Abstract
OBJECTIVE To assess the effect of 17β–estradiol(E2) on cell proliferation, cell invasiveness and its regulation of MTA3, Snail and matrix metalloproteinase 2 (MMP–2) expression in the ovarian clear cell adenocarcinoma cell line ES–2, and to further investigate the mechanism involved.
METHODS We first investigated expression of ERα, ERβ, PR and E-cadherin of ES–2 cells by RT–PCR and Western blots. Before all experiments, the ES–2 cells were grown in medium depleted of steroid for more than 7 days. Following treatment with 10−7, 10−8 and 10−9M E2, cell viability of the ES–2 cells was determined by the MTT method, and the cell cycle distribution and apoptosis were examined by flow cytometry (FCM). Invasion and mobility assays were performed using modified Boyden chambers. MTA3, Snail and MMP –2 mRNA expression was measured by RT –PCR, and Snail, MMP –2 protein levels were determined by IHC. MMP –2 activity was assayed by zymography.
RESULTS RT –PCR and Western Blots showed that the expression of ERα and E –cadherin mRNA and protein in the ES –2 cells was negative, while ERβ and PR expression was positive. E2 at 10−7,10−8 or 10−9M stimulated cell proliferation. A level of 10−8M E2 reduced the proportion of G0–G1 phase cells and increased the proportion of cells in the S phase, but it had no effect on apoptosis. Invasiveness and mobility of the ES–2 cells was significantly increased by 10−8M E2. Treatment with 10−8M E2 led to reduced MTA3 mRNA expression, and elevated Snail and MMP–2 mRNA and protein levels.
CONCLUSION E2 enhanced invasion by the ES–2 cells. The effects observed maybe mediated by down-regulation of MTA3 and up-reguation of Snail and MMP–2.
- estrogens
- clear cell adenocarcinoma
- Snail
- matrix metalloproteinase 2
Clear cell adenocarcinoma (CCA) constitutes only 5~11% of surface epithelial ovarian cancer, and up to 60% of the patients with CCA have stage I disease at diagnosis. Nevertheless, patients with CCA have poorer prognoses than do those with other pathological types of epithelial ovarian carcinoma. A significant proportion of women (20~50%) with stage I CCA have recurrences and die of their malignancies. Therefore oncologists are highly concerned regarding the characteristics of drug-resistence, early recurrence, and poor prognosis of ovarian CCA.[1] It has been reported that unopposed hyperestrogenism, either endogenous or exogenous, is a significant risk factor for the development of ovarian CCA.[2,3]
The transcription factor Snail super family comprises Snail and Slug. In 2000, it was reported that the Snail protein directly represses E-cadherin expression and may be involved inthe development of cancer.[4,5] Yokoyama et al.[6] showed that Snail contributes to the increased invasion not only through the inhibition of cell-cell adhesion but also through the up-regulation of matrix metalloproteinase 2(MMP-2) expression. Thus, it was suggested that Snail played an important role in adhesion, invasion and metastasis of tumor cells. The report by Fujita et al.[7] showed that MTA3 directly represses Snail expression and identified MTA3 as an ER-regulated component of the Mi2/NuRD complex and that the MTA3 protein was abundant only in cells expressing estrogen receptors. These fingdings might explain how ER status controls invasion and metastasis in human tumors.
In this paper, we have examined the in vitro effect of estrogen on cell proliferation, cell cycle distribution, cell mobility and invasion, and its regulation of MTA3, Snail and MMP-2 expression of ovarian clear cell adenocarcinoma ES-2 cells.
MATERIALS AND METHODS
Reagents
17β-Estradiol(E2), activated charcoal, dextran, methyl thiazolyl tetrazoliun (MTT) and fibronectin were purchased from Sigma. Anti-MMP-2 mouse monoclonal antibodies were purchased from Neomarker. Anti-snail goat polyclonal antibodies were procured from Santa Cruz. Anti-E-cadherin mouse monoclonal antibodies were obtained from Zymed. EnVision solution and HRP-labeled rabbit anti-goat IgG were purchased from DAKO. TRIZOL was the product of Gibco. A reverse transcript kit, Taq DNA polymerase and dNTPs were purchased from MBI Fermentas.
Cell culture
The human ovarian clear cell adenocarcinoma cell line, ES-2, purchased from the American Type Culture Collection, was routinely grown in 5% CO2 in air at 37℃. Growth medium was McCoy’s 5A medium (Gibco) supplemented with 10% fetal bovine serum (Hyclone), lOOu/ml penicillin G and 100µg/ml streptomycin.
The serum was pretreated with dextran-coated activated charcoal to remove steroid hormones as reported. [8] Before all experiments, the ES-2 cell line was grown in media depleted of steroid for 7 days. E2 was dissolved in 100% DMSO and DMSO was added to the culture medium at a final concentration of 0.01% vol.
Cell viability assay by MTT
Cells were plated at a density of 1×104/well in 96-well plates. Four hours later, medium was removed, and medium containing 10−7, 10−8 and 10−9M E2 was added. Parallel cultures treated with vehicle without DMSO and vehicle containing DMSO (0.01%vol). At 72 and 120 h later, 20 µl MTT (5 mg/ml) was added to each well. The medium was aspirated 4 h later, and 150 µl DMSO was added to each well. Finally, optical densities were measured at 490 nm, and changing rates were calculated by the equation: changing rates =OD of each group/OD of vehicle containing DMSO.
Examination of cell cycle distribution and apoptosis by flow cytometry
After cells were incubated in vehicle (DMSO 0.01% vol) or medium with the addition of 10−8M E2, ES-2 cells were collected, washed twice with PBS, fixed in 70% ice-cold ethanol for 30 min at room temperature, and then treated with 200 µl of 1% RNase for 15 min at 37℃. The pellets were resuspended in PBS containing 50 µg/ml of propidium iodide. The staining was performed at 4℃ for at least 30 min, and samples were analyzed by a flow cytometr(Becton Dickinson, USA).
Invasion assay
The invasion assay was performed by using modified Boyden chambers (BD) separated by a porous polycarbonate filter (8 µm pore size), coated with 10µg matrigel on the upper side and 5 µg fibronectin on the lower side of the membrane. The lower chamber was filled with 800 µl vehicle (DMSO 0.01%vol) or medium with 10−8M E2. After 72 h of incubation in control medium with only DMSO or medium with 10−8M E2, 200 µl of trpsinized cells(5×104/ml) suspended by hormone-stripped medium were seeded in the upper chamber. After 72 h the filters were fixed and stained by Gemsa. The total number of cells that crossed the membrane was counted in 5 different fields under a light microscope at ×200 magnification. The results were expressed as x̅± s for each group. Each experiment was performed in triplicate wells.
Migratory capacity assay
The steps were the same as with the invasion assay, expect that the upper side of the membrane was not coated with matrigel and that cells were fixed and stained after 24 h of incubation.
Design and synthesis of PCR primers
Primers for MTA3 and E-cadherin were synthesized according to sequences reported.[7] Primers for Snail, MMP-2, ERα, ERβ, PR and GAPDH were designed and synthesized by the Shanghai Bioasia Biotechnology Co.(Shanghai, China). Primer sequences are shown in Table 1.
RT–PCR
The RNA was extracted from ES-2 cells cultivated in medium with 10% fetal bovine serum or in medium with 10% activated charcoal-treated serum. Then after 0.5, 1, 2, 4, 8, 12, 16 and 24 h of incubation in vehicle (DMSO 0.01%vol) or in medium with l0−8M E2, total RNA was extracted by Trizol. RT was performed according to the manufacturer’s instructions, 1 µg RNA being added to 20 µl of reaction mixture. The PCR reaction mixture consisted of the following: 1 µl RNA template, 2.5 µl 10×PCR buffer, 1.5 µl 25 mM Mg2+, 1µl 10 mM dNTP, 1 µl 10 µM primer×2, 1 µl 5 U/ µl Taq DNA polymerase. Samples were subjected to 3 min at 95℃ for pre-denaturing, followed by 30 thermal cycles of 1 min at 94℃ for denaturing, 1 min for annealing (temperature shown in Table 1), 1 min at 72℃ for extension, and an additional 10 min at 72℃ for final extension after the last cycle. GAPDH was amplified simultaneously as the internal marker. The PCR products were electrophoresedon a 1.5% agarose gel.
Detection of Snail and MMP –2 proteins by immunohistochemical staining
Snail staining was performed by a 2-step method. ES-2 cells were fixed after 72 h of incubation in vehicle (DMSO 0.01%vol) or in medium with 10−8M E2, rinsed with PBS, treated with 0.3% hydrogen peroxide and incubated with 10% nonfat dry milk. The slides were then incubated with primary antibodies (at a dilution of 1:50) at 4℃ over night. After washing with PBS, they were incubated with HRP-labeled rabbit anti-goat IgG, andthen stained with diaminobenzidine. MMP–2 staining was performed by an EnVision method. Following treatment with 0.3% hydrogen peroxide, the slides were incubated with a primary antibodies (all at a dilution of 1:50) at 4℃ over night, washed with PBS, and incubated with the EnVision solution. No staining was obtained when PBS was used instead of the primary antibody.
Determination of MMP activity by zymography
After incubation in medium without serum for 24 h, the ES-2 cells were incubated in either vehicle (no DMSO), vehicle (DMSO 0.01%vol) or medium with 10−8M E2 for 24 h. Then the supernatants were collected. Gelatin was added to a standard acylamide polymerization mixture at a final concentration of 1mg/ml. Ten µg of each sample was loaded, and gels were run at 30 mA for 2~3 h. Following electrophoresis, the gels were rinsed in 2.5% Triton-X 100. Then the gels were incubated for 18 h at 37℃ in reaction buffer(50 mmol/L Tris, 200 mmol/L NaCl, 10 mmol/L CaCl2, pH 7.5). After incubation, the gels were stained with Coomassie Blue R-250 and then destained. The absorption density of the MMP lytic bands on the zymographic gels were scanned and analyzed.
Western blot analysis
Cells were broken in a lysis buffer: 50 mmol/L Tris-HCl pH 8.0, 20 mmol/L EDTA, 1% SDS, and 100 mmol/L NaCl. The samples were boiled for 10 min, centrifuged, and the supernatants collected. Ten µg of each sample was loaded on a 10% SDS-PAGE gel. After electrophoresis, the proteins were transferred onto a polyvinyl difluoride membrane, and the membrane incubated in blocking buffer containing 5% nonfat dry milk for 2 h at room temperature. Then the membrane was probed with a primary antibody in blocking buffer (ERα 1:100, ERβ 1:500, PR 1:200, E-cad 1:500) overnight at 4℃, followed by probing with the secondary antibody in blocking buffer (at a dilution of 1:2000) for 2 h at room temperature. Signal intensity was detected by an enhanced chemiluminescence detection kit.
Statistical analyses
Data were analyzed with Student’s t Test by software SPSS 11.0. Differences were considered to be significant at P <0.05.
RESULTS
Expression of steroid receptors and E-cadherin in ES–2 cells
The expression of ERα and E-cadherin was not detected by RT-PCR or Western blots, while the expression of ERβ and PR mRNA and protein were positive.
The effect of E2 on cell proliferation and cell cycle distribution
The MTT assay showed that compared with vehicle, 10−7, 10−8 and 10−9M E2 all stimulated proliferation of ES-2 cells significantly after 5-days of incubation (P=0.006, 0.000, 0.002, respectively).There was no significant difference among the effects of the 3 concentrations of E2. When incubated for only 3 days, there were trends of stimulation of proliferation, but the differences were of no significant importance. The results are summarized in Table 2..
Cell cycle distribution and apoptosis were determined 48 h after treatment with 10−8M E2. Compared with the vehicle alone (DMSO 0.01%vol), 10−8M E2 changed the cell cycle distribution, reduced the proportion of cells in the G0-G1 phases(P =0.047), and increased the proportion of cells in the S phase (P= 0.024), but it had no effect on apoptosis. The results are summarized in Table 3.
Effect of E2 on cell invasiveness and migratory capacity
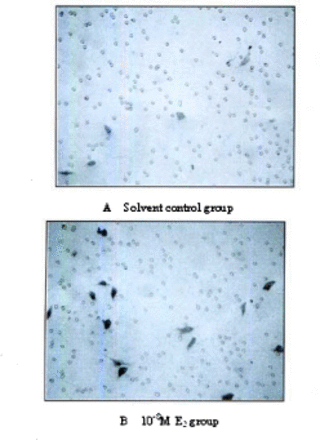
The invasion and migratory capacity of ES-2 cells was significantly facilitated after treated with 10−8 E2 for 72 h. In the invasion assay, the number of cells per field that passed through the membranes was 4.1±0.9 in the control group (DMSO 0.01%vol), and 11.2±0.8 in the 10−8M E2 group (Fig.1)(P=0.001). And in the migratory capacity assay, the number was 12.2±2.5 in the control group and 58.5±2.6 in the 10−8M E2 group Fig.2(P=0.000).
Results of invasion assay. Cells across the membranes of the modified Boyden chambers (original magnification×400).
Results of cell mobility assay. Cells across the membranes of the modified Boyden chambem (original magnification×400).
Effect of E2 on expression of MTA3 and Snail mRNA and expression of Snail protein
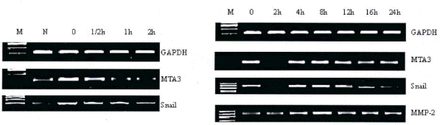
The expression of MTA3 and Snail mRNAs was determined by RT-PCR. The results showed that the level of MTA3 and Snail mRNA was elevated after cultivation for 7 days in hormone-stripped medium. When treated with 10−8 E2, the level of MTA3 and Snail mRNAwent down gradually by the first 2 h.
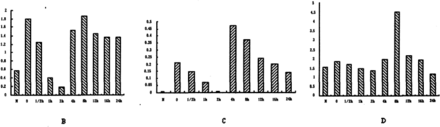
The level of MTA3 and Snail mRNA at each time point during the first 24 h is shown in Fig.3. A, B, C). The expression of MTA3 mRNA went down gradually over the first 2 h, and reached its lowest level at 2 h. Then its expression gradually recovered to the level before E2 treatment. The production of Snail mRNA went down gradually in the first 2 h and then rose to a peak value at 4 h, and then it recovered to the level before E2 treatment. When treated with vehicle (DMSO 0.01% vol), the expression of MTA3 mRNA did not change, and the expression of Snail mRNA slightly went down, but there were no significant differences.
Change of expression of MTA3, Snail and MMP-2 mRNA in ES-2 cells treated with 10−8M E2. M: Marker; N: Cells grown in normal medium (McCoy’s 5A medium supplemented with 10% fetal bovine serum); 0: Cells grown in media depleted of steroids.
Change of expression of MTA3 mRNA in ES-2 cells treated with 10−8M E2. Fig.3C. Change of expression of Snail mRNA in ES-2 cells treated with 10−8M E2. Fig.3D. ChangeofexpressionofMMP-2mRNAinES?2cellstreatedwith 10−8M E2.
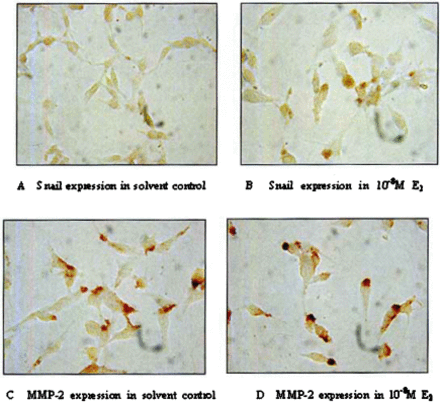
The expression of Snail protein was determined by immunohistochemical staining. There wasslight Snail staining in the nuclei of ES-2 cells in the control group (DMSO 0.01% vol). After incubation in 10−8M E2 for 24 h, positive staining appeared in the cytoplasm of the ES-2 cells, whereas the staining in the nuclei did not change (Fig.4. A, B).
Effect of E2on Snail and MMP-2 expression (immunohistochemistry staining; original magnification×400).
Effect of E2 on expresston of MMP –2 mRNA and protein and MMP activity
We performed RT-PCR to clarify the effect of E2 on expression of MMP-2 mRNA. There were no significant differences in the MMP-2 mRNA level in ES-2 cells cultivated in normal medium with 10% fetal bovine serum compared to cells cultivated in hormone-stripped medium. MMP-2 mRNA expression went up at 4 h after the addition of E2, reached a peak value at 8 h, and then it gradually returned to the level before E2 treatment (Fig.3.D). In the control group (DMSO 0.01%vol), the value of MMP-2 mRNA did not change.
Weak positive staining of MMP-2 was detected by immunohistochemical staining in cytoplasms in the control group (DMSO 0.01%vol). After incubation in 10−8M E2 for 24 h, enhanced staining of MMP–2 was detected in the cytoplasm of the cells (Fig.4. C, D).
Gelatin zymography results (Fig.5) showed that ES-2 cells secreted MMP-2 in the active type (67Kd), as well as a little MMP-9 (92 Kd). After incubation in 10-8M E2 for 24h, the secretion of MMP-2 and MMP-9 increased significantly compared to the control group without DMSO (P =0.007, 0.025). There was little difference between the secretion in the control group with or without DMSO.
Change in the activity of MMPs in ES-2 cells treated with 10−8M E2(zymography). 1: ES-2 cells grown in control medium (no DMSO); 2: ES-2 cells grown in control medium (DMSO 0.01%vol); 3: ES-2 cells grown in 10−8ME2.
DISCUSSION
The present study showed that 10−7, 10−8 or 10−9M E2 stimulated cell proliferation, and that 10−8M E2 reduced the proportion of G0-G1 phase cells, increased the proportion of S phase cells, but had no effect on apoptosis. The results also suggested that 10−8M E2 significantly enhanced invasion and mobility of ES-2 cells,and that cells treated with 10−8M E2 led to reduced expression of MTA3 mRNA, and elevated Snail and MMP–2 mRNA and protein expression. Therefore we conclude that E2 increased invasiveness of the ES-2 cells, and that the effect maybe mediated by down-regulation of MTA3 and up-reguation of Snail and MMP-2.
Effect of estrogen on ovarian CCA
Past studies have indicated that there is certain relationship among ovarian CCA, estrogen and progesterone. The relationship between prenatal exposure to diethylstilbestrol (DES) in the first trimester of pregnancy and CCA of the vagina and cervix has been firmly established. [9] In addition unopposed postmenopausal estrogen replacement therapy has been reported to be a risk factor associated with ovarian clear cell tumors, while no clear association has been shown between the risk of ovarian cancer and the use of hormone replacement therapy with both estrogen and progesterone.[2] Hyperestrogenism has also been shown to be a significant risk factor for the development of ovarian CCA from endometriosis. [3] However, ovarian CCA shows a distinctive immunoprofile characterized by immuno-negativity for ERa and low immunoreactivity for PR.[10] Nevertheless, there is little known regarding whether estrogen has some effect on the growth of CCA, and if so by which mechanism the effect of estrogens is mediated. The ES-2 cell line utilized in our research is negative for ERa, conforming to the characteristics of ovarian CCA.
The relationship between Snail and adhesion, invasion and metastasis of tumor cells
The transcription factor Snail, first described in Drosophila in 1984, is the prototype of a family of zinc finger proteins.[11] In 1994, Corbo et al.[12] reported that Snail family members were implicated in the triggering of epithelial-mesenchymal transitions (EMT) during embryonic development. Snail was described as contributing to repression of transcription of the cell adhesion molecule E-cadherin, and that the induction of EMT by Snail was mediated by the direct transcriptional repression of E-cadherin.[4,5] These results indicated that Snail might be involved in the development of cancer. Snail was also reported to down-regulate aromatase expression, and to affect the serum estrogen level.[13] Yokoyama et al.[6] showed that over-expression of Snail led to higher levels of MMP-2 activity and its gene expression. Thus Snail contributed to the increased invasion not only through the inhibition of cell-cell adhesion by down-regulation of E-cadherin but also by promotion of cell invasion through up-regulation of MMP-2 expression, playing an important role in adhesion, invasion and metastasis of tumor cells. The transcription factor Snail super family is comprised of Snail and Slug.
Our study showed that in ES-2 cells treated with 10−8M E2, Snail mRNA expression went down gradually in the first 2 h, and then went up and came to a peak value at 4 h. With the regulation of Snail, MMP-2 mRNA expression went up at 4 h after the addition of estrogen, and reached a peak value at 8 h. Immunohistochemical staining showed enhanced staining of MMP-2 in the cytoplasm of ES-2 cells, and gelatin zymography results showed that the secretion of MMP-2 increased significantly after incubation for 24 h in 10−8M E2, which might be caused by an up-regulation effect on MMP-2 by Snail.
The degradation of the extracellular matrix (ECM) is an essential step in infiltration bytumor cells, and MMP-2 is among the most important enzymes that can lead to this degradation.Our study suggests that Snail plays a significant role in estrogen-induced reinforcement of invasion by ES-2 cell, and that Snail might serve as an important target in the treatment of ovarian CCA.
Direct repressive effect of MTA3 on Snail
Metastasis-associated genes (MTAs) comprise a novel gene family with a growing number of recognized members. Currently, there are 3 known genes encoding 6 isoforms(MTAl, MTAls, MTA-ZG29p, MTA2, MTA3, MTA3L).[7] However, MTA proteins do not seem to possess enzymatic activity, so the mechanism of their function remains a mystery. The report by Fujita et al.,[7] identified MTA3 as an ER-regulated component of the Mi2/NuRD complex (nuclear remodeling and deacetylation complex), and showed that MTA3 directly repressed transcription from Snail, but not from Slug. A chromatin immunoprecipitation assay showed that MTA3 associated with the Snail promoter through interaction with unidentified DNA binding proteins, suggesting MTA3 directly repressed Snail transcription.
In our study, MTA3 mRNA expression gradually went down in the ES-2 cells after treatment with 10−8M E2, and reached the lowest level at 2 h. With decreased repression of Snail transcription by MTA3, Snail mRNA expression went up and reached a peak value at 4 h, and increased Snail protein expression was observed by immunohistochemical staining after incubation in 10−8M E2 for 24 h, which reflected the direct repressive effect of MTA3 on Snail.
MTA3 serving as a key component of an estrogendependent pathway
Fujita et al.[7] reported that ER up-regulated MTA3 expression, and that MTA3 protein was abundant only in cells expressing estrogen receptors. This ER signaling up-regulates MTA3 levels to negatively modulate Snail-mediated repression of E-cadherin, suggesting MTA3 serves as a key component of this estrogen-dependent pathway.
We selected an ERα negative, ERβ and PR positive cell line to observe the effect of E2 on MTA3, Snail and MMP-2 expression without the participation of ERα. Contrary to ERα positive cell lines, MTA3 mRNA levels of this ERα negative and ERa positive ES-2 cell line gradually went down after treatment with 10−8 M E2, andreached the lowest level at 2 h. Then Snail mRNA and protein values, as well as MMP-2 mRNA and protein expression and enzyme activity gradually increased, which can be contributed to the different effect of ERα and ERβ on MTA3.
Different effects of ERα and ERβ on transcription
ERα and ERβregulate gene transcription in two ways. The first is mediated by classical estrogen response elements (ERE). Upon activation by cognate ligands, ERα and ERβcan form homodimers (αα or ββ) as well as heterodimers (αβ), and then bind to ERE.[14,15] The ER also mediates gene transcription from an API enhancer element that requires ligand and the API transcription factors Fos and Jun for transcriptional activation. [16] ER α and ER α signal in opposite ways when complexed with estradiol from an API site: with ERα, E2 activates transcription, whereas with ERβ E2 inhibites transcription. So MTA3 expression of ERa positive T47D cells was reported [7] to decrease in hormone stripped medium with thedecrease in ERa, and to increase with the increase in ERa at the first 2 h of treatment withE2. The cell line used in the present study was ERa negative and ERβ positive, so MTA3 mRNA expression elevated in hormone stripped medium, and decreased in the first 2 h after treatment with estrogen. This difference was attributed to different roles of ERα and ERβ in regulation of transcription.
In the present study, it was shown that E2 enhanced invasion by ES-2 cells through upregulation of MMP-2, and that the effect maybe mediated by down-regulation of MTA3 mediated repression of Snail through ERβ. Thus we suggest that snail plays an essential role in estrogen-induced reinforcement of cell invasion of ovarian CCA, and might serve as a novel target in its treatment. However, it is still unknown whether Snail is the most important factor that manipulates the expression of MMP-2, and it is also unknown whether reinforcement of cell invasion and up-regulation of MMP-2 by estrogen can be folly blocked by repression of Snail expression.
- Received March 7, 2005.
- Accepted May 20, 2005.
- Copyright © 2005 by Tianjin Medical University Cancer Institute & Hospital and Springer