Abstract
OBJECTIVE To investigate the correlation between polymorphism of the 5’-untranslated region (5’-UTR) of thymidylate synthase genes, as well as the lifestyle, and the susceptibility of gastric carcinoma.
METHODS A case-control study, with 60 cases of gastric carcinoma and 170 cases of general risk population-based controls from Nantong, Jiangsu province, China, was conducted. The epidemiological data, such as living habits of the cancer patients, were collected. DNA of peripheral blood leukocytes was obtained from all of the subjects. The TS 5’-UTR tandem repeat genotype was detected using polymerase chain reaction (PCR).
RESULTS There were three TS 5’-UTR genotypes in the group of gastric cancer cases (2R/2R, 2R/3R and 3R/3R) and six TS 5’-UTR genotypes in the group of the controls (2R/2R, 2R/3R, 3R/3R, 2R/4R, 2R/5R and 3R/4R). The genotypic frequencies were respectively 5.0%, 43.3% and 51.7% in the gastric cancer group. Compared with the parameters in the control group, i.e., 4.7%, 31.7%, 60.6%, 1.2%, 1.2% and 0.6%. There were no significant differences between the two groups. Compared with the 3R/3R-genotype individuals who where non-smokers, drank alcohol twice or less each week, drank tea and did not intake pickled food (PF), the risk of gastric cancer significantly went up in the 2R/2R or 2R/3R-genotype people who had habits of smoking, drinking alcohol more than twice each week, no tea drinking but with frequent intake of PF. The adjusted ORs were as follows, 3.79 (95% CI: 2.45~8.64), 3.41 (95% CI: 1.21~8.47), 5.99 (95% CI: 3.01~14.7), and 3.61 (95% CI: 1.81~8.78).
CONCLUSION There is an obvious correlation between the polymorphisms of TS 5’-UTR genotypes and the lifestyle of individuals in the development of gastric carcinoma.
keywords
Introduction
Thymidylate synthase (TS) can be used to intra-cellularly catalyze deoxyuridylic acid, so as to produce deoxythymidine monophosphate after methylation, and regulate a balance between nucleotides, which is necessary during the course of cell DNA duplicating and reparative process. The TS is also one of the enzymes that play a key role in folic acid metabolism and circulation. Therefore, TS is of significant importance to cell proliferation and differentiation, and is an important target enzyme in tumor chemotherapy[1]. TS 5’-untranslated region (UTR) has polymorphisms, and the polymorphisms of the TS gene may result in a change of enzymic activity or function, thus change the susceptibility of individuals to cancers and their prognosis[2]. There is a 28 bp polymorphic repeat sequence at the upstream end of the TS 5’-UTR transcription initiation site, and its frequencies of repetition include two repetitions (2R), three repetitions (3R) or more[3]. However, the latter seldom occurs. It was shown by previous study that the 3R-allelotype mRNA transcription and the efficiency of protein expression were both higher, compared to 2R[4,5], and the high expression of TS protein facilitated the excess cell proliferation, thus escaped senescence and apoptosis[6]. Nevertheless, the relationship between polymorphisms of the TS 5’-UTR and susceptibility of gastric cancer remains uncertain. Since our city has been regarded as a high-risk area of gastrointestinal cancer, the relationship between polymorphisms of the thymidylate synthase gene and susceptibility of gastric cancer was analyzed using molecular epidemiology while the investigation was conducted, so as to further study the correlations among the polymorphism of TS 5’-UTR gene, living habit (LH) and susceptibility of gastric cancer.
Patients and Methods
Patients
Sixty gastric cancer patients, randomized and pathologically diagnosed in the Tumor Hospital Affiliated to Nantong University from June 2006 to March 2007, were classified as the gastric-cancer group (GCG), while 170 people (general-risk population of same age) without medical records of cancers, who received health examinations in the hospital, were divided into the control group (CG). The habits such as smoking, alcohol and tea drinking, as well as eating habit of every object were investigated. All the research objects were of the Han people. None were consanguineous relatives.
Methods
Extraction of DNA
A sodium ethylene diamine tetracetate anticoagulation cuvette was used to collect venous blood samples and extract leukocytes. A QIAamp DNA extracting kit was used to obtain leukocyte DNA. The DNA was kept in an -80°C cryogenic box for storage.
Genotyping
PCR method was used to detect the TS 5’-UTR genotype, and the PCR volume was 20 μl, containing 100 ng DNA, 10 × PCR buffer solution 2.0 μl, 1.5 mol/L MgCl2, 1 UDNA polymerase, 200 μmol/L dNTPs. The upstream primer of TS 5′-UTR was 5’-GTG GCT CCT GCG TTT CCC CC-3’, and downstream primer was 5′-GCT CCG AGC CGG CCA CAG GCA TGG CGC GG-3’. Both the primers were 300 nmol/L. PCR conditions were as follows: an initial denaturation at 94°C was conducted for 5 min; then a process at 94°C for 1 min, 63°C for 1 min and 72°C for 2 min. After a total of 35 cycles, the process at 72°C was extended for 10 min. A 2%-agarose gel electrophoresis of the PCR product was performed. The 2R and 3R allelic amplification primers were expected to be 215 bp and 243 bp, respectively.
Statistical analysis
Stata 8.0 software was used for conducting the statistical analysis and a fourfold table χ2 test for comparison of the incidence rate, P < 0.05 indicated that there was a statistically significance in the difference between the groups. The Mantel-Haenszel χ2 test was employed to analyze the distribution difference of the genotypic frequency and statistical significance of the relative risk (RR). The RR was signified using the odds ratio (OR) and the 95% confidence interval (95% CI), a multifactorial and unconditioned logistic regression model was used to calculate and regulate the OR and its 95% CI (reference). The LH items were divided into smoking (never smoke or smoke regularly), alcohol drinking (occasionally or frequently), tea drinking (never or regularly), and other eating habits (never and often eat pickled food [PF]) while the analysis was conducted. The smokers were defined as those who at least smoked 1 cigarette everyday, with a successional period of 6 months, and the rest were non-smokers, and the phrases “frequent alcohol drinking” and “to often eat a food” were defined as a intake frequency of 2 times/week. No tea drinking indicated a monthly tea consumption of less than 10 g. Then the rest ought to be tea drinkers.
Results
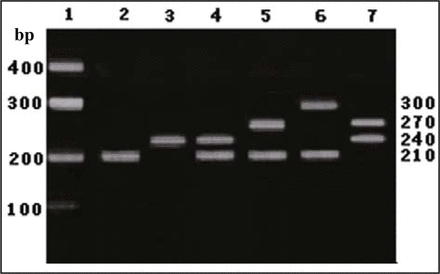
TS 5’-UTR genotypic distribution (Fig.1)
TS 5’-UTR genotype was detected using polymerase chain reaction (PCR). 1, The markers respectively represent 100 bp, 200 bp, 300 bp and 400 bp; 2, 2R/2R genotype; 3, 3R/3R genotype; 4, 2R/3R genotype; 5, 2R/4R genotype; 6, 2R/5R genotype; 7, 3R/4R genotype.
Results of TS 5’-UTR tandem repetitive sequence genotyping have shown 4 PCR segments (approximately 210, 240, 270, 300 bp), and sequence analysis has confirmed that they respectively represented 2 (2R), 3 (3R), 4 (4R) and 5 (5R) repetitive alleles with same 28bp, thus forming the 6 genotypes, i.e., 2R/2R, 2R/3R, 3R/3R, 2R/4R, 2R/5R and 3R/4R.
Test of equilibrium in GCG and CG (Table 1)
Genotypic distribution of genders, ages and habits such as smoking, alcohol and tea drinking, and foods between group with gastric cancer cases and group of controls.
There were 43 male and 17 female cases in the GCG, and in CG 128 male and 42 female, respectively. There were no statistical differences in gender and age distribution between GCG and CG. It was found upon analysis of the people’s habits with respect to cigarettes, alcohol and tea consumptions that the smokers accounted for 76.7% of the cases in GCG, which was significantly higher compared to that in CG (56.5%), P = 0.006. Tobacco other than cigareretts needs to be adressed formally. The patients who frequently drank alcohol accounted for 50.0% in GCG, which was significantly higher than the percentage of the subjects who had this habit and belonged to the CG (26.5%), P = 0.002. The subjects having the habit of tea drinking accounted for 52.9% in the CG, significantly higher compared to GCG (36.7%), P = 0.03. In GCG, the percentage of the patients with frequent intake of PF amounted to 65.0%, apparently higher than that in CG (42.4%), P = 0.008.
TS test of genetic equilibrium and genotypic distribution in GCG and CG (Table 1)
The test of genetic equilibrium for the frequency of TS genotypic distribution was conducted in GCG and CG, and there were no statistical differences in the observed value and the expected value of the TS genotypic frequency in the two groups, P value > 0.05, indicating that both the groups were in conformity with the Hardy-Weinberg ratio. In GCG, the TS 5’-UTR genetypes included 2R/2R, 2R/3R and 3R/3R, and the genotypic frequencies were 5.0%, 43.3% and 51.7%, respectively. In the CG, TS 5’-UTR geneopes included 2R/2R, 2R/3R, 3R/3R, 2R/4R, 2R/5R and 3R/4R, and the genotypic frequencies were 4.7%, 31.7%, 60.6%, 1.2%, 1.2% and 0.6%, respectively. There were no significant differences in genotypic distribution between the two groups (P = 0.161).
Relationships among TS genotype and smoking, alcohol drinking, tea drinking and eating habits, and susceptibility of gastric cancer (Table 2)
Relationship between polymorphism of TS5’-UTR gene and susceptibility of gastric cancer referring to various living habits.
In our study, compared to the people carrying the 3R/3R genotype without a habit of frequent smoking, there was an significant increase in risk of gastric cancer in those of 2R/2R and 2R/3R genotypes with a frequent smoking habit and the adjusted OR value was 3.79 (95% CI: 2.45~8.64). In comparison with the people of 3R/3R genotype who had an alcohol drinking habit of less than twice a week, the risk of gastric cancer significantly rose in those carrying the 2R/2R and 2R/3R genotypes, with a regular alcohol drinking habit of over twice a week and the adjusted OR value was 3.41 (95% CI: 1.21~8.47). Compared to the people of 3R/3R genotype who also had a tea drinking habit, the risk of gastric cancer obviously rose in those of the 2R/2R and 2R/3R genotypes without the tea drinking habit, and the adjusted OR was 5.99 (95% CI:3.01~14.7). Compared with those of 3R/3R genotype, with infrequent intake of PF, the risk of gastric cancer apparently rose in the people of 2R/2R and 2R/3R genotypes with a frequent intake of PF, the adjusted OR was 3.61(95% CI:1.81~8.78) (Table 2). However, it was shown in an analysis of the correlations between the habit of smoking, and those of drinking alcohol and tea and the susceptibility of gastric cancer by the same genotype that the significant rise of gastric-cancer risk could only be found in the population carrying the 2R/2R and 2R/3R genotypes, who also had a regular smoking habit (P = 0.008) (Table 2). It was demonstrated that there was an overt synergistic effect among TS 5’-UTR genotype and smoking, alcohol drinking and tea drinking.
Discussion
Over the past several decades, there were great changes in the incidence spectrum of cancers in China and overseas, possibly resulting from the change of environmental factors. However, cancers are only found in a few people from the population who live in the same environment, showing that there are differences in the hereditary susceptibility of cancers among various individuals[6]. Therefore, the onset of cancer might be the consequence of an interaction between environmental and hereditary factor.
It has been shown by epidemiologic study that dietary factors play an important role in carcinogenesis. The existence of susceptible individuals in the population is because of the presence of genetic polymorphisms related to the oncogenesis. The interactions between gene and gene and between gene and environment are of important influence in the morbidity of cancers. i) Interaction between gene and gene: it was shown in the studies of Zhang et al.[7] that TS 5’-UTR 2R and TS 3′-UTR 6 bp allele may play a synergistic effect in the etiology of gastric cancer in south of China, with a positive correlation. ii) Interaction between gene and environment: it was found by Gao et al.[8] that there was an obvious synergistic action between the TS 6bp/-6bp genotype and the LH of smoking, alcohol drinking, and the choice of not drinking tea in the increase of gastric cancer risk. iii) Influence of the folic acid level. A folic acid level of less than 140 ng/ml in human RBC or a folic acid level of less than 3 ng/ml in the blood can induce chromosome damage and increase the risk of carcinogenesis. The risk of colorectal cancer was significantly raised in the TS 5’-UTR 2R allele carrier and those with a low intake of folic acid, with an interaction between the two populations[9]. Since the coupling pathway of thymidylate synthesis is clear, therefore, the studies on the interaction between the TS gene polymorphisms and dietary factors and its relationship with cancers and other diseases can not only provide valid evidence for the influence on cancers, which is results from the interaction between the genetic and environmental factors, but at the same time can be of significant importance in the prevention, treatment and theoretical study of cancers.
It has been confirmed by epidemiological research that the dietary factors can play an important role in carcinogenesis. Although the roles of vitamin-B, such as folic acid, and methionine may only be part of the dietary factors in the carcinogenesis, just as the antitumous effect of the green tea, which can provide both some vitamins with antitumous effect and some antioxidants with the same effect. It is stated that oncogenesis is the result of an interaction between the gene and environment, so the environmental factors are the initiating agents of oncogenesis, and susceptibility to tumors depends on the hereditary features of individuals. TS can regulate the supply and demand equilibrium of 4 nucleotides, which is needed by for DNA repair, and can play a significant role in the folic acid metabolism[8]. Numerous studies demonstrate that sufficient folic acid supply would be of significant importance to cancer prevention, and a low-dose folic acid might increase the risk of oncogenesis in general population, especially, those who have the habit of heavy alcohol drinking[10]. It was found in our study that there is a definite correlation between the habits, such as frequent smoking, alcohol drinking and intake of PF, the choice of not drinking tea and the incidence of gastric cancer, with a statistical significance among all factors This means among all factor one can find one significant correlation, with statistically significant correlation between all factors has a different meaning. This demonstrates in our study that the dietary factors play a definite role in gastric cancer.
Though the effect of TS 5’-UTR gene polymorphism on the expression and function of the enzyme remains unclear, it was shown in some reports that both the mRNA transcription and the efficiency of protein expression of the 3R allelotype were higher compared to 2R[4,5]. Chen et al.[11] believed that the risk of colorectal cancer significantly increased in the population with a low TS expression, as their level of folic acid metabolism was low. It was shown in our study that compared with the patients of 3R/3R genotype with the habit of not smoking, less than two alcohol drinking events per week, tea drinking and no intake of PF, the risk of gastric cancer significantly rose in the patients of 2R/2R, 2R/3R genotype who have the habits of smoking, ingesting alcohol more than twice a week but no tea drinking, and regular intake of PF, and the adjusted ORs were 3.79 (95% CI: 2.45~8.64), 3.41 (95% CI: 1.21~8.47), 5.99 (95% CI: 3.01~14.7) and 3.61 (95% CI: 1.81~8.78), respectively. In our study, there was no statistical significance in the difference of the TS 5′-UTR genotypic frequency distribution between the GCG and CG, however, it could be seen, after a combined analysis of the LH, such as smoking, alcohol drinking, tea drinking and intake of PF, etc. that the habits of smoking, alcohol drinking, choice of not drinking tea and intake of PF and the TS 5′-UTR genotype synthetically played an obvious synergistic effect in the onset of gastric cancer. This indicates that TS 5′-UTR genotype itself may fail to increase the susceptibility of gastric cancer, nevertheless, following the effect of environmental factors, the hereditary factors can have a synergistic a effect on the susceptibility of gastric cancer.
In conclusion, the polymorphism of genes and the LH have an apparent synergistic effect on the susceptibility to gastric cancer. Therefore, to investigate gene polymorphism and the susceptibility of tumor development will be of a definite significance for incidence, in prevention and treatment of tumors.
Footnotes
The work was supported by a grant from Nantong Municipal Bureau of Public Health, Jiangsu Province, China (2006 [No.29]).
- Received August 29, 2008.
- Accepted November 11, 2008.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer