Abstract
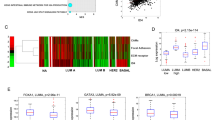
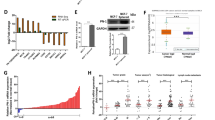
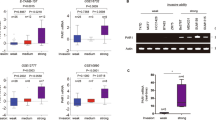
SIPA1 (signal-induced proliferation-associated protein 1) is a GTPase activation protein that can catalyze the hydrolysis of Rap1 bound GTP to GDP. Recently attention has been paid to a potential role for SIPA1 in cancer metastasis; however, the underlying mechanism of how changes in SIPA1 levels may lead to increased metastasis remains poorly understood. In this study, we showed that SIPA1 was mainly localized to the nuclei in highly invasive breast cancer tumor tissue and MDA-MB-231 cells. Knockdown of SIPA1 in MDA-MB-231 altered cell morphology and cell proliferation ability. Furthermore, this study is the first to establish that nuclear SIPA1 can interact with the integrin β1 promoter and activate its transcription; this interaction appears to be important for SIPA1-dependent MDA-MB-231 cell adhesion and invasion. We also demonstrated that the phosphorylation of FAK, Akt and the expression of MMP9, downstream signaling molecules of integrin β1, were decreased upon SIPA1 knockdown, and MDA-MB-231 cell invasion was impaired. Taken together, these results suggest nuclear SIPA1 contributes to breast cancer cell invasion through the regulation of integrin β1 signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Redig AJ, McAllister SS . Breast cancer as a systemic disease: a view of metastasis. J Intern Med 2013; 274: 113–126.
Colzani E, Liljegren A, Johansson AL, Adolfsson J, Hellborg H, Hall PF et al. Prognosis of patients with breast cancer: causes of death and effects of time since diagnosis, age, and tumor characteristics. J Clin Oncol 2011; 29: 4014–4021.
Valastyan S, Weinberg RA . Tumor metastasis: molecular insights and evolving paradigms. Cell 2011; 147: 275–292.
Drukker CA, Bueno-de-Mesquita JM, Retel VP, van Harten WH, van Tinteren H, Wesseling J et al. A prospective evaluation of a breast cancer prognosis signature in the observational RASTER study. Int J Cancer 2013; 133: 929–936.
Kurachi H, Wada Y, Tsukamoto N, Maeda M, Kubota H, Hattori M et al. Human SPA-1 gene product selectively expressed in lymphoid tissues is a specific GTPase-activating protein for Rap1 and Rap2. Segregate expression profiles from a rap1GAP gene product. J Biol Chem 1997; 272: 28081–28088.
Valles AM, Beuvin M, Boyer B . Activation of Rac1 by paxillin-Crk-DOCK180 signaling complex is antagonized by Rap1 in migrating NBT-II cells. J Biol Chem 2004; 279: 44490–44496.
Gao L . Ras-Associated protein-1 regulates extracellular signal-regulated kinase activation and migration in melanoma cells: two processes important to melanoma tumorigenesis and metastasis. Cancer Res 2006; 66: 7880–7888.
Lyle KS, Raaijmakers JH, Bruinsma W, Bos JL, de Rooij J . cAMP-induced Epac-Rap activation inhibits epithelial cell migration by modulating focal adhesion and leading edge dynamics. Cell signalling 2008; 20: 1104–1116.
Bailey CL, Kelly P, Casey PJ . Activation of Rap1 promotes prostate cancer metastasis. Cancer Res 2009; 69: 4962–4968.
Malchinkhuu E, Sato K, Maehama T, Ishiuchi S, Yoshimoto Y, Mogi C et al. Role of Rap1B and tumor suppressor PTEN in the negative regulation of lysophosphatidic acid-induced migration by isoproterenol in glioma cells. Mol. Biol Cell 2009; 20: 5156–5165.
Hernandez-Varas P, Colo GP, Bartolome RA, Paterson A, Medrano-Fernandez I, Arellano-Sanchez N et al. Rap1-GTP-interacting adaptor molecule (RIAM) protein controls invasion and growth of melanoma cells. J Biol Chem 2011; 286: 18492–18504.
McSherry EA, Brennan K, Hudson L, Hill AD, Hopkins AM . Breast cancer cell migration is regulated through junctional adhesion molecule-A-mediated activation of Rap1 GTPase. Breast Cancer Res 2011; 13: R31.
Park YG, Zhao X, Lesueur F, Lowy DR, Lancaster M, Pharoah P et al. Sipa1 is a candidate for underlying the metastasis efficiency modifier locus Mtes1. Nature Genet 2005; 37: 1055–1062.
Crawford NP, Ziogas A, Peel DJ, Hess J, Anton-Culver H, Hunter KW . Germline polymorphisms in SIPA1 are associated with metastasis and other indicators of poor prognosis in breast cancer. Breast Cancer Res 2006; 8: R16.
Farina A, Hattori M, Qin J, Nakatani Y, Minato N, Ozato K . Bromodomain protein Brd4 binds to GTPase-activating SPA-1, modulating its activity and subcellular localization. Mol Cell Biol 2004; 24: 9059–9069.
Shimizu Y, Hamazaki Y, Hattori M, Doi K, Terada N, Kobayashi T et al. SPA-1 controls the invasion and metastasis of human prostate cancer. Cancer Sci 2011; 102: 828–836.
Alsarraj J, Faraji F, Geiger TR, Mattaini KR, Williams M, Wu J et al. BRD4 short isoform interacts with RRP1B, SIPA1 and components of the LINC complex at the inner face of the nuclear membrane. PloS One 2013; 8: e80746.
Crawford NP, Alsarraj J, Lukes L, Walker RC, Officewala JS, Yang HH et al. Bromodomain 4 activation predicts breast cancer survival. Proc Natl Acad Sci USA 2008; 105: 6380–6385.
Alsarraj J, Walker RC, Webster JD, Geiger TR, Crawford NP, Simpson RM et al. Deletion of the proline-rich region of the murine metastasis susceptibility gene Brd4 promotes epithelial-to-mesenchymal transition- and stem cell-like conversion. Cancer Res 2011; 71: 3121–3131.
Su L, Hattori M, Moriyama M, Murata N, Harazaki M, Kaibuchi K et al. AF-6 controls integrin-mediated cell adhesion by regulating Rap1 activation through the specific recruitment of Rap1GTP and SPA-1. J Biol Chem 2003; 278: 15232–15238.
Ji K, Ye L, Toms AM, Hargest R, Martin TA, Ruge F et al. Expression of signal-induced proliferation-associated gene 1 (SIPA1), a RapGTPase-activating protein, is increased in colorectal cancer and has diverse effects on functions of colorectal cancer cells. Cancer Genomics Proteomics 2012; 9: 321–327.
Mathieu V, Pirker C, Schmidt WM, Spiegl-Kreinecker S, Lotsch D, Heffeter P et al. Aggressiveness of human melanoma xenograft models is promoted by aneuploidy-driven gene expression deregulation. Oncotarget 2012; 3: 399–413.
Marques IJ, Weiss FU, Vlecken DH, Nitsche C, Bakkers J, Lagendijk AK et al. Metastatic behaviour of primary human tumours in a zebrafish xenotransplantation model. BMC cancer 2009; 9: 128.
Guo W, Giancotti FG . Integrin signalling during tumour progression. Nature reviews Molecular cell biology 2004; 5: 816–826.
Sahai E . Mechanisms of cancer cell invasion. Curr Opin Genet Dev 2005; 15: 87–96.
Cavallaro U, Christofori G . Cell adhesion and signalling by cadherins and Ig-CAMs in cancer. Nat Rev Cancer 2004; 4: 118–132.
Ruoslahti E . Fibronectin and its integrin receptors in cancer. Adv Cancer Res 1999; 76: 1–20.
Hood JD, Cheresh DA . Role of integrins in cell invasion and migration. Nat Rev Cancer 2002; 2: 91–100.
Mitra SK, Schlaepfer DD . Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol 2006; 18: 516–523.
Mitra SK, Hanson DA, Schlaepfer DD . Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Biol 2005; 6: 56–68.
Maity G, Fahreen S, Banerji A, Roy Choudhury P, Sen T, Dutta A et al. Fibronectin-integrin mediated signaling in human cervical cancer cells (SiHa). Mol Cell Biochem 2010; 336: 65–74.
Sen T, Dutta A, Maity G, Chatterjee A . Fibronectin induces matrix metalloproteinase-9 (MMP-9) in human laryngeal carcinoma cells by involving multiple signaling pathways. Biochimie 2010; 92: 1422–1434.
Maity G, Choudhury PR, Sen T, Ganguly KK, Sil H, Chatterjee A . Culture of human breast cancer cell line (MDA-MB-231) on fibronectin-coated surface induces pro-matrix metalloproteinase-9 expression and activity. Tumour Biol 2011; 32: 129–138.
Scott MS, Troshin PV, Barton GJ . NoD: a Nucleolar localization sequence detector for eukaryotic and viral proteins. BMC Bioinformatics 2011; 12: 317.
Chou KC, Shen HB . A new method for predicting the subcellular localization of eukaryotic proteins with both single and multiple sites: Euk-mPLoc 2.0. PloS One 2010; 5: e9931.
Carragher NO, Frame MC . Focal adhesion and actin dynamics: a place where kinases and proteases meet to promote invasion. Trends Cell Biol 2004; 14: 241–249.
Bos JL, Rehmann H, Wittinghofer A . GEFs and GAPs: critical elements in the control of small G proteins. Cell 2007; 129: 865–877.
Tsygankova OM, Prendergast GV, Puttaswamy K, Wang Y, Feldman MD, Wang H et al. Downregulation of Rap1GAP contributes to Ras transformation. Mol Cell Biol 2007; 27: 6647–6658.
Zuo H, Gandhi M, Edreira MM, Hochbaum D, Nimgaonkar VL, Zhang P et al. Downregulation of Rap1GAP through epigenetic silencing and loss of heterozygosity promotes invasion and progression of thyroid tumors. Cancer Res 2010; 70: 1389–1397.
Zheng H, Gao L, Feng Y, Yuan L, Zhao H, Cornelius LA . Down-regulation of Rap1GAP via promoter hypermethylation promotes melanoma cell proliferation, survival, and migration. Cancer Res 2009; 69: 449–457.
Mitra RS, Goto M, Lee JS, Maldonado D, Taylor JM, Pan Q et al. Rap1GAP promotes invasion via induction of matrix metalloproteinase 9 secretion, which is associated with poor survival in low N-stage squamous cell carcinoma. Cancer Res 2008; 68: 3959–3969.
Carmona G, Gottig S, Orlandi A, Scheele J, Bauerle T, Jugold M et al. Role of the small GTPase Rap1 for integrin activity regulation in endothelial cells and angiogenesis. Blood 2009; 113: 488–497.
Tsukamoto N, Hattori M, Yang H, Bos JL, Minato N . Rap1 GTPase-activating protein SPA-1 negatively regulates cell adhesion. J Biol Chem 1999; 274: 18463–18469.
Hung MC, Link W . Protein localization in disease and therapy. J Cell Sci 2011; 124: 3381–3392.
Katagiri K, Maeda A, Shimonaka M, Kinashi T . RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat Immunol 2003; 4: 741–748.
Bivona TG, Wiener HH, Ahearn IM, Silletti J, Chiu VK, Philips MR . Rap1 up-regulation and activation on plasma membrane regulates T cell adhesion. J Cell Biol 2004; 164: 461–470.
Lindberg K, Strom A, Lock JG, Gustafsson JA, Haldosen LA, Helguero LA . Expression of estrogen receptor beta increases integrin alpha1 and integrin beta1 levels and enhances adhesion of breast cancer cells. J Cell Physiol 2010; 222: 156–167.
Chen C, Peng J, Xia HS, Yang GF, Wu QS, Chen LD et al. Quantum dots-based immunofluorescence technology for the quantitative determination of HER2 expression in breast cancer. Biomaterials 2009; 30: 2912–2918.
Camp RL, Rimm EB, Rimm DL . Met expression is associated with poor outcome in patients with axillary lymph node negative breast carcinoma. Cancer 1999; 86: 2259–2265.
Cayrol C, Clerc P, Bertrand C, Gigoux V, Portolan G, Fourmy D et al. Cholecystokinin-2 receptor modulates cell adhesion through beta 1-integrin in human pancreatic cancer cells. Oncogene 2006; 25: 4421–4428.
Tu H, Xu C, Zhang W, Liu Q, Rondard P, Pin JP et al. GABAB receptor activation protects neurons from apoptosis via IGF-1 receptor transactivation. J Neurosci 2010; 30: 749–759.
Hsu TI, Wang MC, Chen SY, Yeh YM, Su WC, Chang WC et al. Sp1 expression regulates lung tumor progression. Oncogene 2012; 31: 3973–3988.
Liu JF, Crepin M, Liu JM, Barritault D, Ledoux D . FGF-2 and TPA induce matrix metalloproteinase-9 secretion in MCF-7 cells through PKC activation of the Ras/ERK pathway. Biochem Biophys Res Commun 2002; 293: 1174–1182.
Nelson JD, Denisenko O, Bomsztyk K . Protocol for the fast chromatin immunoprecipitation (ChIP) method. Nat Protocol 2006; 1: 179–185.
Keely S, Glover LE, MacManus CF, Campbell EL, Scully MM, Furuta GT et al. Selective induction of integrin beta1 by hypoxia-inducible factor: implications for wound healing. FASEB J 2009; 23: 1338–1346.
Mizugaki H, Sakakibara-Konishi J, Ikezawa Y, Kikuchi J, Kikuchi E, Oizumi S et al. gamma-Secretase inhibitor enhances antitumour effect of radiation in Notch-expressing lung cancer. Br J Cancer 2012; 106: 1953–1959.
Kunapuli P, Chitta KS, Cowell JK . Suppression of the cell proliferation and invasion phenotypes in glioma cells by the LGI1 gene. Oncogene 2003; 22: 3985–3991.
Liang CC, Park AY, Guan JL . In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protocols 2007; 2: 329–333.
Freeman GH, Halton JH . Note on an exact treatment of contingency, goodness of fit and other problems of significance. Biometrika 1951; 38: 141–149.
Acknowledgements
This work was supported partly by grants of National Basic Research Program of China (2011CB910401), NSFC (31271504) and 2009DFA31940 from The Ministry of Science and Technology, China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, Y., Gong, Y., Hu, D. et al. Nuclear SIPA1 activates integrin β1 promoter and promotes invasion of breast cancer cells. Oncogene 34, 1451–1462 (2015). https://doi.org/10.1038/onc.2014.36
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.36
This article is cited by
-
A novel transcription factor SIPA1: identification and verification in triple-negative breast cancer
Oncogene (2023)
-
Identification of the fatty acid synthase interaction network via iTRAQ-based proteomics indicates the potential molecular mechanisms of liver cancer metastasis
Cancer Cell International (2020)
-
Long non-coding RNA p10247, high expressed in breast cancer (lncRNA-BCHE), is correlated with metastasis
Clinical & Experimental Metastasis (2018)
-
Distinct effects of β1 integrin on cell proliferation and cellular signaling in MDA-MB-231 breast cancer cells
Scientific Reports (2016)
-
Comparative expression study of sipa family members during early Xenopus laevis development
Development Genes and Evolution (2016)