342 was used as a nuclear marker. Scale bar denotes 10 μM.
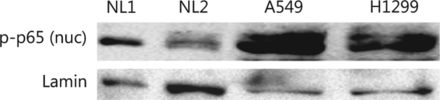
Constitutive over-expression of nuclear NFκB pp65 in NSCLC cell lines. Nuclear fractions from cells of two normal human lung tissues (denoted as NL1 and NL2) and two NSCLC cell lines, A549 and H1299, were compared regarding their endogenous expression of NFκB pp65. Lamin B1 was used as a loading control.
Investigating the possible pathways involved in signaling cascades that may converge to the activation of the NFκB pathway following IR, we also examined levels of RAF1 and of an Akt1-mediated phosphorylated protein expression (Akt1 substrate 1 AKT1S1 or proline-rich Akt substrate25) in cytoplasmic cellular fractions. RAF1 protein levels increased at 4 h after-irradiation in both A549 and H1299 cell lines (3.05 fold/P<0.01 in A549 and 1.9 fold/P<0.001 in H1299). Furthermore, pAkt1S1 protein levels increased sharply as early as 30 min after irradiation (2.04 fold/P<0.001 in A549 and 1.42 fold/P<0.001 in H1299). The levels of the A549 cell line remained elevated up to 7 days later, whereas those of H1299 decreased at the last time point examined (Figures 1 & 2).
Knockdown of the IKK pathway increases radiosensitivity
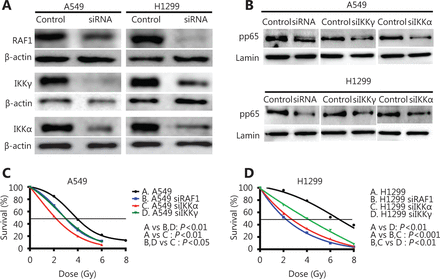
We performed silencing of the RAF1, IKKγ, and IKKα genes using specific siRNAs to examine the role of the IKK pathway in lung cancer radiosensitivity (Figure 3A). All three genes were efficiently repressed in both cell lines (0.68 and 0.12 for siRAF1 in A549 and H1299, respectively; 0.26 and 0.45 for siIKKγ in A549 and H1299, respectively; 0.25 and 0.28 for siIKKα in A549 and H1299, respectively; P<0.05). In addition, we examined the direct connection between IKK members and NFκB by testing the functional impact of the siRNA-driven down-regulation of RAF1, IKKα, and IKKγ on NFκB/phospho-p65 nuclear levels in both cell lines. Nuclear fractions of cell lysates were isolated at 48 h after-siRNA treatment, and the NFκB/pp65 protein levels were examined in each case. All three siRNAs resulted in a marked reduction of the pp65 protein expression (0.41 and 0.24 after siRAF1 in A549 and H1299, respectively, P<0.05; 0.39 and 0.51 after siIKKα in A549 and H1299, respectively, P<0.05; 0.45 and 0.39 after siIKKγ, respectively, P<0.05) in the cell nuclei of the A549 and H1299 cell lines, thereby suggesting the siRNA-induced inhibition of NFκB nuclear translocation (Figure 3B) in both cancer cell lines.
Down-regulation of IKK family members and upstream activator Raf1 increases radiosensitivity of A549 and H1299 lung cancer cell lines. (A) Immunoblot analysis demonstrates the effective down-regulation of RAF1, IKKα, and IKKγ in A549 and H1299 cell lines after a 48 h incubation of cells with the designated pools of siRNAs. si-scrambled treated cells were used as a control, and β-actin was used as a loading control. (B) Immunoblot analysis shows NFκB pp65 nuclear protein levels after a 48 h incubation of cells with the designated siRNA pools against RAF1, IKKα, and IKKγ. si-scrambled treated cells were used as control, and lamin B1 was used as a loading control. C-D) A549 and H1299 cells were transfected with siRNA pools against RAF1, IKKα, and IKKγ, and dose-response viability curves are plotted after treatment with 2, 4, 6 or 8 Gy IR.
Cell viability was assessed 7 days after irradiation with 0, 2, 4, 6, or 8 Gy and after gene suppression was confirmed after silencing the relevant gene. Both lung cancer cell lines showed an important radiosensitization effect after the irradiation of the control and the three cases of gene silencing. While the radiation dose leading to 50% cell survival (lethal dose, LD50) at 7 days after irradiation was calculated, the LD50 in the A549 cells was reduced from 3.95 to 3.03, 3.02, and 2.17 Gy upon RAF1, IKKγ, and IKKα silencing, respectively (23.29%, 23.54%, and 45.06% dose reduction with P<0.01, P<0.001, and P<0.01, respectively). Meanwhile, the LD50 in the H1299 cells was reduced from 6.58 to 2.12, 3.92, and 2.65 Gy upon RAF1, IKKγ, and IKKα silencing, respectively (67.78%, 40.42%, 59.73% dose reduction with P<0.0001, P<0.0001, and P<0.001, respectively) (Figure 3C).
Pharmacological inhibition of the IKK pathway increases radiosensitivity
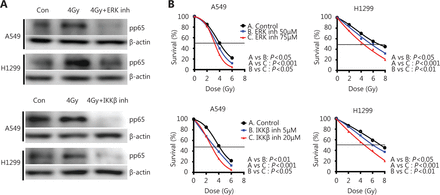
The cells were irradiated in the absence or presence of two drugs: FR180204, a selective ERK inhibitor blocking the RAF1 pathway and IMD-0354, an IKKβ inhibitor blocking IκBα phosphorylation, to examine the direct connection between IKK members and NFκB, as well as the efficacy of the pharmacological inhibitors of the IKK pathway on NFκB/p65 nuclear levels. We chose to pharmacologically inhibit different targets than those used for the siRNA inhibition to assess several aspects of the same pathway and obtain a global view of the mechanism. Both lung cancer cell lines were pre-incubated for at 24 h with either inhibitor and then underwent 4 Gy irradiation, and the NFκB/pp65 protein levels in the nuclear fraction were examined 30 min after irradiation. Both drugs resulted in a marked reduction of pp65 protein accumulation (0.22 and 0.42 after ERK inhibition in A549 and H1299, respectively, P<0.05; 0.13 and 0.21 after IKKβ inhibition in A549 and H1299, respectively, P<0.05) in the cell nuclei, thereby suggesting the inhibition of NFκB radiation-induced translocation (Figure 4A) in both cancer cell lines.
Pharmacological inhibition of IKKβ and upstream activator ERK decreases pp65 protein accumulation in the cell nuclei and increases radiosensitivity of A549 and H1299 lung cancer cell lines. (A) Immunoblot analysis demonstrates the protein levels of pp65 NFκB at 30 min after treating A549 and H1299 cells with 4 Gy IR in the presence or absence of 75 μM of the ERK inhibitor (upper panel) or 20 μM of the IKKβ inhibitor (lower panel). Lamin B1 was used as a loading control, and con denotes the untreated control. (B) A549 and H1299 cells were pre-treated for 24 h with 75 μM of ERK or 20 μM of IKKβ inhibitor and irradiated with 2, 4, 6 or 8 Gy IR. Cell viability was monitored 7 days later. Pairwise comparisons were performed for all cases, and their P values are shown.
The 75 and 20 μM ERK and IKKβ inhibitors, respectively, in the viability experiments resulted in 80%–90% cell viability after 24 h of incubation (data not shown), and these concentrations were applied in the radiosensitization experiments. Cells were pre-treated with either inhibitor for 24 h and irradiated with various doses, and their growth was observed at 7 days after-irradiation. Both drugs sensitized lung cancer cells to radiation in a dose-dependent manner (Figure 4B). In the A549 cell line, 5 and 20 μM of the IKKβ inhibitor resulted in a significant reduction of the LD50 from 4.06 to 3.38 and 2.74 Gy, respectively (with P<0.05 and P<0.001, respectively). In the H1299 cell line, the LD50 was reduced from 7.10 to 5.5 and 4.6 Gy with 50 and 75 μM of the IKKβ inhibitor, respectively (with P<0.05 and P<0.001, respectively). Treating the A549 cells with 50 and 75 nM of the ERK inhibitor also reduced LD50 from 4.10 to 3.6 and 3.2 Gy, respectively (with P<0.01 and P<0.001, respectively). In the H1299 cell line, the LD50 was reduced from 6.72 to 5.52 and 4.4 Gy by 50 and 75 nM of the ERK inhibitor, respectively (with P<0.05 and P<0.001, respectively).
Discussion
We investigated the roles of the IKK and the Ras/ Raf/MEK/ERK pathways in the radiosensitivity of lung cancer cells. First, we noted that the protein level kinetics of members of the IKK complex, IKKα, and IKKγ, as well as IκBα, were altered upon irradiation, thereby affecting NFκB nuclear translocation in a time-dependent manner. The phosphorylation status of IKKα increased within hours after irradiation. However, the duration of this radiation-induced effect depends upon the cell line. A protracted presence of high levels of phospho-IKKα was evident in the A549 cell line for up to 7 days. Meanwhile, levels of phospho-IKKα normalized relatively rapidly in H1299.
This noteworthy difference between the two lung cancer cell lines did not translate to the phosphorylated p65 levels. Nuclear levels dropped within hours in both cell lines after an initial increase. Several studies have suggested that irradiation results in the activation of the NFκB pathway in some cells, but the kinetics of its activation have never been thoroughly examined26,27. Following an initial increase, NFκB nuclear translocation levels decreased 24 h after irradiation and increased again on day 7. The decrease in the nuclear pp65 levels at 4 and at 24 h after-IR was accompanied by a simultaneous increase of cytoplasmic pp65. We believe that the elevated pp65 nuclear levels at 30 min after-irradiation could be a result of a rapid and increased translocation from the cytoplasm to the nucleus due to the increased demand for NFκB-dependent activation, which does not occur at later time points, such as 4 and 24 h after-IR. Moreover, the increased levels of both fractions on day 7 could be a result of secondary radiation effects. This finding reflects a cellular attempt to shift the balance toward survival and regrowth of pathways. Indeed, cell lines experience the nadir of cell depletion and begin to regrow at 6 days to 7 days after irradiation, an effect we have confirmed in multiple cell lines (data not shown23). In addition, we and other researchers have shown that NFκB is more constitutively expressed in NSCLC cell lines than in cells from normal human lung tissues28,29. The over-expression is demonstrated in the nuclear fractions and can therefore be attributed to the higher demand for transcriptional activity in tumor cells than those in normal tissues.
The role of the IKK pathway in radiosensitization is recently gaining attention. The inhibition of an IKK family member (IKKβ) may sensitize tumor cells via both an apoptosis-dependent and apoptosis-independent manner, thus involving or bypassing NFκB30. Radiosensitization occurs after inhibition of IκBα phosphorylation in human brain tumor M054 cells or by using a super-repressor form of IκBα in HT1080 colorectal cancer, fibrosarcoma, glioma, and cervical cancer cells31-35. Moreover, inhibiting IKKβ (but not IKKα) either with a particular IKKβ inhibitor or by shRNA-silencing of IKKβ results in IR-dependent DSB repair inhibition and IR-induced cell death in various cancer cell lines. This effect is believed to occur in an IκBα-NFκB- and apoptosis-independent pathway to a certain extent30,36.
This study confirmed the leading role of the IKK complex in radiosensitivity by increasing the sensitizing effect of IR by the down-regulation of either IKKα or IKKγ. Although IKKγ has no enzymatic activity, its presence as a scaffold seems to be important in the phosphorylation activity of the IKK complex37-40. Similar results were observed in this study after inhibiting IKKβ using IMD-0354, a small-molecule inhibitor. The exact death pathway exploited under the radiosensitizing effect of the IKK inhibition was unclear. Whether this death pathway involves increased apoptosis via activated NFκB or a different cell death pathway demands further investigation.
The Ras/Raf/MEK/ERK and AKT pathways are directly involved in the activation of the IKK complex and release of active NFκB14-16,41. A possible mechanism of RAF1 activation by IR could be due to the strong induction of free radicals (reactive oxygen species or ROS) by radiation in the cytoplasm of cells. ROS directly activates the RAF1 protein and the downstream MEK/ERK pathway42, thus possibly accounting for the phosphorylation of IKKα and the rapid accumulation of pp65 in the cell nuclei. RAF1 and AKT downstream protein AKT1S1 levels significantly increased at minutes after irradiation, thereby suggesting that IR may enhance the phosphorylation activity of the IKK complex by activating both RAS and AKT pathways. Meanwhile, repressing the RAF1 expression using specific siRNAs enhanced A549 and H1299 cancer cell line radiosensitivity. The involvement of the Ras/Raf/MEK/ERK pathway in the IKK-dependent radiosensitization was additionally borne out by the fact that inhibiting ERK with a small-molecule inhibitor also increased the radiosensitization of both lung cancer cell lines at the various IR doses applied. The link between ERK/IKK and radiosensitization was also supported by experiments showing that inhibiting ERK with small-molecule inhibitor strongly reduces the levels of the phosphorylated form of NFκB and pp65 along with their nuclear localization.
Several recent studies have demonstrated that the IKK complex plays a central role in NFκB phosphorylation, translocation, and activation. Moreover, IKK itself is activated upon a vast range of stress signals, such as inflammatory cytokines, radiation, pathogens, and DNA damage. Thus, activation involves a variety of upstream factors and complexes, including AKT and the Ras/Raf/MEK/ERK pathway. IKK activation culminates in NFκB activation independent of the initial trigger. Blocking IKK activation can be more effective than intervening in each of the upstream pathways that comprise the IKK. Dysregulating the IKK-NFκB pathway results in reduced NFκB activity and enhanced radiosensitivity. Therefore, blocking the IKK-NFκB pathway can be useful for therapeutic intervention in radiation oncology.
Footnotes
Conflict of interest statement No potential conflicts of interest are disclosed.
- Received April 26, 2017.
- Accepted July 10, 2017.
- Copyright: © 2017, Cancer Biology & Medicine
This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY) 4.0, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.