Abstract
OBJECTIVE To study the expression level and clinical significance of HMGB1 and RAGE in cervical squamous epithelial carcinoma.
METHODS Real time quantitative polymerase chain reaction (qRT-PCR) was employed to examine the expression of HMGB1 (high mobility group box protein1), and RAGE (receptor for advanced glycation endproducts) in 60 cervical squamous epithelial carcinomas (CSEC), their paraneoplastic tissues (PS) and 30 normal cervix tissues (NCS).
RESULTS The expression of HMGB1 in the CSEC samples and PS was similar (P>0.05), but higher compared to NCS (P<0.05). Overexpression of HMGB1 in the CESC tissues was significantly correlated with the tumor (P<0.05), and the presence of metastasis (P<0.01), but not correlated with the tumor diameter or tumor grade. RAGE expression was not significantly different among these tissue types, and showed no significant correlation with the the tumor stage, diameter or grade. But there was a significant positive correlation between RAGE expression and CSEC metastasis.
CONCLUSION The results suggest that HMGB1 may be related to the proliferation, progression and metastasis of CSEC. The relationship of HMGB1/RAGE may be of importance for CSEC metastasis. HMGB1 presents a new potential gene target for prevention and treatment of CSEC. Study of HMGB1/RAGE expression will offer an experimental foundation for understanding the pathogenesis of CSES.
- HMGB1
- RAGE
- cervical squamous epithelial carcinoma
- qRT-PCR
INTRODUCTION
In China, cervical squamous epithelial carcinoma (CSEC) is one of the most common cancers, causing a serious health problem among women. But the related etiological factors and pathogenesis are not apparent. Previous studies have shown that HMGB1 is expressed in many tumors, and it has been demonstrated that the overexpression of HMGB1 plays a key role in different neoplastic biological processes[1,2]. Expression of the receptor for advanced glycation endproducts, a main receptor of HMGB1, has also been associated with cancers[3]. Therefore we decided to evaluate expression of HMGB1 and RAGE in CSEC by qRT-PCR, and to investigate the relationship of their expression with cancer progression, invasion, metastasis, and especially examine the role of HMGB1/RAGE in metastasis.
MATERIAIS AND METHODS
Samples
Sixty CSEC samples, 60 paraneoplastic samples (PS), and 30 normal cervical samples (NCS) were from the Department of Gynecological Oncology, Tianjin Cancer Hospital. Ages of the patients ranged from 35 to 77 (median 50) years. All samples were identified by pathologic examination. The PS were removed at least 0.5 cm away from the tumor margin, and the NCS were removed form cervical leiomyomas of the uterus. Upon collection, the samples were frozen immediately at -80°C for future analysis.
RNA extraction and cDNA systhesis
Total RNA was extracted from 5 mm diameter samples using a Trizol reagent (Invitrogen Corp.Carlsbad, CA, USA), after which, the concentration of RNA was adjusted to 500 ng/μ1. The extracted RNA (2 μg) was used to synthesize cDNA (reaction volume 20 μl) using a RevertAid First Stain cDNA Synthesis Kit (MBI Fermentas Inc, Lithuania).
Primer sequences
Primers were designed by the soft Primer Express 5.0, synthesized by the Shanghai Sangon Biological Engineering Technology&Services Co. The sequences used for primers were as follows: 5’-AAT ACG AAA AGG ATA TTG CT-3’ (sense); 5’-GCG CTA GAA CCA ACT TAT-3’ (antisense)for HMGB1(226 bp). The sequences 5’-AGG AGG AAG AGG AGG AGC Gt-3’ (sense) and 5’-TGG CAA GGT GGG GTT ATA CAG-3’ (antisense) were used for RAGE (194 bp). The sequences 5’-CCT GGG CAT GGA GTC CTG TG-3’ (sense) and 5’-AGG GGC CGG ACT CGT CAT AC-3’ (antisense) were used for β-actin (289 bp).
qRT-PCR
The qRT-PCR analysis was performed using Rotor-Gene 5700-Fluorescence PCR. The total reaction volume was 20 μl, including 1 μl cDNA, 10 μl QuantiTect SYBR Green PCR buffer, 0.6 μl primers, and 7.8 μl DEPC-H2O. Reaction conditions were as follows: 94°C for 10 s; then 40 cycles at 94°C for 30 s, 60°C(HMGB1, β-actin), and 58°C (RAGE, β-actin) for 30 s, and 72°C for 45 s, followed by 72°C for 10 min. Data analysis using the 2-ΔCt method was as follows: ΔCt=Cttarget–Ctaction, the result of 2-ΔCt value represents the sample relative to the initial amount of cDNA.
Statistical analysis
All experimental data were entered into the data library of SPSS 15.0, which was used to analyze the difference between variables. Statistical significance was calculated using the t test for continuous variables. The one-way analysis of variance (ANOVA) was also used where appropriate. Significance was accepted at a value of P<0.05.
RESULTS
Expression of HMGB1,and RAGE in the CSEC
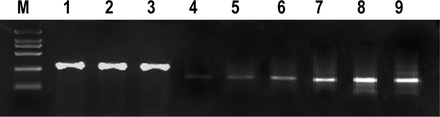
We analyzed 60 CSEC specimens, 60 PS and 30 NCS, to determine the expression of HMGB1, RAGE and β-actin genes. HMGB1 expression in the CSEC specimens and PS was higher compared to the NCS. The expression of RAGE in CSEC specimens and PS was not significantly higher than that of NCS. There were no significant differences in all of the cervical samples (P>0.05, Fig.1, Tables 1,2)
Electrophoresis of β-actin, HMGB1 and RAGE genes by qRT-PCR. M: Marker(2000 bp). 1 2 3: expression of β-actin (289 bp) in NCS, PS and CSEC. 4 5 6: expression of HMGB1(226 bp) in NCS, PS and CSEC. 7 8 9: expression of RAGE (194 bp) in NCS, PS and CSEC.
Expression of HMGB1 in normal, paraneoplastic and CSEC.
Expression of RAGE in normal, paraneoplastic and CSEC.
Correlation among HMGB1, RAGE and clinical tumor parameters
Sixty patients with CSEC were grouped according to the tumor metastasis, diameter, grade and stage. We found that the overexpression of HMGB1 in the samples was significantly correlated with the presence of metastasis and stage, but not with the diameter or grade. RAGE expression was not related with the tumor diameter, grade or stage, but correlated with the presence of metastasis (Table 3).
Correlation among HMGB1, RAGE and clinical parameters.
DISCUSSION
HMGB1 is a basic, multifunction cytokine, non-histone-chromosomal-binding protein found in eukaryotic cells. After histones, HMGB1 is the most abundant chromatin protein[4]. It contains 219 amino acids and has two basic structural units, termed HMGB1 domain A and B (known as the A and B boxes) as well as a carboxy tail[5]. HMGB1 is highly expressed in immature and transformed cells, and is an autocrine/paracrine regulator of invasive migration[6]. New studies have aslo shown that HMGB1,which is expressed in many human neoplasms, including prostatic, colorectal, neuroblastic, pancreatic, thyroidal, gastric, lymphoma and mammary cancer, plays an important role in metastasis and extension of tumors[7-10]. HMGB1 is expressed at extremely low levels in most normal adult tissues, but is overexpressed in many different cancer samples as noted above.
In our present study, we found that the expression of HMGB1 in the CSEC samples and their paraneoplastic tissues was higher compared to normal cervical tissues by using the qRT-PCR technique, suggesting that overexpression of HMGB1 may be related to CSEC proliferation. We aslo found strong up-regulation of HMGB1 in cases with metastases and 2 stages of CSEC, indicating there is a relation between HMGB1 and tumor progression and metastasis.
RAGE is a multi-ligand member of the cell surface and belongs to an immunoglobulin superfamily, that is the main receptor[11]. Elevated RAGE expression also has been reported to be associated with cancer invasion, metastasis and chronic diseases (diabetes mellitus, heart disease and cerebrovascular disease) [12-14]. However we failed to find any significant correlation between RAGE expression and size, histological grade, clinical stage and metastasis of CSEC, but we did find RAGE overexpression in the CSEC metastatic group significantly correlated with HMGB-1 by correlative statistical analysis. These results suggest that the overexpression of HMGB1 may cause modulation of the transcriptional expression of many groups of genes reported to play key roles in different biological processes of CSEC progression, proliferation and metastasis. RAGE has a role in CSEC metastatis. Overexpression of RAGE may form a relationship with HMGB1 to promotate CSEC metastasis.
Recently, Kuniyasu et al.[15] have reported a study of 96 patients with gastric carcinoma by an immunohistochemical method. RAGE immunoreactivity was detected in 62 (65%) of the tumors. In contrast, HMGB1 (amphoterin) expression was detected at a high frequency (82/96, 85%), regardless of the histological type. The results suggested that the relationship between HMGB1 and RAGE plays an important role in gastric tumor metastasis, findings which are similar to our results.
At the present time, few reports related to the HMGB1/RAGE expression in CSEC have been published, so our results showing the HMGB1/RAGE relationship may be of vital importance for CSEC therapy. Although the role of HMGB1 in CSEC is unknown, its level of expression may be used to evaluate CSEC prognosis, and potential for metastasis as well as provide stimulas for further reseach on the pathogenesis of CSES metastasis.
- Received March 26, 2007.
- Accepted August 6, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer