Abstract
OBJECTIVE To study the effect of transforming growth factor ß1 (TGF-β1) on differentiation of rhabdomyosarcoma (RMS) cells
METHODS RD (human embryonal RMS cell line) cells, cultured in differentiation medium containing 9-cis retinoic acid (9CRA), were treated with TGF-ß1. Proliferation of the cells was examined by the MTT assay. The differentiation specific proteins (sarcomeric actin and MyHC) and myogenic transcription factors (MyoD1 and myogenin) in the RD cells were assessed by immunofluorescence staining.
RESULTS Compared to control cells, treatment with lower concentrations of TGF-ß1 (0.1 and 0.2 ng/ml) induced an increase in OD values after 4 d (P<0.01), whereas higher concentrations of TGF-ß1 (2 and 5 ng/ml) led to a reduction of cell viability (P<0.01). After exposure to 9CRA, the viability of the cells decreased significantly (P<0.01), while sarcomeric actin, MyHC and myogenin were induced. These changes were antagonized by TGF-β1 (0.1 ng/ml). No changes were observed in expression of MyoD1.
CONCLUSION The RMS cells, derived from myogenic progenitors are committed to a myogenic fate, but are arrested in the differentiation course by the addition of TGF-ß1 which represses some of the myogenic transcription factors.
- TGF-ß1
- proliferation
- differentiation
- rhabdomyosarcoma
INTRODUCTION
The transforming growth factor ß1 (TGF-ß1) family of peptides is expressed in many normal and malignant tissues, and mediates a diversity of cellular responses encompassing growth, differentiation, apoptosis, and migration[1]. An intriguing feature of TGF-β biology is that its effects are cell type-dependent. For example, TGF-ß1 inhibits proliferation of many epithelial cell types, but accumulation of loss-of-function mutations in the TGF-β1/Smad signaling pathway contributes to the development of carcinoma[2]. TGF-β1 stimulates proliferation of some mesenchymal cells[3], and inhibits myogenic differentiation through a Smad3-mediated transcriptional repression[4]. So, in the light of the evidence for the interplay between proliferation and differentiation within the myogenic lineage[5], it is of interest to explore the possibility that TGF-ß1 might induce the steps towards rhabdomyosarcoma (RMS) tumorigenesis. These neoplasms are a class of myoblastsderived tumors expressing muscle-specific markers.
RMS is the most common soft-tissue malignant tumor in children. Embryonal rhabdomyosarcoma (ERMS) is the commonest subtype, followed by alveolar (ARMS) and pleomorphic (PRMS) subtypes. Although most RMS tumor cells are characterized by expression of the myogenic-promoting transcription factor MyoD, RMS cells fail to undergo terminal differentiation into skeletal muscle[6,7]. This suggests that the etiology of RMS is heterogeneous and complex. Our previous studies demonstrated that the RD cells, a human embryonal RMS cell line, over-expresses receptors and downstream molecules Smads 2, 3, 4[8]. We showed that a high exogenous concentration of TGF-ß1 can suppress the growth of RD cells[9], but virtually little is known as to whether the TGF-β/Smad signaling pathway regulates the differentiation of RMS. The present study aims to examine the relationship between the inhibitory effect of TGF-β1 on differentiation and its effect on proliferation in RMS cells.
MATERIALS AND METHODS
Cell culture
RD cells were obtained from the American Type Culture Collection (ATCC)[10]. The cells were grown in HG-DMEM with kanamycin and 20% fetal calf serum (FCS) in 5% CO2 at 37°C. Differentiation was induced in differentiation medium with kanamycin, 5 μM 9-cis retinoic acid (9CRA) (Sigma Chemical Co.USA) and 2% horse serum. This medium has been as model for myogenic differentiation[11]. Stock concentrations of 5 mM 9CRA were prepared in 95% ethanol and stored at -20°C. The stock was diluted to a final concentration of 5 uM. The cells were plated in growth media and incubated overnight. Then the medium was changed to the treatment conditions to begin the experiment. The treatments were either incubation in differentiation media with 9CRA, or treatment with recombinant human TGF-ß1 (endotoxinfree) (R&D, Wiesbaden, Germany). Control cultures contained 0.1% ethanol.
MTT assay
The effect of TGF-ß1on cell growth was evaluated by a viability assay. RD cells were plated at 4×104 cells per well in differentiation media into each well of 96-well plates, with solvent control (ethanol) or with TGF-ß1 (0.1~5 ng/ml). During the last 4 h of each day, cells were plused with MTT (Sigma, USA) 10 μl/well, followed by incubation at 37°C for 4 h to allow MTT formazan formation. The medium and MTT were replaced by 100 μl of dimethylsulfoxide to dissolve the formazan crystals. After 30 min, the optical density (OD) at 557 nm was determined using a microplate reader (Model 550, BIO-RAD Hercules, CA, USA).
Morphology
Changes in morphology and myotube-like structures, which can be seen at confluence, were taken as an indication of differentiation[11]. Observed morphologies may include star-shaped cells, elongated spindleshaped cells, and small round cells. The treatments were either differentiation media with TGF-ß1(0.1 and 5 ng/ml) or with solvent control (ethanol). After 6 days in culture, photographs were taken using an inverted phase contrast microscope.
Immunofluorescence staining
RD cells, after cultivating on coverslips for 24 h in DMEM containing 10% FCS, were treated with TGF-ß1 (0.1 ng/ml) for 6 days in EMEM containing 5 uM 9CRA. Coverslips were then washed with iced PBS and fixed with 4% paraformaldehyde for 15 min at 4°C. After washing with PBS, the cells were blocked with 5% goat serum in PBS containing 1% bovine serum albumin (BSA) at 37°C for 30 min. Then the cells were incubated at 4°C overnight with mouse anti-sarcomeric actin (Dako, Glostrup, Denmark),-MyHC (Zymed, San Francisco, CA, USA), MyoD1 (Dako, Glostrup, Denmark), and rabbit anti-myogenin (Santa Cruz, USA)in PBS containing 1%BSA. After the cells were washed with PBS, a 1:100 dilution of fluorescein isothiocyanate (FITC)-conjugated rabbit anti-mouse IgG (for mouse anti-sarcomeric actin, MyHC, and MyoD1) and goat anti-rabbit IgG (for rabbit anti-myogenin) were applied as the secondary antibody for 1 h at room temperature. The slides were then washed, air dried, mounted, and photographed with a laser scanning confocal microscope (LSCM, Bio-Rad, USA). At least 200 cells/slide in random fields were counted at ×20 to determine the percentage of positive cells. Each experiment was repeated at least three times.
Statistical analysis
The data were presented in the form of the mean± standard deviation. Analyses were carried out using SPSS 10.0 (SPSS Inc, Chicago, IL, USA) Difference between the control samples and TGF-ß1treated samples were compared using the paired-samples t test. P values <0.05 were considered significant.
RESULTS
Effect of TGF-ß1 on the growth of RD cells in a concentration–dependent manner
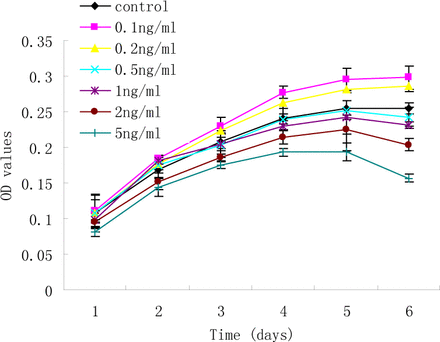
Proliferation curves of RD cells were generated to measure the effect of TGF-ß1 (0.1, 0.2, 0.5, 1, 2, and 5 ng/ml) on growth (Fig.1). RD cells treated with lower concentrations TGF-ß1 (0.1 and 0.2 ng/ml) induced an increase in OD values (P<0.01), whereas higher concentrations of TGF-ß1 (2 and 5 ng/ml) led to reduction of cell viability (P<0.01). Although growth of the RD cells was suppressed by higher concentrations of TGF-ß1, few cell deaths were observed in the cultures, as determined by the uptake of Trypan blue. These results indicate that the induced growth arrest at higher concentrations of TGF-ß1 was not due to an increase in cell death.
Concentration-response to TGF-ß1 on growth of RD cells. RD cells were treated with TGF-ß1 (0.1, 0.2, 0.5, 1, 2, and 5 ng/ml) or without TGF-ß1 for 1~6 d. Cell viability was quantified by the MTT assay. Each experiment was performed three times.
A Low concentration of TGF-ß1 (0.1 ng/ml) antagonizes the growth-inhibitory effect of 9CRA
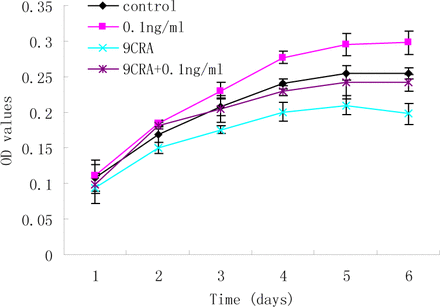
The preliminary findings showed exogenous high concentrations of TGF-ß1 (2 and 5 ng/ml) can suppress the growth of RD cells. To further characterize whether a low concentration of TGF-ß1 has an inhibitory effect on RD cell differentiation, MTT analysis was used to assess the effect of TGF-ß1 (0.1 ng/ml) on viability in the presence of 9CRA. As shown in Fig.2, 9CRA led to a reduction in cell viability in the RD cells (P<0.001), indicating that 9CRA suppresses growth of RD cells. However, there was no significant difference between control and 9CRA+TGF-ß1 (0.1ng/ml) groups throughout the 6-day time course (P>0.05).These results suggest that TGF-ß1 may act as an autocrine growth regulator in RD cells.
MTT analysis of the effect of TGF-ß1 (0.1 ng/ml) on the cell viability in the presence of 9CRA. 9CRA suppressed growth of RD cells (P<0.001), whereas, there was no significant difference between control and 9CRA+TGF-ß1 (0.1 ng/ml) groups throughout the 6-day time period.
Inhibition of myogenic differentiation by TGF-ß1 in RD cells
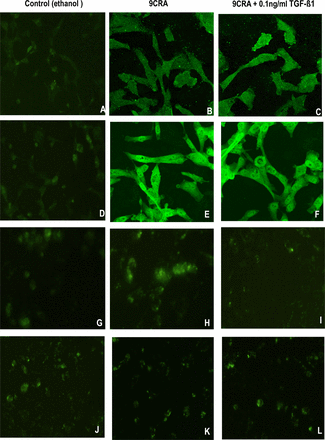
RMS cells show only limited expression of genes associated with terminal differentiation. Specific proteins, such as sarcomeric acitin and MyHC, are markers of myogenic differentiation. Following exposure to 9CRA, the proportion of myotube-like and multinucleated myofiber-like cells increased to a certain extent. In a parallel experiments, immunofluorescence staining was used to detect the expression RD cells showed 20% sarcomeric actin-and 10% MyHC-positive cells (A and D), and 36% sarcomeric actin-and 31% MyHC-positive cells (B and E) with the addition of 9CRA. In the presence of TGF-ß1 (0.1ng/ml) plus 9CRA, about 22% sarcomeric actinand18% MyHC-positive cells were found (C and F).
Immunofluorescence staining was performed at the same time to analyze the expression of myogenic transcription factors myogenin and MyoD1 (Fig.3 G~L). Control RD cells showed 5% myogenin-and 8% MyoD1-positive cells (G and J). With the addition of 9CRA, 10% myogenin-positive cells developed (H), but less myogenin was detected when both 9CRA and TGF-ß1 were applied (4% I). No changes were observed in expression of MyoD1 (J~L).
Inhibition of myogenic differentiation by TGF-ß1. Immunofluorescence analysis of RD cells in control (ethanol) (A, D, G, and J×600), in DMEM containing 5 uM 9CRA without TGF-ß1 (B, E, H, and K) or with 5 uM 9CRA plus TGF-ß1 (0.1 ng/ml) (C, F, I, and L×600) for 6 days. Cells were stained with anti-sarcomeric actin (A, B, and C×600), anti-MyHC (D, E, and F×600), anti-myogenin (G, H, and I×600), and anti-MyoD1 (J, K, and L×600). Positive cells were detected and quantified as described in materials and methods.
In summary, treatment of RD cells with 9CRA resulted in an increase in expression of sarcomeric acitin, MyHC, and myogenin, suggesting a differentiating effect (Table 1). Since 9CRA did not change the expression of MyoD1, 9CRA may induce myogenic differentiation independent of its effects on MyoD1. TGF-ß1 inhibited expression of sarcomeric acitin, MyHC, and myogenin, which were induced by 9CRA alone, indicating that TGF-ß1 may play an important role in myogenic differentiation of RMS cells.
Analysis of myogenic differentiation.
DISCUSSION
Multi-modality therapies involving surgery, chemotherapy, and radiotherapy are necessary in childhood RMS, but the outcome with metastatic RMS remains poor[12]. Therefore, the development of more effective therapeutic tools and strategies are in great need. Since RMS cells are thought to arise as a consequence of regulatory disruption of growth and differentiation, understanding of such a mechanism may open new avenues for the treatment of RMS through the development of targeted therapies.
RMS represents a class of myoblasts-derived tumors, expressing some muscle-specific markers and myogenic promoting transcription factors. However, RMS cells fail to undergo terminal differentiation into skeletal muscle. TGF-ß1 has been reported to negatively regulate muscle cells and RMS cell myogenic differentiation. Some researches have demonstrated different means to produce differentiation in RMS. Alterations of certain signaling pathways, for example those involving IGFs and myostatin, result in similar differentiation of RMS cells[13,14].These findings highlight the complexity of tumorigenicity in RMS.
Ample evidence in the literature has shown that retinoic acid (RA) can induce celluar growth inhibition combined with differentiation, and related agents show promise for the prevention and treatment of several human cancers[15]. RA induces marked myogenic differentiation in skeletal muscle cells[16]. This prompted us to analyze the effects of RA on RMS. Two major histopathological classifications of RMS are embryonal RMS-ERMS and alveolar RMS-ARMS, each of which is characterized by a distinct genetic alteration. The ERMS cell line RD did not respond to all-trans retinoic acid ( ATRA)[17], nevertheless, growth was moderately suppressed by 9CRA[11]. Virtually, nothing is known about the molecular mechanisms by which this occurs.
Our previous studies demonstrated that TGF-ß1 at an exogenously high concentration (5 ng/ml) can inhibit growth of RD cells[8,9].In this current report, higher concentrations of TGF-ß1 (2 and 5 ng/ml) led to a reduction of cell viability of the RD cells. TGF-ß1 used at low concentrations (0.1 and 0.2 ng/ml) induced increases in OD values, and antagonized the growth inhibitory effect of 9CRA. But, under physiological conditions, the function of autocrine TGF-ß1 is not dependent on a high concentration at the cellular level. Both observations suggest a growth-facilitative effect of TGF-ß1 on RMS cells under physiological conditions, and these findings may yield insights into the processes of differentiation-inhibition in RMS. In our study, 9CRA was shown to inhibit RD growth in vitro and induced a marked increase in the expression of sarcomeric actins and MyHC, demonstrating that 9CRA can be an effective inducer of differentiation in RD cell lines. Treatment with TGF-ß1 significantly decreased the expression of muscle specific proteins when compared with the control cells. This observation offers a basis for further study of the effect of TGF-ß1 on the relationship between inhibition of protein expression and differentiation.
The myogenic nuclear regulatory proteins are a group of DNA binding proteins, which act as transcription factors, that induce differentiation by activating muscle specific genes. They are important in the switch from cellular proliferation to differentiation. Muscle specific proteins (sarcomeric actin and MyHC) and myogenic transcription factors (MyoD1 and myogenin) are useful markers of myogenic differentiation in RMS cells[18,19]. That these genes are important for skeletal muscle differentiation has been demonstrated by reports that myogenin knockout mice show severe skeletal muscle defects, despite expressing normal amounts of MyoD[20]. Therefore, myogenin and MyoD appear to have different roles in myogenesis.
In our study, 9CRA induced myogenin expression, but did not change the expression of MyoD1, indicating that 9CRA may induce muscle specific proteins to certain degree independent of its effects on MyoD1. Perhaps this is the reason why the RD cells do not complete differentiation into multinucleated cells. A more complete differentiation response may be achieved by taking advantage of the potential synergistic effects of myogenin and MyoD. TGF-ß1 decreased transcription factor myogenin, which appears to serve as a target for negative regulation of myogenesis by the TGF-ß1 signaling pathway. This pathway controls all downstream events within a differentiation regulatory cascade, such as expression of myogenic differentiation markers (sarcomeric acitin and MyHC). In the present report, we have demonstrated that MyoD1was expressed normally in rhabdomyosarcoma cells but it obviously lacked the biological activity to function as a muscle-specific transcription factor. It is conceivable that this inactivity is either due to posttranscriptional modifications of MyoD1 or to the presence of an inhibitor. A better understanding of howTGF-ß1 modulates growth and differentiation in RMS may have implications for the treatment and prognosis of this neoplasm.
Our recent studies demonstrated that the signaling of TGF-ß1 from the cell surface to nucleus can also be directed through the MAPK (ERK2) pathway as well as the TGF-ß1/Smad signaling pathway[21]. These results suggest that TGF-ß1 promotes tumorigenicity by a mechanism that does not depend strongly on over-expressed receptors (TβR II and TβR I), and that tumor formation involves activation of intracellular MAPK and Rho GTPase activities. Further investigation is needed to determine whether the tumorigenicity of TGF-ß1 in RMS cells is related to modulating the cellular response by the MAPK pathway.
In summary, this study demonstrated that 9CRA can be an effective inducer of growth suppression and enhance muscle-specific protein expression in the RD RMS cell line. In addition, evidence of an increase in OD values induced by TGF-ß1used at low concentrations was observed. Additionally, TGF-ß1 antagonizes the differentiation-inducing effect of 9CRA. Together, these findings offer a basis for further study of the autocrine nature of TGF-ß1 and differentiation-inhibition in RMS.
Footnotes
This work was supported by the grants from the Jiangshu Provincial Higher Institution Natural Science Foundation (No.2134605), Soochow Universtiy Medicine Development Foundation (No.EE134513), and the Jiangshu Provincial Post-Doctoral Foundation (No.51208).
- Received August 17, 2007.
- Accepted October 8, 2007.
- Copyright © 2007 by Tianjin Medical University Cancer Institute & Hospital and Springer