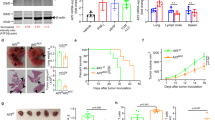
Abstract
Dendritic cells (DCs), a type of professional antigen-presenting cells, are responsible for initiation and maintenance of immune responses. Here we report that a substantial proportion of DCs in tumor-bearing mice and people with cancer have high amounts of triglycerides as compared with DCs from tumor-free mice and healthy individuals. In our studies, lipid accumulation in DCs was caused by increased uptake of extracellular lipids due to upregulation of scavenger receptor A. DCs with high lipid content were not able to effectively stimulate allogeneic T cells or present tumor-associated antigens. DCs with high and normal lipid levels did not differ in expression of major histocompatibility complex and co-stimulatory molecules. However, lipid-laden DCs had a reduced capacity to process antigens. Pharmacological normalization of lipid abundance in DCs with an inhibitor of acetyl-CoA carboxylase restored the functional activity of DCs and substantially enhanced the effects of cancer vaccines. These findings suggest that immune responses in cancer can be improved by manipulating the lipid levels in DCs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gabrilovich, D.I. The mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat. Rev. Immunol. 4, 941–952 (2004).
Shurin, M. & Chatta, G. Immunobiology of dendritic cells in cancer. in Tumor-induced immune suppression. Mechanisms and theraputic reversal. (eds. Gabrilovich, D.I. and Hurwitz, A.) 101–130 (Springer, New York, 2008).
Calder, P.C. & Burdge, G.C. Fattty acids. in Bioactive lipids (eds. Nicolaou, A. & Kokotos, G.) 1–36 (Bridgewater: The Oily Press, 2004).
Shaikh, S.R. & Edidin, M. Polyunsaturated fatty acids, membrane organization, T cells and antigen presentation. Am. J. Clin. Nutr. 84, 1277–1289 (2006).
Knight, S.C. Specialized perinodal fat fuels and fashions immunity. Immunity 28, 135–138 (2008).
Sanderson, P., MacPherson, G.G., Jenkins, C.H. & Calder, P.C. Dietary fish oil diminishes the antigen presentation activity of rat dendritic cells. J. Leukoc. Biol. 62, 771–777 (1997).
Touitou, E., Godin, B., Karl, Y., Bujanover, S. & Becker, Y. Oleic acid, a skin penetration enhancer, affects Langerhans cells and corneocytes. J. Control. Release 80, 1–7 (2002).
Säemann, M.D. et al. Bacterial metabolite interference with maturation of human monocyte-derived dendritic cells. J. Leukoc. Biol. 71, 238–246 (2002).
Zeyda, M. et al. Polyunsaturated fatty acids block dendritic cell activation and function independently of NF-κB activation. J. Biol. Chem. 280, 14293–14301 (2005).
Aliberti, J., Hieny, S., Reis e Sousa, C., Serhan, C.N. & Sher, A. Lipoxin-mediated inhibition of IL-12 production by DCs: a mechanism for regulation of microbial immunity. Nat. Immunol. 3, 76–82 (2002).
Weatherill, A.R. et al. Saturated and polyunsaturated fatty acids reciprocally modulate dendritic cell functions mediated through TLR4. J. Immunol. 174, 5390–5397 (2005).
Shamshiev, A.T. et al. Dyslipidemia inhibits Toll-like receptor–induced activation of CD8α-negative dendritic cells and protective Th1 type immunity. J. Exp. Med. 204, 441–452 (2007).
Angeli, V. et al. Dyslipidemia associated with atherosclerotic disease systemically alters dendritic cell mobilization. Immunity 21, 561–574 (2004).
Packard, R.R. et al. CD11c+ dendritic cells maintain antigen processing, presentation capabilities and CD4+ T cell priming efficacy under hypercholesterolemic conditions associated with atherosclerosis. Circ. Res. 103, 965–973 (2008).
Perrot, I. et al. Dendritic cells infiltrating human non-small cell lung cancer are blocked at immature stage. J. Immunol. 178, 2763–2769 (2007).
Halvorson, D.L. & McCune, S.A. Inhibition of fatty acid synthesis in isolated adipocytes by 5-(tetradecyloxy)-2-furoic acid. Lipids 19, 851–856 (1984).
Nagaraj, S. et al. Dendritic cell–based full-length survivin vaccine in treatment of experimental tumors. J. Immunother. 30, 169–179 (2007).
de Winther, M.P., van Dijk, K.W., Havekes, L.M. & Hofker, M.H. Macrophage scavenger receptor class A: a multifunctional receptor in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 20, 290–297 (2000).
Peiser, L., Mukhopadhyay, S. & Gordon, S. Scavenger receptors in innate immunity. Curr. Opin. Immunol. 14, 123–128 (2002).
Husemann, J., Loike, J.D., Anankov, R., Febbraio, M. & Silverstein, S.C. Scavenger receptors in neurobiology and neuropathology: their role on microglia and other cells of the nervous system. Glia 40, 195–205 (2002).
Hagemann, T. et al. Ovarian cancer cells polarize macrophages toward a tumor-associated phenotype. J. Immunol. 176, 5023–5032 (2006).
Becker, M., Cotena, A., Gordon, S. & Platt, N. Expression of the class A macrophage scavenger receptor on specific subpopulations of murine dendritic cells limits their endotoxin response. Eur. J. Immunol. 36, 950–960 (2006).
Jin, J.O. et al. Ligand of scavenger receptor class A indirectly induces maturation of human blood dendritic cells via production of tumor necrosis factor-alpha. Blood 113, 5839–5847 (2009).
Delimaris, I. et al. Oxidized LDL, serum oxidizability and serum lipid levels in patients with breast or ovarian cancer. Clin. Biochem. 40, 1129–1134 (2007).
Motta, M. et al. Antibodies against ox-LDL serum levels in patients with hepatocellular carcinoma. Panminerva Med. 45, 69–73 (2003).
Youn, J.I., Nagaraj, S., Collazo, M. & Gabrilovich, D.I. Subsets of myeloid-derived suppressor cells in tumor-bearing mice. J. Immunol. 181, 5791–5802 (2008).
Hogquist, K.A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Barnden, M.J., Allison, J., Heath, W.R. & Carbone, F.R. Defective TCR expression in transgenic mice constructed using cDNA-based α- and β-chain genes under the control of heterologous regulatory elements. Immunol. Cell Biol. 76, 34–40 (1998).
Nefedova, Y. et al. Hyperactivation of STAT3 is involved in abnormal differentiation of dendritic cells in cancer. J. Immunol. 172, 464–474 (2004).
Nefedova, Y., Cheng, P., Alsina, M., Dalton, W.S. & Gabrilovich, D.I. Involvement of Notch-1 signaling in bone marrow stroma-mediated de novo drug resistance of myeloma and other malignant lymphoid cell lines. Blood 103, 3503–3510 (2004).
Acknowledgements
This work was supported by US National Institutes of Health grant 1R21AI070598 to D.I.G., National Institutes of Health grants HL70755, HL094488 and OH008282 to V.E.K. and, in part, by the flow cytometry core of H. Lee Moffitt Cancer Center. Poly-IC was provided by A. Salazar (Oncovir).
Author information
Authors and Affiliations
Contributions
D.L.H. performed initial experiments and participated in the writing of the paper; W.C., Y.N., S.V.N., S.N., A.C. and B.L. performed experiments investigating the mechanism and immunological consequences of lipid accumulation in DCs and analyzed the data; V.E.K. and V.A.T. designed and performed experiments with mass spectrometry analysis of lipid content, analyzed the data and participated in writing the paper; E.C. and H.-I.C. designed and performed experiments with B16F10 model and analyzed the data; S.C.K. participated in the design of the original experiments and participated in writing the paper; T.P., T.V.M., J.C.M. and S.A. participated in experiments evaluating human samples; M.F. and R.L.F. participated in experiments evaluating human samples and participated in writing the paper; D.I.G. designed the study, analyzed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Methods and Supplementary Discussion (PDF 486 kb)
Rights and permissions
About this article
Cite this article
Herber, D., Cao, W., Nefedova, Y. et al. Lipid accumulation and dendritic cell dysfunction in cancer. Nat Med 16, 880–886 (2010). https://doi.org/10.1038/nm.2172
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2172
This article is cited by
-
Immunosurveillance encounters cancer metabolism
EMBO Reports (2024)
-
Fatty acid metabolism of immune cells: a new target of tumour immunotherapy
Cell Death Discovery (2024)
-
The fatty acid-related gene signature stratifies poor prognosis patients and characterizes TIME in cutaneous melanoma
Journal of Cancer Research and Clinical Oncology (2024)
-
The impact of lipid metabolism on breast cancer: a review about its role in tumorigenesis and immune escape
Cell Communication and Signaling (2023)
-
Endoplasmic reticulum stress mediates the myeloid-derived immune suppression associated with cancer and infectious disease
Journal of Translational Medicine (2023)