Abstract
OBJECTIVE This study was conducted to investigate ATP-induced growth inhibition in human leukemic cells KG1a.
METHODS ATP inhibited cell growth was analyzed by MTS assay. Externalization of phosphatidylserine could be detected by Annexin-V-FITC apoptosis staining after activation of the P2X7 receptor. P2X7 mediated pore formation was detected in KG1a cells by Yo-Pro-1 uptake assay.
RESULTS ATP inhibited cell growth in a dose-dependent manner. The cytotoxic effect could be blocked by P2X7 antagonists, oxidized ATP (oATP) and KN62. Externalization of phosphatidylserine could be detected in a time-dependent manner. P2X7 mediated pore formation could be detected in KG1a cells. These effects could not be observed in P2X7 null Ramos cells.
CONCLUSION The results and our previously reports that mRNA, protein expression and calcium response of the P2X7 receptor in KG1a cells, suggested that extracellular ATP effectively induces growth inhibition through apoptosis in KG1a cells by activation of P2X7 receptor, and that may be mediated by extracellular Ca2+ influx and pore formation.
keywords
Introduction
Extracellular nucleotides exert diverse effects on a variety of tissues and cell types via specific receptors named P2 receptors, which consists of P2X and P2Y families. P2X ionotropic receptors form nonselective monovalent cation channels that allow, in most cases, anion or Ca2+ influx from the extracellular space. Seven members of the P2X family have been cloned in mammals (P2X1-7). The P2Y receptors are coupled to trimeric G proteins and are composed of eight subtypes in mammals (P2Y1, P2Y2, P2Y4, P2Y6, P2Y11-14). The P2X7 receptor is a member of the P2X family, and the presence of P2X7 receptor has been found in excitable tissues and in hematopoietic-derived immune cells[1-4]. The natural agonist of P2X7 receptor is ATP, but the 3’-O-(4-benzoyl) benzoyl-ATP (BzATP), a synthetic, P2X7-selective agonist, is a more potent agonist than ATP.
Upon stimulation of agonist, the P2X7 receptor facilitates a rapid, bidirectional flux of cations, thereby triggering depolarization, and massive influx of Ca2+. Furthermore, continued stimulation of the P2X7 allows formation of a large, nonspecific pore, allowing permeability of molecules up to 900 Da[5-7]. Agonists of P2X7 receptor can induce cell death in cells of the immune system, including thymocytes, T cells, macrophages, monocytes and dendritic cells. Cell death may occur through either necrosis or apoptosis[8-10]. Indeed, apoptotic as well as necrotic cell death patterns have been observed after treatment with ATP in several murine and human cell lines[11-13]. ATP can induce cell death in glomerular mesangial cells, which may have pathogenic implications for some forms of kidney disease[14,15]. ATP has been shown to be growth inhibitory or cytotoxic for several mammalian cell lines such as transformed mouse leukemia cells[16], as well as in human cell lines such as some leukemia cells[17]. Treatment with ATP was discussed as an alternative cytostatic principle which could find possible application in therapy of human diseases[18]. However, the mechanisms underlying the cytotoxic effects of extracellular ATP have not been fully elucidated, although several mechanisms have been described, suggesting that the mode of ATP-mediated cell death strongly depends upon the cell type used for the study[19,20].
We have previously reported the expression and characteristics of the P2X7 receptor positive (P2X7+) cell line KG1a[21]. We have since been investigating the inhibition of cell proliferation, apoptosis and the mechanisms involved in these effects by extracellular ATP in KG1a cells. The present study was carried out to establish the role of apoptosis in this growth inhibitory effect. The data presented here shown that agonists of the P2X7 receptor can induce growth inhibition and apoptosis of KG1a cells.
Materials and Methods
Drugs and reagents
Adenosine 5’-triphosphate (ATP), 3’-O-(4 benzoylbenzoyl) - ATP (BzATP), D-glucose, bovine serum albumin (BSA), Yo-Pro-1, 1-[N, O-Bis (5-isoquinolinesulfonyl)-N-methyl-L-tyrosyl]-4-phenylpiperazine (KN-62), oxidized ATP (oATP), 3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), and N-methyl-D-glucamine (NMDG) were purchased from Sigma Chemical (St. Louis, MO). Annexin V-FITC apoptosis detection kit I was purchased from BD Pharmingen.
Cell culture
KG1a and Ramos cells were maintained in RPMI 1640 medium supplemented with 10% fetal calf serum (FCS), glutamine (2 mM), penicillin (100 U/ml) and streptomycin (100 μg/ml), at 37°C in humidified air with 5% carbon dioxide.
Assay of cell viability
Cell viability was determined by MTT method. Cells were plated in 96-well culture plates in triplicate at a density of 1 × 105 per well and cultured at 37°C for 24 h. After exposure to different concentrations of agonists, ATP or BzATP, cells were cultured for an additional 20 h. Then, cells were incubated with MTT for 4 h. Finally, cells were lysed in DMSO and the absorbance at 546 nm was measured with a microplate reader. In blocking experiments, cells were treated with the antagonists of P2X7, KN-62 and oATP, 1 h prior to the treatment with agonists. Blank control samples were also set when cells were without agonist or antagonist treatment. All data are expressed as the mean ± SD.
Detection of exposure of phosphatidylserine on the outside of the cell membrane
To detect the early membrane changes of apoptosis, the annexin-V-FITC apoptosis staining kit was used for flow cytometric detection of phosphatidylserine externalisation on the outside of the cell membrane. In brief, cells were suspended in medium, and treated with drugs for the indicated time. The cells were washed with annexin-binding buffer and incubated for 15 min at room temperature with 0.25 mg/ml of FITC-annexin-V and 10 mM propidium iodide. At least 10,000 cells were analyzed and apoptosis (FITC-positive and propidium iodide negative-cells) was assessed by flow cytometry (FACScan, Becton Dickinson).
Yo-Pro-1 uptake
Pore formation mediated by P2X7 receptor was detected by uptake of Yo-Pro-1, a fluorescence DNA chelating dye[22]. Cells resuspended at 1 × 106 cells/ml in the Locke’s solution (154 mM NaCl, 5.6 mM KCl, 2.2 mM CaCl2, 1.2 mM MgCl2, 10 mM glucose, 5 mM HEPES, pH 7.4)[23], containing 10 μM Yo-Pro-1 were transferred into a 10 × 10 mm quartz cuvette placed in the thermostat-regulated sample chamber of a dual-excitation beam spectrofluorometer (F-4500; Hitachi, Tokyo). Cells were continuously stirred with a circular stir and stimulated with agonists at 37°C. The fluorescences change in Yo-Pro-1-containing cell suspension was continuously monitored with an excitation wavelength of 490 nm, and emission of 509 nm. At the end of the experiments, excessive digitonin was added to the cell suspension to obtain maximal fluorescence.
Results
Effect of P2X7 receptor agonists on KG1a cell death
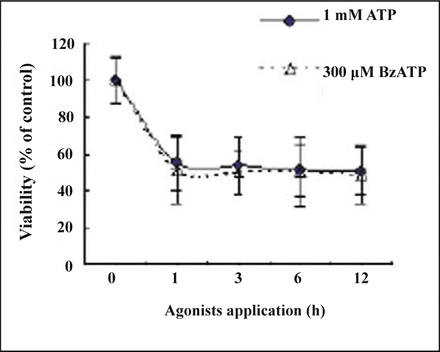
To examine the cytotoxic effect of P2X7 receptor agonists ATP and BzATP on KG1a cells, cell death was assessed using the MTT method. ATP and BzATP were added as a single dose can induce KG1a cells death obviously (Fig.1). Incubation of cells with 1 mM ATP can produce an obvious reduction in cell viability. ATP is an agonist for both P2X and P2Y receptors, but the P2X7 receptor has a higher threshold of ATP activation (≥ 1 mM) than other P2 receptors[24,25]. We also treated the cells with 300 μM BzATP, the specific, complete and most potent agonist for the P2X7, and we obtained similar results as 1 mM ATP treated. These results suggested that agonists used in our experiment induce cell death by stimulation of P2X7 receptor.
Cells were incubated with 300 μM Bz-ATP or 1 mM ATP for indicated times, then washed with agonist-free medium and cell viability measured 24 hrs after agonist exposure. Bars represent the mean ± SD from three experiments.
In KG1a cells, 1 mM ATP induced reduction of cell viability. The reduction after 1, 3, 6, and 12 h after ATP exposure was calculated as 46.7%, 48.5%, 50.5% and 52.1% of the untreated control culture, respectively, while at the concentration of 300 μM BzATP the cell viability was 45.8%, 47.1%, 48.4% and 49.6 % of the untreated control culture. We found that the agonists ATP and BzATP have the same effect in inducing KG1a cell death, and they can induce dramatic reduction in cell viability after 1-hour incubation.
Dose-dependent cytotoxic effect of P2X7 receptor agonists on KG1a cells
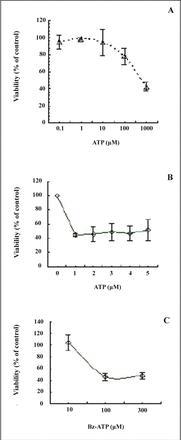
To examine P2X7 receptor agonists ATP and BzATP-induced cytotoxic effect in more detail, the dose-dependence of treatment with agonists of P2X7 receptor was investigated (Fig.2). A 24h-incubation with ATP showed a cell growth inhibitory effect in a dose-dependent manner (Fig.2A). Cytotoxicity was found at concentrations reaching 1 mM ATP. Cell viability and cell growth kinetics of the cell culture were not significantly altered in concentrations below 1 mM ATP. These data supported the hypothesis that the cytotoxic effect of the high concentration ATP was mediated by P2X7 receptor. To further corroborate this hypothesis, we performed experiments with higher concentrations of ATP (Fig.2B). Cytotoxic effect could also be observed at concentrations exceeding 1 mM, but cytotoxicity was not further increased.
Concentration response curve for the decrease in cell viability produced by ATP and BzATP measured after 24 h incubation with the agonist. Cell viability was determined by succinatetetrazolium reductase activity. Bars represent the mean ± SD from three experiments.
After 24 h incubation of KG1a cells with BzATP also showed a cell growth inhibitory effect in a dose-dependent manner (Fig.2C). There was no effect in decreasing in cell number when the BzATP concentration was below 100 μM. We detected a decrease in cell number following up to 100 μM BzATP treatment.
Effect of P2X7 antagonists on agonists induced cell death in KG1a cells
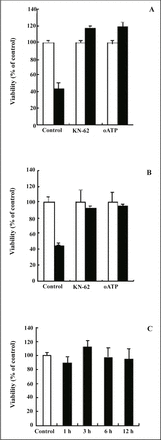
There are currently very few pharmacological blockers for the P2X7 receptor. The most potent one is KN-62, an isoquinoline derivative. A less potent agent but one more suited for long term studies on cell proliferation than KN-62, is oxidized ATP (oATP), an ATP analog that covalently inhibits and thus irreversibly blocks P2X7. We tested the effect both of KN-62 and oATP on cells viability. Fig.3A shows that treatment with an optimal concentration of 300 mM oATP and 3 μM KN-62, can inhibit cell death induced by 1 mM ATP. Treatment with 1 mM ATP for 24 h killed over 41.44% KG1a cells. This reduction in cell viability by ATP was completely inhibited by adding oATP or KN-62 to the culture medium. Both treatments are known to block P2X7 activation. Fig.3B shows the same treatment of P2X7 receptor antagonists to the cytotoxic effect induced by 300 μM BzATP in KG1a cells. 300 μM BzATP can kill 44.56.5% of the cells and antagonists can inhibit this cytotoxic effect. These results directly demonstrate activation of P2X7 receptor-induced cell death.
Cells were pre-treated with 300 mM oxidised ATP or 3 μM KN-62 for 1 h, then exposed to agonists ATP (A) and BzATP (B) for 24 h. Cell viability was measured 24 h after treatment (■) and in time matched untreated cells (□). (C): Effect of the Bz-ATP application on Ramos cell viability, cells were incubated with 300 μM Bz-ATP for indicated times.
P2X7 receptor agonists failed to induce Ramos cell death
We also investigated whether P2X7 receptor agonist BzATP could produce Ramos cell (a P2X7 null cell line[21]) death. Fig.3C shows the time-dependence of this effect when cells were incubated with 300 μM Bz-ATP. Treatment with 100 mM Bz-ATP for increasing periods of time did not produce progressive reduction in cell viability.
The role of apoptosis in P2X7 receptor induced leukemia cell death
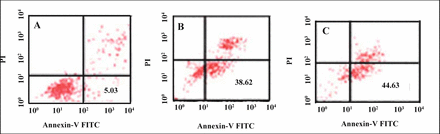
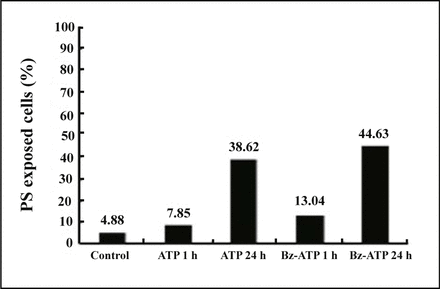
To distinguish the decrease in the number of cells following treatment with ATP and BzATP could be due to either a lower rate of proliferation, or constant proliferation but a higher level of cell death, we investigated the induction of phosphatidylserine (PS) translocation to the cell surface of KG1a cells. The exposure of phosphatidylserine on the outside of the cell membrane has been reported as an index of early apoptosis. The externalization of phosphatidylserine was demonstrated by flow cytometry using FITC-labeled annexin V, which has a high and selective affinity for phosphatidylserine. Compared with untreated control cells, treatment of KG1a cells for 24 h with 1 mM ATP or 100 μM BzATP resulted in a significant increase in annexin V expression (Figs. 4&5). The results showed a shift in expression of PS, and therefore apoptotic cell death, after 1 h to 24 h of treatment with 1 mM ATP, from 7.85% to 38.62% of all cells. 100 μM BzATP can cause 13.04% or 44.63% of cells expressing annexin V after stimulation for 1 h or 24 h, respectively. These findings showed that stimulation of the P2X7 receptor by agonists induces phosphatidylserine translocation to the surface of KG1a cells as early as 1 h after exposure to ATP and BzATP, confirming a role for this receptor in apoptotic cell death.
Cells were treated without (A) or with ATP (B) and Bz-ATP (C) for 24 h. Phosphatidylserine on the outside cell membrane was detected using the annexin-V/PI staining kit.
The cells were treated with or without P2X7 receptor agonists for the time on abscissa. After treatment, cells were labeled using FITC-annexin V and analysis by flow cytometry as described in the Materials and Methods.
P2X7 receptor-mediated pore formation analysis
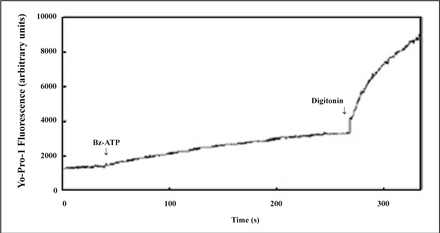
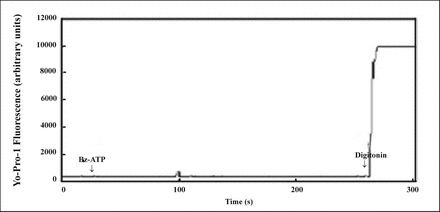
To study the channel dilation of P2X7 receptor under the agonist stimulation, studies were undertaken to measure Yo-Pro-1 accumulation. BzATP (100 μM) could increase Yo-Pro-1 fluorescence during prolonged stimulation (Fig.6). The result indicated that large pores were formed in P2X7+ leukemia cells KG1a by prolonged application of agonist. We examined whether Yo-Pro-1 uptake occurs during addition of P2X7 receptor agonist in P2X7- leukemia cells Ramos. BzATP (100 μM) did not increase Yo-Pro-1 fluorescence during a 4 min exposure (Fig.7). In contrast, membrane disruption by digitonin (50 μg/ml) markedly increased Yo-Pro-1 fluorescence. Yo-Pro-1 or ethidium bromide uptake through the P2X7 receptors is reported to be facilitated at low concentrations of external divalent cations[22]. However, in our experiment, even in a low divalent cation medium (Mg2+-free and 0.18 mM CaCl2), BzATP was unable to stimulate the Yo-Pro-1 uptake in Ramos cells (data not shown).
BzATP 100μM and digitonin were added as indicated (arrows) while Yo-Pro-1 uptake in KG1a cells was measured by fluorescence. Yo-Pro-1 uptake was detected with BzATP, and significant fluorescence was detected with digitonin.
No uptake was detected with BzATP, and significant fluorescence was detected with digitonin.
Discussion
Receptors for extracellular nucleotides have recently become a focus of interest in immunology and hematology due to their high level of expression in blood cells and the vast potential of therapeutic applications afforded by their modulation[26]. The P2X7 receptor is expressed in many different cell types including hematopoietic cells, in which its activation can stimulate proliferation[27], or cause cell death by necrosis or apoptosis[28-30]. However, whether stimulation of this receptor can induce leukemia cell death is still poorly understood.
We previously reported that the expression pattern of P2X7 receptor in a panel of leukemia cells. RT-PCR and FACS analysis confirmed the presence of P2X7 mRNA and protein in KG1a cells. Furthermore, stimulation of P2X7 receptor agonists ATP and BzATP could induce [Ca2+]i increase in KG1a cells[21]. On the contrary, no [Ca2+]i increase was observed in Ramos cells, which were negative for P2X7 mRNA and protein (P2X7-). These data suggested that P2X7 receptor was functional on KG1a cells. In this study, we have examined the potential apoptotic role of P2X7 receptor which is present on leukemia cells. Our data indicated the presence of functional P2X7 receptor on KG1a cells, activation of which induces growth inhibition through apoptosis. Apoptosis studies with agonists (ATP, BzATP) and antagonists (oATP, KN-62) specific for the P2X7 receptor distinguished this receptor from other P2 receptors. Furthermore, ATP failed to induce cell death in P2X7- Ramos cells.
Following exposure to the P2X7 receptor agonist, KG1a cells were killed. This was accompanied by exposure of phosphatidylserine on the outside of cell membrane, suggesting that death was occurring by apoptosis. The cytotoxic effect of ATP on KG1a cells was dose dependent, higher ATP concentrations resulted in higher cytotoxic effects in KG1a cells. The inhibition rate on KG1a cells when treated with 5 mM ATP was comparable to that treated with 1 mM ATP. Similar results were obtained when cells were treated with BzATP.
Extracellular ATP could induce a sustained increase of [Ca2+]i, when stimulated KG1a cells with 1 mM ATP or 100 μM BzATP in[21]. In hepatocytes, ATP-induced cell death is dependent on a rise in intracellular calcium, leading to accumulation of Ca2+ in mitochondria, and loss of mitochondrial membrane potential[31]. This may lead to cytochrome c release and the activation of a cascade of cysteine proteases (caspases) which are known to be involved in programmed cell death. Consistent with this, we demonstrated the elevation of intracellular Ca2+ in KG1a cells, and that activation of P2X7 receptors in KG1a cells leads to cell death.
At present, it is not clear how cell death, which occurred over many hours, is related to the almost immediate changes in membrane ion permeability and elevation in [Ca2+]i. We also tested the P2X7 receptor pore formation. Our results indicated that detectable pore formation could be observed in P2X7+ KG1a but not in P2X7- Ramos cells, the P2X7 receptor in KG1a cells is permeable to YO-PRO-1. The formation of a large cytolytic pore following P2X7 receptor activation has been observed in several cell types, suggesting that opening of the pore is a further conformational change of the P2X7 receptor [32,33]. One possible mechanism of P2X7-mediated cell death has been proposed dependent on the formation of the cytolytic pore[34]. In this case, the positive coupling we observed between receptor activation and pore formation in KG1a cells might account for the long delay between receptor activation and cell death. Further studies will be necessary to elucidate the exact role of intracellular Ca2+, mitochondrial cytochrome c release, pore formation and the activation of caspases in P2X7 receptor-triggered cell death of KG1a cells.
In conclusion, we have shown here that ATP and BzATP could induce cell death in P2X7+ human leukaemic cell line KG1a, and this effect is dependent upon time and concentration. Agonist mediated apoptosis via P2X7 receptor may be mediated by extracellular Ca2+ influx and pore formation in KG1a cells.
Footnotes
This work was supported by a grant from the National Natural Science Foundation of China (No.30671092).
- Received July 11, 2008.
- Accepted October 16, 2008.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer