Key Points
-
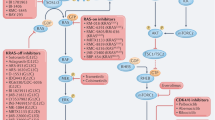
The oncoprotein BRAF is frequently activated due to genetic alterations in tumours promoting deregulation of RAF–MEK–ERK signalling. Targeting BRAF with inhibitors is a validated therapeutic strategy for a substantial proportion of cancer patients.
-
RAF inhibitors alone or in combination with MEK inhibitors have elicited dramatic responses and prolonged the survival of patients with melanoma whose tumours harbour mutationally activated BRAF-V600. However, their effectiveness is limited by the development of drug resistance, frequently by mechanisms that promote reactivation of RAF–ERK signalling in the presence of the drug.
-
In BRAF-V600 tumours other than melanoma, or in tumours carrying BRAF alterations other than the BRAF-V600 mutation, current clinical RAF inhibitors have shown modest effectiveness.
-
RAF inhibitors have unique biochemical properties that account for their wide therapeutic window, on-target toxicities and major mechanisms of resistance.
-
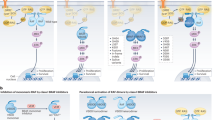
RAF dimerization is a common mechanism of both intrinsic and acquired resistance to current clinical RAF inhibitors vemurafenib and dabrafenib, which stabilize the αC-helix of RAF kinase in the OUT position. These compounds effectively inhibit monomeric mutant BRAF but fail to inhibit dimeric RAF.
-
Structurally, inhibitor resistance due to RAF dimerization is the result of negative allostery for inhibitor binding to the second protomer of the RAF dimer, once the first is bound to an inhibitor.
-
Next-generation RAF inhibitors that stabilize the αC-helix of RAF kinase in the active (IN) position will inhibit RAF monomers and dimers, but they are predicted to have a narrow therapeutic window due to inhibition of wild-type BRAF in normal cells. Thus, combinatorial approaches with current clinical inhibitors may be beneficial.
-
Paradoxical pathway activation (allosteric priming) is a critical adverse event observed with most RAF inhibitors in the presence of RAS. Its effect on downstream signalling is currently ameliorated with the combined use of MEK inhibitors.
-
Several structurally diverse, next-generation RAF inhibitors are under preclinical or clinical development and may be effective in BRAF-mutant tumours that are resistant to current clinical RAF inhibitors.
Abstract
The discovery that a subset of human tumours is dependent on mutationally deregulated BRAF kinase intensified the development of RAF inhibitors to be used as potential therapeutics. The US Food and Drug Administration (FDA)-approved second-generation RAF inhibitors vemurafenib and dabrafenib have elicited remarkable responses and improved survival of patients with BRAF-V600E/K melanoma, but their effectiveness is limited by resistance. Beyond melanoma, current clinical RAF inhibitors show modest efficacy when used for colorectal and thyroid BRAF-V600E tumours or for tumours harbouring BRAF alterations other than the V600 mutation. Accumulated experimental and clinical evidence indicates that the complex biochemical mechanisms of RAF kinase signalling account both for the effectiveness of RAF inhibitors and for the various mechanisms of tumour resistance to them. Recently, a number of next-generation RAF inhibitors, with diverse structural and biochemical properties, have entered preclinical and clinical development. In this Review, we discuss the current understanding of RAF kinase regulation, mechanisms of inhibitor action and related clinical resistance to these drugs. The recent elucidation of critical structural and biochemical aspects of RAF inhibitor action, combined with the availability of a number of structurally diverse RAF inhibitors currently in preclinical and clinical development, will enable the design of more effective RAF inhibitors and RAF-inhibitor-based therapeutic strategies, tailored to different clinical contexts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lavoie, H. & Therrien, M. Regulation of RAF protein kinases in ERK signalling. Nat. Rev. Mol. Cell Biol. 16, 281–298 (2015).
Dhillon, A. S., Hagan, S., Rath, O. & Kolch, W. MAP kinase signalling pathways in cancer. Oncogene 26, 3279–3290 (2007).
Wellbrock, C., Karasarides, M. & Marais, R. The RAF proteins take centre stage. Nat. Rev. Mol. Cell Biol. 5, 875–885 (2004).
Matallanas, D. et al. Raf family kinases: old dogs have learned new tricks. Genes Cancer 2, 232–260 (2011).
Roberts, P. J. & Der, C. J. Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 26, 3291–3310 (2007).
Young, A. et al. Ras signaling and therapies. Adv. Cancer Res. 102, 1–17 (2009).
Baines, A. T., Xu, D. & Der, C. J. Inhibition of Ras for cancer treatment: the search continues. Future Med. Chem. 3, 1787–1808 (2011).
Caunt, C. J., Sale, M. J., Smith, P. D. & Cook, S. J. MEK1 and MEK2 inhibitors and cancer therapy: the long and winding road. Nat. Rev. Cancer 15, 577–592 (2015).
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002).
Wan, P. T. et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell 116, 855–867 (2004). This paper provides the first crystal structure of BRAF-V600E bound to inhibitor, along with biochemical insight into the mechanism of BRAF activation by mutations.
Garnett, M. J., Rana, S., Paterson, H., Barford, D. & Marais, R. Wild-type and mutant B-RAF activate C-RAF through distinct mechanisms involving heterodimerization. Mol. Cell 20, 963–969 (2005).
Foster, S. A. et al. Activation mechanism of oncogenic deletion mutations in BRAF, EGFR, and HER2. Cancer Cell 29, 477–493 (2016).
Holderfield, M., Deuker, M. M., McCormick, F. & McMahon, M. Targeting RAF kinases for cancer therapy: BRAF-mutated melanoma and beyond. Nat. Rev. Cancer 14, 455–467 (2014).
Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 161, 1681–1696 (2015).
Kimura, E. T. et al. High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res. 63, 1454–1457 (2003).
Rajagopalan, H. et al. Tumorigenesis: RAF/RAS oncogenes and mismatch-repair status. Nature 418, 934 (2002).
Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 511, 543–550 (2014).
Tiacci, E. et al. BRAF mutations in hairy-cell leukemia. N. Engl. J. Med. 364, 2305–2315 (2011).
Badalian-Very, G. et al. Recurrent BRAF mutations in Langerhans cell histiocytosis. Blood 116, 1919–1923 (2010).
Solit, D. B. et al. BRAF mutation predicts sensitivity to MEK inhibition. Nature 439, 358–362 (2006). This paper provides preclinical evidence supporting the targeting of BRAF–ERK signalling in tumours harbouring the mutation BRAF-V600E.
Hauschild, A. et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380, 358–365 (2012).
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011).
Bollag, G. et al. Clinical efficacy of a RAF inhibitor needs broad target blockade in BRAF-mutant melanoma. Nature 467, 596–599 (2010). This paper reports on the discovery and early clinical results of the first FDA-approved RAF inhibitor, vemurafenib.
Long, G. V. et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N. Engl. J. Med. 371, 1877–1888 (2014).
Larkin, J. et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N. Engl. J. Med. 371, 1867–1876 (2014).
Carlino, M. S. et al. Antiproliferative effects of continued mitogen-activated protein kinase pathway inhibition following acquired resistance to BRAF and/or MEK inhibition in melanoma. Mol. Cancer Ther. 12, 1332–1342 (2013).
Moriceau, G. et al. Tunable-combinatorial mechanisms of acquired resistance limit the efficacy of BRAF/MEK cotargeting but result in melanoma drug addiction. Cancer Cell 27, 240–256 (2015).
Villanueva, J. et al. Concurrent MEK2 mutation and BRAF amplification confer resistance to BRAF and MEK inhibitors in melanoma. Cell Rep. 4, 1090–1099 (2013).
Yang, H. et al. RG7204 (PLX4032), a selective BRAFV600E inhibitor, displays potent antitumor activity in preclinical melanoma models. Cancer Res. 70, 5518–5527 (2010).
Karoulia, Z. et al. An integrated model of RAF inhibitor action predicts inhibitor activity against oncogenic BRAF signaling. Cancer Cell 30, 485–498 (2016). This study provides a unified mechanistic model of RAF inhibitor action and a roadmap for the clinical use of next-generation RAF inhibitors based on their structural properties.
Yao, Z. et al. BRAF mutants evade ERK-dependent feedback by different mechanisms that determine their sensitivity to pharmacologic inhibition. Cancer Cell 28, 370–383 (2015).
Gentilcore, G. et al. Effect of dabrafenib on melanoma cell lines harbouring the BRAF(V600D/R) mutations. BMC Cancer 13, 17 (2013).
Flaherty, K. T. et al. Inhibition of mutated, activated BRAF in metastatic melanoma. N. Engl. J. Med. 363, 809–819 (2010).
McArthur, G. A. et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a phase 3, randomised, open-label study. Lancet Oncol. 15, 323–332 (2014).
Klein, O. et al. BRAF inhibitor activity in V600R metastatic melanoma. Eur. J. Cancer 49, 1073–1079 (2013).
Dahlman, K. B. et al. BRAF(L597) mutations in melanoma are associated with sensitivity to MEK inhibitors. Cancer Discov. 2, 791–797 (2012).
Prahallad, A. et al. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature 483, 100–103 (2012).
Corcoran, R. B. et al. EGFR-mediated re-activation of MAPK signaling contributes to insensitivity of BRAF mutant colorectal cancers to RAF inhibition with vemurafenib. Cancer Discov. 2, 227–235 (2012). This paper shows that insensitivity of colorectal BRAF-V600E tumours to RAF inhibitors is caused by relief of negative feedback and upstream activation of RTK signalling, most commonly through EGFR.
Montero-Conde, C. et al. Relief of feedback inhibition of HER3 transcription by RAF and MEK inhibitors attenuates their antitumor effects in BRAF-mutant thyroid carcinomas. Cancer Discov. 3, 520–533 (2013). This paper shows that insensitivity of thyroid BRAF-V600E tumours to RAF inhibitors is caused by relief of negative feedback and upstream activation of RTK signalling, most commonly through HER3.
Warne, P. H., Viciana, P. R. & Downward, J. Direct interaction of Ras and the amino-terminal region of Raf-1 in vitro. Nature 364, 352–355 (1993).
Cutler, R. E. Jr, Stephens, R. M., Saracino, M. R. & Morrison, D. K. Autoregulation of the Raf-1 serine/threonine kinase. Proc. Natl Acad. Sci. USA 95, 9214–9219 (1998).
Vojtek, A. B., Hollenberg, S. M. & Cooper, J. A. Mammalian Ras interacts directly with the serine/threonine kinase Raf. Cell 74, 205–214 (1993).
Moodie, S. A., Willumsen, B. M., Weber, M. J. & Wolfman, A. Complexes of Ras. GTP with Raf-1 and mitogen-activated protein kinase kinase. Science 260, 1658–1661 (1993).
Weber, C. K., Slupsky, J. R., Kalmes, H. A. & Rapp, U. R. Active Ras induces heterodimerization of cRaf and BRaf. Cancer Res. 61, 3595–3598 (2001).
Avruch, J. et al. Ras activation of the Raf kinase: tyrosine kinase recruitment of the MAP kinase cascade. Recent Prog. Horm. Res. 56, 127–155 (2001).
Kornev, A. P. & Taylor, S. S. Dynamics-driven allostery in protein kinases. Trends Biochem. Sci. 40, 628–647 (2015).
Hatzivassiliou, G. et al. RAF inhibitors prime wild-type RAF to activate the MAPK pathway and enhance growth. Nature 464, 431–435 (2010). This study provides insight into the unique biochemical mechanism of RAF inhibitor action.
Tsai, J. et al. Discovery of a selective inhibitor of oncogenic B-Raf kinase with potent antimelanoma activity. Proc. Natl Acad. Sci. USA 105, 3041–3046 (2008).
Lavoie, H. et al. Inhibitors that stabilize a closed RAF kinase domain conformation induce dimerization. Nat. Chem. Biol. 9, 428–436 (2013).
Rajakulendran, T., Sahmi, M., Lefrancois, M., Sicheri, F. & Therrien, M. A dimerization-dependent mechanism drives RAF catalytic activation. Nature 461, 542–545 (2009). This paper provides mechanistic insight into the mechanism of RAF activation by dimerization.
Freeman, A. K., Ritt, D. A. & Morrison, D. K. Effects of Raf dimerization and its inhibition on normal and disease-associated Raf signaling. Mol. Cell 49, 751–758 (2013).
Poulikakos, P. I. et al. RAF inhibitor resistance is mediated by dimerization of aberrantly spliced BRAF(V600E). Nature 480, 387–390 (2011). This paper provides information identifying the expression of splice variants of BRAF-V600E, which cause clinical resistance to RAF inhibitors by enhanced homodimerization, and confirming that RAF dimerization is a common mechanism of resistance to RAF inhibitors.
Roring, M. et al. Distinct requirement for an intact dimer interface in wild-type, V600E and kinase-dead B-Raf signalling. EMBO J. 31, 2629–2647 (2012).
Thevakumaran, N. et al. Crystal structure of a BRAF kinase domain monomer explains basis for allosteric regulation. Nat. Struct. Mol. Biol. 22, 37–43 (2015).
Haling, J. R. et al. Structure of the BRAF-MEK complex reveals a kinase activity independent role for BRAF in MAPK signaling. Cancer Cell 26, 402–413 (2014).
Poulikakos, P. I., Zhang, C., Bollag, G., Shokat, K. M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 464, 427–430 (2010). This paper provides insight into the unique biochemical mechanism of RAF inhibitor action.
Heidorn, S. J. et al. Kinase-dead BRAF and oncogenic RAS cooperate to drive tumor progression through CRAF. Cell 140, 209–221 (2010). This paper shows that mutant BRAF with impaired kinase activity can cooperate with active RAS to promote tumour progression.
Holderfield, M. et al. RAF inhibitors activate the MAPK pathway by relieving inhibitory autophosphorylation. Cancer Cell 23, 594–602 (2013).
Halaban, R. et al. PLX4032, a selective BRAF(V600E) kinase inhibitor, activates the ERK pathway and enhances cell migration and proliferation of BRAF melanoma cells. Pigment Cell Melanoma Res. 23, 190–200 (2010).
Joseph, E. W. et al. The RAF inhibitor PLX4032 inhibits ERK signaling and tumor cell proliferation in a V600E BRAF-selective manner. Proc. Natl Acad. Sci. USA 107, 14903–14908 (2010).
Holderfield, M., Nagel, T. E. & Stuart, D. D. Mechanism and consequences of RAF kinase activation by small-molecule inhibitors. Br. J. Cancer 111, 640–645 (2014).
Poulikakos, P. I. & Rosen, N. Mutant BRAF melanomas — dependence and resistance. Cancer Cell 19, 11–15 (2011).
Samatar, A. A. & Poulikakos, P. I. Targeting RAS-ERK signalling in cancer: promises and challenges. Nat. Rev. Drug Discov. 13, 928–942 (2014).
Zhang, J., Yang, P. L. & Gray, N. S. Targeting cancer with small molecule kinase inhibitors. Nat. Rev. Cancer 9, 28–39 (2009).
Hu, J. et al. Allosteric activation of functionally asymmetric RAF kinase dimers. Cell 154, 1036–1046 (2013).
Girotti, M. R. et al. Paradox-breaking RAF inhibitors that also target SRC are effective in drug-resistant BRAF mutant melanoma. Cancer Cell 27, 85–96 (2015).
Nakamura, A. et al. Antitumor activity of the selective pan-RAF inhibitor TAK-632 in BRAF inhibitor-resistant melanoma. Cancer Res. 73, 7043–7055 (2013).
Peng, S. B. et al. Inhibition of RAF isoforms and active dimers by LY3009120 leads to anti-tumor activities in RAS or BRAF mutant cancers. Cancer Cell 28, 384–398 (2015).
Okuzumi, T. et al. Inhibitor hijacking of Akt activation. Nat. Chem. Biol. 5, 484–493 (2009).
Cameron, A. J., Escribano, C., Saurin, A. T., Kostelecky, B. & Parker, P. J. PKC maturation is promoted by nucleotide pocket occupation independently of intrinsic kinase activity. Nat. Struct. Mol. Biol. 16, 624–630 (2009).
Pearce, L. R. et al. Characterization of PF-4708671, a novel and highly specific inhibitor of p70 ribosomal S6 kinase (S6K1). Biochem. J. 431, 245–255 (2010).
Andraos, R. et al. Modulation of activation-loop phosphorylation by JAK inhibitors is binding mode dependent. Cancer Discov. 2, 512–523 (2012).
Zhang, C. et al. RAF inhibitors that evade paradoxical MAPK pathway activation. Nature 526, 583–586 (2015).
Su, F. et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N. Engl. J. Med. 366, 207–215 (2012).
Nazarian, R. et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature 468, 973–977 (2010). This paper provides evidence identifying RAS mutation and RTK upregulation as major mechanisms of acquired clinical resistance of BRAF-V600E melanomas to RAF inhibitors.
Diedrich, B. et al. Discrete cytosolic macromolecular BRAF complexes exhibit distinct activities and composition. EMBO J. 36, 646–663 (2017).
Ross, J. S. et al. The distribution of BRAF gene fusions in solid tumors and response to targeted therapy. Int. J. Cancer 138, 881–890 (2016).
Jones, D. T. et al. Tandem duplication producing a novel oncogenic BRAF fusion gene defines the majority of pilocytic astrocytomas. Cancer Res. 68, 8673–8677 (2008).
Palanisamy, N. et al. Rearrangements of the RAF kinase pathway in prostate cancer, gastric cancer and melanoma. Nat. Med. 16, 793–798 (2010).
Chen, S. H. et al. Oncogenic BRAF deletions that function as homodimers and are sensitive to inhibition by RAF dimer inhibitor LY3009120. Cancer Discov. 6, 300–315 (2016).
Zheng, G. et al. Clinical detection and categorization of uncommon and concomitant mutations involving BRAF. BMC Cancer 15, 779 (2015).
Nieto, P. et al. A Braf kinase-inactive mutant induces lung adenocarcinoma. Nature 548, 239–243 (2017).
Yao, Z. et al. Tumours with class 3 BRAF mutants are sensitive to the inhibition of activated RAS. Nature 548, 234–238 (2017).
Shi, H. et al. Melanoma whole-exome sequencing identifies (V600E)B-RAF amplification-mediated acquired B-RAF inhibitor resistance. Nat. Commun. 3, 724 (2012).
Villanueva, J. et al. Acquired resistance to BRAF inhibitors mediated by a RAF kinase switch in melanoma can be overcome by cotargeting MEK and IGF-1R/PI3K. Cancer Cell 18, 683–695 (2010).
Shi, H. et al. Acquired resistance and clonal evolution in melanoma during BRAF inhibitor therapy. Cancer Discov. 4, 80–93 (2014).
Emery, C. M. et al. BRAF-inhibitor associated MEK mutations increase RAF-dependent and-independent enzymatic activity. Mol. Cancer Res. http://dx.doi.org/10.1158/1541-7786.MCR-17-0211 (2017).
Wagle, N. et al. Dissecting therapeutic resistance to RAF inhibition in melanoma by tumor genomic profiling. J. Clin. Oncol. 29, 3085–3096 (2011).
Lito, P. et al. Relief of profound feedback inhibition of mitogenic signaling by RAF inhibitors attenuates their activity in BRAFV600E melanomas. Cancer Cell 22, 668–682 (2012).
Obenauf, A. C. et al. Therapy-induced tumour secretomes promote resistance and tumour progression. Nature 520, 368–372 (2015).
Straussman, R. et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 487, 500–504 (2012). This paper shows that adaptive response to RAF inhibitors is promoted by growth factors in the tumour microenvironment and upstream RTK activation.
Wilson, T. R. et al. Widespread potential for growth-factor-driven resistance to anticancer kinase inhibitors. Nature 487, 505–509 (2012).
Kopetz, S. et al. Phase II pilot study of vemurafenib in patients with metastatic BRAF-mutated colorectal cancer. J. Clin. Oncol. 33, 4032–4038 (2015).
Hyman, D. M. et al. Vemurafenib in multiple nonmelanoma cancers with BRAF V600 mutations. N. Engl. J. Med. 373, 726–736 (2015).
Hall-Jackson, C. A. et al. Paradoxical activation of Raf by a novel Raf inhibitor. Chem. Biol. 6, 559–568 (1999).
Lyons, J. F., Wilhelm, S., Hibner, B. & Bollag, G. Discovery of a novel Raf kinase inhibitor. Endocr. Relat. Cancer 8, 219–225 (2001).
Escudier, B. et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 356, 125–134 (2007).
Llovet, J. M. et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 359, 378–390 (2008).
King, A. J. et al. Demonstration of a genetic therapeutic index for tumors expressing oncogenic BRAF by the kinase inhibitor SB-590885. Cancer Res. 66, 11100–11105 (2006).
Hoeflich, K. P. et al. Antitumor efficacy of the novel RAF inhibitor GDC-0879 is predicted by BRAFV600E mutational status and sustained extracellular signal-regulated kinase/mitogen-activated protein kinase pathway suppression. Cancer Res. 69, 3042–3051 (2009).
Shelton, J. G. et al. Differential effects of kinase cascade inhibitors on neoplastic and cytokine-mediated cell proliferation. Leukemia 17, 1765–1782 (2003).
Chin, P. C. et al. The c-Raf inhibitor GW5074 provides neuroprotection in vitro and in an animal model of neurodegeneration through a MEK-ERK and Akt-independent mechanism. J. Neurochem. 90, 595–608 (2004).
Falchook, G. S. et al. Dabrafenib in patients with melanoma, untreated brain metastases, and other solid tumours: a phase 1 dose-escalation trial. Lancet 379, 1893–1901 (2012).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01909453(2017).
Okaniwa, M. et al. Discovery of a selective kinase inhibitor (TAK-632) targeting pan-RAF inhibition: design, synthesis, and biological evaluation of C-7-substituted 1,3-benzothiazole derivatives. J. Med. Chem. 56, 6478–6494 (2013).
Sun, Y. et al. A brain-penetrant RAF dimer antagonist for the noncanonical BRAF oncoprotein of pediatric low-grade astrocytomas. Neuro Oncol. 19, 774–785 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02437227(2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01425008 (2016).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02723006(2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02327169 (2017).
Henry, J. R. et al. Discovery of 1-(3,3-dimethylbutyl)-3-(2-fluoro-4-methyl-5-(7-methyl-2-(methylamino)pyrido[2,3- d]pyrimidin-6-yl)phenyl)urea (LY3009120) as a pan-RAF inhibitor with minimal paradoxical activation and activity against BRAF or RAS mutant tumor cells. J. Med. Chem. 58, 4165–4179 (2015).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02014116 (2017).
Tang, Z. et al. BGB-283, a novel RAF kinase and EGFR inhibitor, displays potent antitumor activity in BRAF-mutated colorectal cancers. Mol. Cancer Ther. 14, 2187–2197 (2015).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02610361 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02607813 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02428712 (2017).
Sievert, A. J. et al. Paradoxical activation and RAF inhibitor resistance of BRAF protein kinase fusions characterizing pediatric astrocytomas. Proc. Natl Acad. Sci. USA 110, 5957–5962 (2013).
Le, K., Blomain, E. S., Rodeck, U. & Aplin, A. E. Selective RAF inhibitor impairs ERK½ phosphorylation and growth in mutant NRAS, vemurafenib-resistant melanoma cells. Pigment Cell Melanoma Res. 26, 509–517 (2013).
Basile, K. J., Le, K., Hartsough, E. J. & Aplin, A. E. Inhibition of mutant BRAF splice variant signaling by next-generation, selective RAF inhibitors. Pigment Cell Melanoma Res. 27, 479–484 (2014).
Waizenegger, I. C. et al. A novel RAF kinase inhibitor with DFG-out-binding mode: high efficacy in BRAF-mutant tumor xenograft models in the absence of normal tissue hyperproliferation. Mol. Cancer Ther. 15, 354–365 (2016).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03118817 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02405065 (2017).
James, J. et al. CEP-32496: a novel orally active BRAF(V600E) inhibitor with selective cellular and in vivo antitumor activity. Mol. Cancer Ther. 11, 930–941 (2012).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01877811 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02928224 (2017).
Long, G. V. et al. Increased MAPK reactivation in early resistance to dabrafenib/trametinib combination therapy of BRAF-mutant metastatic melanoma. Nat. Commun. 5, 5694 (2014).
Carlino, M. S. et al. Differential activity of MEK and ERK inhibitors in BRAF inhibitor resistant melanoma. Mol. Oncol. 8, 544–554 (2014).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02872259 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01719380 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01543698 (2017).
US National Library of Medicine.ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02159066 (2017).
Topalian, S. L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Robert, C. et al. Pembrolizumab versus ipilimumab in advanced melanoma. N. Engl. J. Med. 372, 2521–2532 (2015).
Hoos, A. Development of immuno-oncology drugs — from CTLA4 to PD1 to the next generations. Nat. Rev. Drug Discov. 15, 235–247 (2016).
Hugo, W. et al. Genomic and transcriptomic features of response to anti-PD-1 therapy in metastatic melanoma. Cell 165, 35–44 (2016).
Mandalà, M. & Massi, D. Immunotolerance as a mechanism of resistance to targeted therapies in melanoma. Handb. Exp. Pharmacol. http://dx.doi.org/10.1007/164_2017_5 (2017).
Rushworth, L. K., Hindley, A. D., O'Neill, E. & Kolch, W. Regulation and role of Raf-1/B-Raf heterodimerization. Mol. Cell. Biol. 26, 2262–2272 (2006).
Acknowledgements
The authors are grateful to Bogos Agianian for generating the movies. E.G. is supported by NIH grant R01CA178394 and awards from the Melanoma Research Alliance, the Pershing Square Sohn Cancer Research Alliance, the Irma T. Hirschl Trust, the Gabrielle's Angels Foundation for Cancer Research, and the Sidney Kimmel Foundation for Cancer Research. P.I.P. is supported by NIH grant R01CA204314, the Sidney Kimmel Foundation for Cancer Research, the Melanoma Research Foundation, the Dermatology Foundation and the Melanoma Research Alliance.
Author information
Authors and Affiliations
Contributions
Z.K., E.G. and P.I.P. contributed equally to researching the literature, to writing the article and to reviewing and editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary S1 (movie)
αC-OUT RAF inhibitors fail to inhibit dimeric RAF (9,713 KB) Vemurafenib binds the first protomer within the RAF dimer and stabilizes the αC-helix in the OUT position. However, the same movement is not sterically possible in the second protomer without disrupting the dimer. The αC-helix-IN conformation of the second protomer prevents binding of a second vemurafenib, leading to persistence of cellular RAF activity. (MOV 9713 kb)
Supplementary S2 (movie)
αC-IN RAF inhibitors potently inhibit dimeric RAF (13,593 KB) In contrast to vemurafenib (and other αC-OUT RAF inhibitors), RAF inhibitors that stabilize the αC-helix in the IN position, such as AZ628, occupy and inhibit both protomers within the dimer, thus effectively suppressing cellular RAF activity. (MOV 13593 kb)
Glossary
- Langerhans cell histiocytosis
-
(LCH). A myeloid neoplasia characterized by inflammatory lesions containing pathological dendritic cells, frequently harbouring the BRAF-V600E mutation.
- Protomer
-
The structural unit of an oligomeric protein; in the case of wild-type BRAF, a functional BRAF dimer is composed of two protomers.
- Steric clashes
-
When atoms from different residues come into close proximity, the resultant repulsion between the atoms leads to a change in conformation.
- Negative allostery
-
Also known as allosteric inhibition, occurs when binding of one ligand to a substrate (in this case a BRAF protomer) decreases the affinity of another ligand at a different site (that is, the other protomer).
- Therapeutic window
-
The range of concentrations of a drug in the patient that provides safe effective therapy. The therapeutic window is wide when the minimum toxic concentration is much higher than the minimum effective concentration.
- Allosteric priming
-
A phenomenon recently observed with several small-molecule inhibitors, by which binding of the inhibitor induces the active conformation of the target kinase, which results in its increased interaction with upstream regulators and consequent kinase priming and activating phosphorylation.
- Steric effects
-
A phenomenon arising as a result of the fact that each residue and its atoms occupy a certain amount of space in a defined conformation.
- Residence time
-
The period for which the target kinase is occupied by the small-molecule inhibitor.
- Off-rate
-
The rate of dissociation of the small-molecule inhibitor from the kinase.
- Cross-resistance
-
The development of tumour resistance to a potential therapy after treatment of a patient with a different therapeutic agent.
Rights and permissions
About this article
Cite this article
Karoulia, Z., Gavathiotis, E. & Poulikakos, P. New perspectives for targeting RAF kinase in human cancer. Nat Rev Cancer 17, 676–691 (2017). https://doi.org/10.1038/nrc.2017.79
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.79
This article is cited by
-
Combinatorial strategies to target RAS-driven cancers
Nature Reviews Cancer (2024)
-
BRAF — a tumour-agnostic drug target with lineage-specific dependencies
Nature Reviews Clinical Oncology (2024)
-
Rebound growth of BRAF mutant pediatric glioma cells after MAPKi withdrawal is associated with MAPK reactivation and secretion of microglia-recruiting cytokines
Journal of Neuro-Oncology (2024)
-
Targeting CRAF kinase in anti-cancer therapy: progress and opportunities
Molecular Cancer (2023)
-
In vitro and in vivo analyses on anti-NSCLC activity of apatinib: rediscovery of a new drug target V600E mutation
Cancer Cell International (2023)