Key Points
-
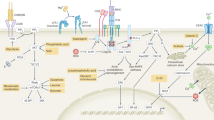
Distinct T cell lineages have unique metabolic needs. CD4+ and CD8+ effector T cells require specific metabolic programmes that differ from those that are required by memory or regulatory T cells.
-
T cell activation, differentiation and function require the upregulation of appropriate metabolic programmes.
-
The MYC, hypoxia-inducible factor 1α, mammalian target of rapamycin and AMP-activated protein kinase pathways regulate cellular metabolic function and direct the outcome of T cell differentiation.
-
Targeting these metabolic pathways represents a novel means of regulating immune responses.
Abstract
Over the past decade, our understanding of T cell activation, differentiation and function has markedly expanded, providing a greater appreciation of the signals and pathways that regulate these processes. It has become clear that evolutionarily conserved pathways that regulate stress responses, metabolism, autophagy and survival have crucial and specific roles in regulating T cell responses. Recent studies suggest that the metabolic pathways involving MYC, hypoxia-inducible factor 1α (HIF1α), AMP-activated protein kinase (AMPK) and mammalian target of rapamycin (mTOR) are activated upon antigen recognition and that they are required for directing the consequences of T cell receptor engagement. The purpose of this Review is to provide an integrated view of the role of these metabolic pathways and of canonical T cell signalling pathways in regulating the outcome of T cell responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gerondakis, S., Fulford, T. S., Messina, N. L. & Grumont, R. J. NF-κB control of T cell development. Nature Immunol. 15, 15–25 (2014).
Srikanth, S. & Gwack, Y. Orai1–NFAT signalling pathway triggered by T cell receptor stimulation. Mol. Cells 35, 182–194 (2013).
Macian, F. et al. Transcriptional mechanisms underlying lymphocyte tolerance. Cell 109, 719–731 (2002).
Schwartz, R. H. Models of T cell anergy: is there a common molecular mechanism? J. Exp. Med. 184, 1–8 (1996).
Fields, P. E., Gajewski, T. F. & Fitch, F. W. Blocked Ras activation in anergic CD4+ T cells. Science 271, 1276–1278 (1996).
Kane, L. P., Andres, P. G., Howland, K. C., Abbas, A. K. & Weiss, A. Akt provides the CD28 costimulatory signal for up-regulation of IL-2 and IFN-γ but not TH2 cytokines. Nature Immunol. 2, 37–44 (2001).
Dolmetsch, R. E., Lewis, R. S., Goodnow, C. C. & Healy, J. I. Differential activation of transcription factors induced by Ca2+ response amplitude and duration. Nature 386, 855–858 (1997).
Chen, L. & Flies, D. B. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nature Rev. Immunol. 13, 227–242 (2013).
Powell, J. D., Lerner, C. G. & Schwartz, R. H. Inhibition of cell cycle progression by rapamycin induces T cell clonal anergy even in the presence of costimulation. J. Immunol. 162, 2775–2784 (1999).
MacIver, N. J., Michalek, R. D. & Rathmell, J. C. Metabolic regulation of T lymphocytes. Annu. Rev. Immunol. 31, 259–283 (2013). This is an outstanding and comprehensive review of metabolism in T cells.
Wang, R. et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 35, 871–882 (2011). This is a seminal paper on the role of MYC in controlling T cell metabolism.
Osthus, R. C. et al. Deregulation of glucose transporter 1 and glycolytic gene expression by c-Myc. J. Biol. Chem. 275, 21797–21800 (2000).
Dang, C. V. MYC, metabolism, cell growth, and tumorigenesis. Cold Spring Harb. Perspect. Med. 3, a014217 (2013).
Gao, P. et al. c-Myc suppression of miR-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature 458, 762–765 (2009).
Li, F. et al. Myc stimulates nuclearly encoded mitochondrial genes and mitochondrial biogenesis. Mol. Cell. Biol. 25, 6225–6234 (2005).
Wang, G. L., Jiang, B. H., Rue, E. A. & Semenza, G. L. Hypoxia-inducible factor 1 is a basic-helix–loop–helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl Acad. Sci. USA 92, 5510–5514 (1995).
Semenza, G. L. Regulation of metabolism by hypoxia-inducible factor 1. Cold Spring Harb. Symp. Quant. Biol. 76, 347–353 (2011).
Iyer, N. V. et al. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α. Genes Dev. 12, 149–162 (1998).
Kim, J. W., Tchernyshyov, I., Semenza, G. L. & Dang, C. V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell. Metab. 3, 177–185 (2006).
Papandreou, I., Cairns, R. A., Fontana, L., Lim, A. L. & Denko, N. C. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell. Metab. 3, 187–197 (2006).
Sun, Q. et al. Mammalian target of rapamycin up-regulation of pyruvate kinase isoenzyme type M2 is critical for aerobic glycolysis and tumor growth. Proc. Natl Acad. Sci. USA 108, 4129–4134 (2011).
Laplante, M. & Sabatini, D. M. mTOR signaling in growth control and disease. Cell 149, 274–293 (2012).
Laplante, M. & Sabatini, D. M. Regulation of mTORC1 and its impact on gene expression at a glance. J. Cell Sci. 126, 1713–1719 (2013). This is an outstanding and comprehensive review of mTOR signalling and function.
Sancak, Y. et al. Ragulator–Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell 141, 290–303 (2010).
Sancak, Y. et al. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science 320, 1496–1501 (2008).
Kim, E., Goraksha-Hicks, P., Li, L., Neufeld, T. P. & Guan, K. L. Regulation of TORC1 by Rag GTPases in nutrient response. Nature Cell Biol. 10, 935–945 (2008).
Zinzalla, V., Stracka, D., Oppliger, W. & Hall, M. N. Activation of mTORC2 by association with the ribosome. Cell 144, 757–768 (2011).
Duvel, K. et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol. Cell 39, 171–183 (2010). This exhaustive work defines the regulation of metabolism by mTOR. Although it is a primary research article, it reads like a review.
Masui, K. et al. mTOR complex 2 controls glycolytic metabolism in glioblastoma through FoxO acetylation and upregulation of c-Myc. Cell. Metab. 18, 726–739 (2013).
Hardie, D. G. AMP-activated protein kinase: an energy sensor that regulates all aspects of cell function. Genes Dev. 25, 1895–1908 (2011).
Yuan, H. X., Xiong, Y. & Guan, K. L. Nutrient sensing, metabolism, and cell growth control. Mol. Cell 49, 379–387 (2013).
Munday, M. R. Regulation of mammalian acetyl-CoA carboxylase. Biochem. Soc. Trans. 30, 1059–1064 (2002).
Inoki, K., Zhu, T. & Guan, K. L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115, 577–590 (2003).
Gwinn, D. M. et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 30, 214–226 (2008).
Kim, J., Kundu, M., Viollet, B. & Guan, K. L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nature Cell Biol. 13, 132–141 (2011).
Faubert, B. et al. Loss of the tumor suppressor LKB1 promotes metabolic reprogramming of cancer cells via HIF-1α. Proc. Natl Acad. Sci. USA 111, 2554–2559 (2014).
Cunningham, J. T. et al. mTOR controls mitochondrial oxidative function through a YY1–PGC-1α transcriptional complex. Nature 450, 736–740 (2007).
Deberardinis, R. J., Lum, J. J. & Thompson, C. B. Phosphatidylinositol 3-kinase-dependent modulation of carnitine palmitoyltransferase 1A expression regulates lipid metabolism during hematopoietic cell growth. J. Biol. Chem. 281, 37372–37380 (2006).
Ben-Sahra, I., Howell, J. J., Asara, J. M. & Manning, B. D. Stimulation of de novo pyrimidine synthesis by growth signaling through mTOR and S6K1. Science 339, 1323–1328 (2013).
Robitaille, A. M. et al. Quantitative phosphoproteomics reveal mTORC1 activates de novo pyrimidine synthesis. Science 339, 1320–1323 (2013).
Bental, M. & Deutsch, C. Metabolic changes in activated T cells: an NMR study of human peripheral blood lymphocytes. Magn. Reson. Med. 29, 317–326 (1993).
Jones, R. G. & Thompson, C. B. Revving the engine: signal transduction fuels T cell activation. Immunity 27, 173–178 (2007). Along with other articles by these authors, this review is paradigm shifting by proposing that metabolism is not dictated by activation but rather that metabolism guides the activation and function of T cells.
Warburg, O. On respiratory impairment in cancer cells. Science 124, 269–270 (1956).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013). This paper demonstrates that aerobic glycolysis is required for T cell effector function but not for proliferation or survival.
Jacobs, S. R. et al. Glucose uptake is limiting in T cell activation and requires CD28-mediated Akt-dependent and independent pathways. J. Immunol. 180, 4476–4486 (2008).
Frauwirth, K. A. et al. The CD28 signaling pathway regulates glucose metabolism. Immunity 16, 769–777 (2002). This is one of the first papers demonstrating a crucial role for immunological signals in regulating cellular metabolism.
Carr, E. L. et al. Glutamine uptake and metabolism are coordinately regulated by ERK/MAPK during T lymphocyte activation. J. Immunol. 185, 1037–1044 (2010).
Kidani, Y. et al. Sterol regulatory element-binding proteins are essential for the metabolic programming of effector T cells and adaptive immunity. Nature Immunol. 14, 489–499 (2013).
Bensinger, S. J. et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111 (2008).
O'Shea, J. J. & Paul, W. E. Mechanisms underlying lineage commitment and plasticity of helper CD4+ T cells. Science 327, 1098–1102 (2010).
Michalek, R. D. et al. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 186, 3299–3303 (2011). This is an early and important study in terms of defining the differences in metabolism between different T cell subsets.
Huang, C. Y., Bredemeyer, A. L., Walker, L. M., Bassing, C. H. & Sleckman, B. P. Dynamic regulation of c-Myc proto-oncogene expression during lymphocyte development revealed by a GFP–c-Myc knock-in mouse. Eur. J. Immunol. 38, 342–349 (2008).
Shi, L. Z. et al. HIF1α-dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and TReg cells. J. Exp. Med. 208, 1367–1376 (2011).
Dang, E. V. et al. Control of TH17/TReg balance by hypoxia-inducible factor 1. Cell 146, 772–784 (2011). References 54 and 55 comprehensively examine the role of HIF1α in regulating T H 17 and T Reg cell differentiation.
Ikejiri, A. et al. Dynamic regulation of TH17 differentiation by oxygen concentrations. Int. Immunol. 24, 137–146 (2012).
Kryczek, I. et al. Human TH17 cells are long-lived effector memory cells. Sci. Transl. Med. 3, 104ra100 (2011).
Muranski, P. et al. Th17 cells are long lived and retain a stem cell-like molecular signature. Immunity 35, 972–985 (2011).
Mazumdar, J. et al. O2 regulates stem cells through Wnt/β-catenin signalling. Nature Cell Biol. 12, 1007–1013 (2010).
Lukashev, D. et al. Cutting edge: hypoxia-inducible factor 1α and its activation-inducible short isoform I.1 negatively regulate functions of CD4+ and CD8+ T lymphocytes. J. Immunol. 177, 4962–4965 (2006).
Georgiev, P. et al. Genetic deletion of the HIF-1α isoform I.1 in T cells enhances antibacterial immunity and improves survival in a murine peritonitis model. Eur. J. Immunol. 43, 655–666 (2013).
Delgoffe, G. M. et al. The mTOR kinase differentially regulates effector and regulatory T cell lineage commitment. Immunity 30, 832–844 (2009). This is the first paper to use genetic tools to demonstrate the crucial role of mTOR in regulating T cell differentiation.
Delgoffe, G. M. et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nature Immunol. 12, 295–303 (2011).
Lee, K. et al. Mammalian target of rapamycin protein complex 2 regulates differentiation of Th1 and Th2 cell subsets via distinct signaling pathways. Immunity 32, 743–753 (2010).
Heikamp, E. B. et al. The AGC kinase SGK1 regulates TH1 and TH2 differentiation downstream of the mTORC2 complex. Nature Immunol. 15, 457–464 (2014).
Yang, K. et al. T cell exit from quiescence and differentiation into Th2 cells depend on Raptor–mTORC1-mediated metabolic reprogramming. Immunity 39, 1043–1056 (2013).
Zeng, H. et al. mTORC1 couples immune signals and metabolic programming to establish TReg cell function. Nature 499, 485–490 (2013). References 63–67 demonstrate the ability of mTORC1 and mTORC2 signalling complexes to selectively regulate CD4+ T cell differentiation.
Kurebayashi, Y. et al. PI3K–Akt–mTORC1–S6K1/2 axis controls Th17 differentiation by regulating Gfi1 expression and nuclear translocation of RORγ. Cell Rep. 1, 360–373 (2012).
MacIver, N. J. et al. The liver kinase B1 is a central regulator of T cell development, activation, and metabolism. J. Immunol. 187, 4187–4198 (2011).
Frauwirth, K. A. & Thompson, C. B. Regulation of T lymphocyte metabolism. J. Immunol. 172, 4661–4665 (2004).
Sukumar, M. et al. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J. Clin. Invest. 123, 4479–4488 (2013).
Cham, C. M. & Gajewski, T. F. Glucose availability regulates IFN-γ production and p70S6 kinase activation in CD8+ effector T cells. J. Immunol. 174, 4670–4677 (2005). References 71 and 72 demonstrate the requirement of glycolysis for T cell effector function by using an inhibitor of glycolysis, 2-deoxy-d-glucose. Reference 71 indicates that inhibition of glycolysis in activated CD8+ T cells promotes the differentiation of long-lived memory cells.
Cham, C. M., Driessens, G., O'Keefe, J. P. & Gajewski, T. F. Glucose deprivation inhibits multiple key gene expression events and effector functions in CD8+ T cells. Eur. J. Immunol. 38, 2438–2450 (2008).
Bianchi, T., Gasser, S., Trumpp, A. & MacDonald, H. R. c-Myc acts downstream of IL-15 in the regulation of memory CD8 T-cell homeostasis. Blood 107, 3992–3999 (2006).
Finlay, D. K. et al. PDK1 regulation of mTOR and hypoxia-inducible factor 1 integrate metabolism and migration of CD8+ T cells. J. Exp. Med. 209, 2441–2453 (2012). This study demonstrates that mTORC1 activity controls glucose uptake and glycolysis by regulating the expression of HIF1α.
Doedens, A. L. et al. Hypoxia-inducible factors enhance the effector responses of CD8+ T cells to persistent antigen. Nature Immunol. 14, 1173–1182 (2013).
Sinclair, L. V. et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nature Immunol. 14, 500–508 (2013). This is an elegant study demonstrating the upregulation of an amino acid transporter upon TCR engagement and reports the functional consequences of deleting this gene.
Tamas, P. et al. Regulation of the energy sensor AMP-activated protein kinase by antigen receptor and Ca2+ in T lymphocytes. J. Exp. Med. 203, 1665–1670 (2006).
Mayer, A., Denanglaire, S., Viollet, B., Leo, O. & Andris, F. AMP-activated protein kinase regulates lymphocyte responses to metabolic stress but is largely dispensable for immune cell development and function. Eur. J. Immunol. 38, 948–956 (2008).
Rolf, J. et al. AMPKα1: A glucose sensor that controls CD8 T-cell memory. Eur. J. Immunol. 43, 889–896 (2013).
Zhang, L. et al. TSC1/2 signaling complex is essential for peripheral naive CD8+ T cell survival and homeostasis in mice. PLoS ONE 7, e30592 (2012).
Yang, K., Neale, G., Green, D. R., He, W. & Chi, H. The tumor suppressor Tsc1 enforces quiescence of naive T cells to promote immune homeostasis and function. Nature Immunol. 12, 888–897 (2011).
O'Brien, T. F. et al. Regulation of T-cell survival and mitochondrial homeostasis by TSC1. Eur. J. Immunol. 41, 3361–3370 (2011).
Pearce, E. L. et al. Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460, 103–107 (2009).
van der Windt, G. J. et al. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity 36, 68–78 (2012). References 84 and 85 are seminal studies by the same group that define the role of metabolism in regulating CD8+ T cell memory.
van der Windt, G. J. et al. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc. Natl Acad. Sci. USA 110, 14336–14341 (2013).
Gubser, P. M. et al. Rapid effector function of memory CD8+ T cells requires an immediate-early glycolytic switch. Nature Immunol. 14, 1064–1072 (2013).
Araki, K. et al. mTOR regulates memory CD8 T-cell differentiation. Nature 460, 108–112 (2009).
Rao, R. R., Li, Q., Odunsi, K. & Shrikant, P. A. The mTOR kinase determines effector versus memory CD8+ T cell fate by regulating the expression of transcription factors T-bet and Eomesodermin. Immunity 32, 67–78 (2010). References 88 and 89 demonstrate the ability of rapamycin to enhance CD8+ T cell memory generation.
Li, Q. et al. A central role for mTOR kinase in homeostatic proliferation induced CD8+ T cell memory and tumor immunity. Immunity 34, 541–553 (2011).
Berezhnoy, A., Castro, I., Levay, A., Malek, T. R. & Gilboa, E. Aptamer-targeted inhibition of mTOR in T cells enhances antitumor immunity. J. Clin. Invest. 124, 188–197 (2014).
He, S. et al. Characterization of the metabolic phenotype of rapamycin-treated CD8+ T cells with augmented ability to generate long-lasting memory cells. PLoS ONE 6, e20107 (2011).
Ben-Shoshan, J., Maysel-Auslender, S., Mor, A., Keren, G. & George, J. Hypoxia controls CD4+CD25+ regulatory T-cell homeostasis via hypoxia-inducible factor-1α. Eur. J. Immunol. 38, 2412–2418 (2008).
Clambey, E. T. et al. Hypoxia-inducible factor-1α-dependent induction of FoxP3 drives regulatory T-cell abundance and function during inflammatory hypoxia of the mucosa. Proc. Natl Acad. Sci. USA 109, E2784–2793 (2012).
Battaglia, M., Stabilini, A. & Roncarolo, M. G. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood 105, 4743–4748 (2005).
Kang, J., Huddleston, S. J., Fraser, J. M. & Khoruts, A. De novo induction of antigen-specific CD4+CD25+Foxp3+ regulatory T cells in vivo following systemic antigen administration accompanied by blockade of mTOR. J. Leukoc. Biol. 83, 1230–1239 (2008).
Kopf, H., de la Rosa, G. M., Howard, O. M. & Chen, X. Rapamycin inhibits differentiation of Th17 cells and promotes generation of FoxP3+ T regulatory cells. Int. Immunopharmacol. 7, 1819–1824 (2007).
Liu, G., Yang, K., Burns, S., Shrestha, S. & Chi, H. The S1P1–mTOR axis directs the reciprocal differentiation of TH1 and TReg cells. Nature Immunol. 11, 1047–1056 (2010).
Haxhinasto, S., Mathis, D. & Benoist, C. The AKT–mTOR axis regulates de novo differentiation of CD4+Foxp3+ cells. J. Exp. Med. 205, 565–574 (2008).
Procaccini, C. et al. An oscillatory switch in mTOR kinase activity sets regulatory T cell responsiveness. Immunity 33, 929–941 (2010).
Powell, J. D., Heikamp, E. B., Pollizzi, K. N. & Waickman, A. T. A modified model of T-cell differentiation based on mTOR activity and metabolism. Cold Spring Harb. Symp. Quant. Biol. http://dx.doi.org/10.1101/sqb.2013.78.020214 (2013).
Powell, J. D. & Zheng, Y. Dissecting the mechanism of T-cell anergy with immunophilin ligands. Curr. Opin. Investig. Drugs 7, 1002–1007 (2006).
Bopp, T. et al. NFATc2 and NFATc3 transcription factors play a crucial role in suppression of CD4+ T lymphocytes by CD4+ CD25+ regulatory T cells. J. Exp. Med. 201, 181–187 (2005).
Zhang, H. et al. Digoxin and other cardiac glycosides inhibit HIF-1α synthesis and block tumor growth. Proc. Natl Acad. Sci. USA 105, 19579–19586 (2008).
Huh, J. R. et al. Digoxin and its derivatives suppress TH17 cell differentiation by antagonizing RORγt activity. Nature 472, 486–490 (2011). This study uses digoxin, which is a small-molecule compound that specifically inhibits the transcription of the gene that encodes RORγt. Digoxin can inhibit both mouse and human T H 17 cell differentiation, which suggests that it may be a novel therapeutic agent for autoimmune diseases.
Fujita-Sato, S. et al. Structural basis of digoxin that antagonizes RORγt receptor activity and suppresses Th17 cell differentiation and interleukin (IL)-17 production. J. Biol. Chem. 286, 31409–31417 (2011).
Xiao, S. et al. Small-molecule RORγt antagonists inhibit T helper 17 cell transcriptional network by divergent mechanisms. Immunity 40, 477–489 (2014).
Bandukwala, H. S. et al. Selective inhibition of CD4+ T-cell cytokine production and autoimmunity by BET protein and c-Myc inhibitors. Proc. Natl Acad. Sci. USA 109, 14532–14537 (2012).
Bai, A. et al. Novel anti-inflammatory action of 5-aminoimidazole-4-carboxamide ribonucleoside with protective effect in dextran sulfate sodium-induced acute and chronic colitis. J. Pharmacol. Exp. Ther. 333, 717–725 (2010).
Nath, N. et al. 5-aminoimidazole-4-carboxamide ribonucleoside: a novel immunomodulator with therapeutic efficacy in experimental autoimmune encephalomyelitis. J. Immunol. 175, 566–574 (2005).
Nath, N. et al. Metformin attenuated the autoimmune disease of the central nervous system in animal models of multiple sclerosis. J. Immunol. 182, 8005–8014 (2009).
Touzot, M., Soulillou, J. P. & Dantal, J. Mechanistic target of rapamycin inhibitors in solid organ transplantation: from benchside to clinical use. Curr. Opin. Organ. Transplant 17, 626–633 (2012).
Waickman, A. T. & Powell, J. D. mTOR, metabolism, and the regulation of T-cell differentiation and function. Immunol. Rev. 249, 43–58 (2012).
Sarbassov, D. D. et al. Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol. Cell 22, 159–168 (2006).
Turner, A. P. et al. Sirolimus enhances the magnitude and quality of viral-specific CD8+ T-cell responses to vaccinia virus vaccination in rhesus macaques. Am. J. Transplant 11, 613–618 (2011).
Thoreen, C. C. et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J. Biol. Chem. 284, 8023–8032 (2009).
Feldman, M. E. et al. Active-site inhibitors of mTOR target rapamycin-resistant outputs of mTORC1 and mTORC2. PLoS Biol. 7, e38 (2009).
Gao, W. et al. Contrasting effects of cyclosporine and rapamycin in de novo generation of alloantigen-specific regulatory T cells. Am. J. Transplant 7, 1722–1732 (2007).
Levitsky, J. et al. Systemic immunoregulatory and proteogenomic effects of tacrolimus to sirolimus conversion in liver transplant recipients. Hepatology 57, 239–248 (2013).
Hsieh, M. M. et al. Allogeneic hematopoietic stem-cell transplantation for sickle cell disease. N. Engl. J. Med. 361, 2309–2317 (2009).
Chen, J. Q. & Russo, J. Dysregulation of glucose transport, glycolysis, TCA cycle and glutaminolysis by oncogenes and tumor suppressors in cancer cells. Biochim. Biophys. Acta 1826, 370–384 (2012).
Schwartz, R. H. Costimulation of T lymphocytes: the role of CD28, CTLA-4, and B7/BB1 in interleukin-2 production and immunotherapy. Cell 71, 1065–1068 (1992).
Loh, C. et al. Calcineurin binds the transcription factor NFAT1 and reversibly regulates its activity. J. Biol. Chem. 271, 10884–10891 (1996).
Chen, F. E. & Ghosh, G. Regulation of DNA binding by Rel/NF-κB transcription factors: structural views. Oncogene 18, 6845–6852 (1999).
Jenkins, M. K. & Schwartz, R. H. Antigen presentation by chemically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo. J. Exp. Med. 165, 302–319 (1987).
Acknowledgements
The authors thank F. Pan for critical review of this manuscript. The authors' work is supported by the US National Institutes of Health (grant R01AI077610-01A2). The authors apologize to those colleagues whose work has not been cited owing to space constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Glucose transporter type 1
-
(GLUT1). A unidirectional transporter that facilitates the transport of glucose across the plasma membrane.
- Lactate dehydrogenase A
-
(LDHA). An enzyme that catalyses the conversion of pyruvate to lactate.
- Hexokinase 2
-
An enzyme that initiates the first reaction of glycolysis by phosphorylating glucose to produce glucose-6-phosphate.
- Pyruvate dehydrogenase kinase 1
-
(PDK1). An enzyme that phosphorylates and inactivates pyruvate dehydrogenase, thereby inhibiting the catalysis of pyruvate to acetyl-CoA and preventing the initiation of the tricarboxylic acid cycle.
- Phosphofructokinase 1
-
A rate-limiting enzyme of glycolysis that requires ATP to convert fructose-6-phosphate into fructose-1,6-bisphosphate.
- Carnitine palmitoyltransferase 1A
-
(CPT1A). A rate-limiting mitochondrial enzyme that is necessary for fatty acid oxidation. CPT1A catalyses the transfer of the acyl group of long-chain fatty acids to acylcarnitine, which allows for its transport from the cytosol to the mitochondria.
- Autophagy
-
An evolutionarily conserved process in which acidic double-membrane-bound vacuoles sequester intracellular contents (such as damaged organelles and macromolecules) and target them for degradation through fusion with secondary lysosomes.
- α-ketoglutarate
-
A key intermediate of the tricarboxylic acid cycle that can be derived from glutaminolysis.
Rights and permissions
About this article
Cite this article
Pollizzi, K., Powell, J. Integrating canonical and metabolic signalling programmes in the regulation of T cell responses. Nat Rev Immunol 14, 435–446 (2014). https://doi.org/10.1038/nri3701
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3701
This article is cited by
-
Fatty acid metabolism of immune cells: a new target of tumour immunotherapy
Cell Death Discovery (2024)
-
Management of Atopy with Dupilumab and Omalizumab in CADINS Disease
Journal of Clinical Immunology (2024)
-
Exploiting autophagy balance in T and NK cells as a new strategy to implement adoptive cell therapies
Molecular Cancer (2023)
-
Reductive carboxylation epigenetically instructs T cell differentiation
Nature (2023)
-
Role of T Cells in the Pathogenesis of Rheumatoid Arthritis: Focus on Immunometabolism Dysfunctions
Inflammation (2023)