Abstract
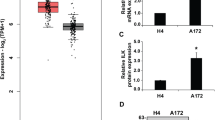
The Fyn related kinase (FRK), a member of intracellular Src-related tyrosine kinases, was recently reported to function as a potent tumor suppressor in several cancer types. However, the expression level and functional significance of FRK in human malignant glioma, which is characterized by high migration and invasion potential, have never been investigated. We reported here that FRK reduced cell migration and invasion via inhibiting the c-Jun N-terminal protein kinase (JNK)/c-Jun signaling pathway in glioma cells. The mRNA and protein levels of FRK were significantly down-regulated in human primary glioma tissues. In addition, over-expression of FRK inhibited migration and invasion of glioma cells and excretion of the matrix metalloprotease 2 (MMP2), an index of migration and invasion. Furthermore, over-expression of FRK inhibited phosphorylation of JNK and c-Jun, which play important role in cell migration and invasion. Finally, the effects of FRK on cell migration and invasion and JNK/c-Jun inhibition were abolished by anisomycin, a JNK specific activator. In summary, these results clearly indicate that FRK may play a protective role against the progression of glioma by suppressing cell migration and invasion, suggesting that FRK needs to be further studied in its detail mechanism and clinical significant.





Similar content being viewed by others
Abbreviations
- FRK:
-
The Fyn related kinase
- JNK:
-
c-Jun NH2-terminal kinase
- MMPs:
-
Matrix metalloproteases
References
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507
Buonerba C, Di Lorenzo G, Marinelli A, Federico P, Palmieri G, Imbimbo M, Conti P, Peluso G, De Placido S, Sampson JH (2011) A comprehensive outlook on intracerebral therapy of malignant gliomas. Crit Rev Oncol Hematol 80:54–68
Sherman JH, Hoes K, Marcus J, Komotar RJ, Brennan CW, Gutin PH (2011) Neurosurgery for brain tumors: update on recent technical advances. Curr Neurol Neurosci Rep 11:313–319
Onishi M, Ichikawa T, Kurozumi K, Date I (2011) Angiogenesis and invasion in glioma. Brain Tumor Pathol 28:13–24
Kim CS, Jung S, Jung TY, Jang WY, Sun HS, Ryu HH (2011) Characterization of invading glioma cells using molecular analysis of leading-edge tissue. J Kor Neurosurg Soc 50:157–165
Charles NA, Holland EC, Gilbertson R, Glass R, Kettenmann H (2011) The brain tumor microenvironment. Glia 59:1169–1180
Tysnes BB, Mahesparan R (2001) Biological mechanisms of glioma invasion and potential therapeutic targets. J Neurooncol 53:129–147
Wheeler DL, Iida M, Dunn EF (2009) The role of Src in solid tumors. Oncologist 14:667–678
Craven RJ, Cance WG, Liu ET (1995) The nuclear tyrosine kinase Rak associates with the retinoblastoma protein pRb. Cancer Res 55:3969–3972
Anneren C, Lindholm CK, Kriz V, Welsh M (2003) The FRK/RAK-SHB signaling cascade: a versatile signal-transduction pathway that regulates cell survival, differentiation and proliferation. Curr Mol Med 3:313–324
Meyer T, Xu L, Chang J, Liu ET, Craven RJ, Cance WG (2003) Breast cancer cell line proliferation blocked by the Src-related Rak tyrosine kinase. Int J Cancer 104:139–146
Oberg-Welsh C, Anneren C, Welsh M (1998) Mutation of C-terminal tyrosine residues Y497/Y504 of the Src-family member Bsk/Iyk decreases NIH3T3 cell proliferation. Growth Factors 16:111–124
Craven RJ, Cance WG, Liu ET (1995) The nuclear tyrosine kinase Rak associates with the retinoblastoma protein pRb. Cancer Res 55:3969–3972
Chandrasekharan S, Qiu TH, Alkharouf N, Brantley K, Mitchell JB, Liu ET (2002) Characterization of mice deficient in the Src family nonreceptor tyrosine kinase Frk/rak. Mol Cell Biol 22:5235–5247
Yim EK, Peng G, Dai H, Hu R, Li K, Lu Y, Mills GB, Meric-Bernstam F, Hennessy BT, Craven RJ, Lin SY (2009) Rak functions as a tumor suppressor by regulating PTEN protein stability and function. Cancer Cell 15:304–314
Yim EK, Siwko S, Lin SY (2009) Exploring Rak tyrosine kinase function in breast cancer. Cell Cycle 8:2360–2364
Chaw KC, Manimaran M, Tay FE, Swaminathan S (2007) Matrigel coated polydimethylsiloxane based microfluidic devices for studying metastatic and non-metastatic cancer cell invasion and migration. Biomed Microdevices 9:597–602
Gabelloni P, Da Pozzo E, Bendinelli S, Costa B, Nuti E, Casalini F, Orlandini E, Da Settimo F, Rossello A, Martini C (2010) Inhibition of metalloproteinases derived from tumours: new insights in the treatment of human glioblastoma. Neuroscience 168:514–522
Kamino M, Kishida M, Kibe T, Ikoma K, Iijima M, Hirano H, Tokudome M, Chen L, Koriyama C, Yamada K, Arita K, Kishida S (2011) Wnt-5a signaling is correlated with infiltrative activity in human glioma by inducing cellular migration and MMP-2. Cancer Sci 102:540–548
Jaalinoja J, Herva R, Korpela M, Hoyhtya M, Turpeenniemi-Hujanen T (2000) Matrix metalloproteinase 2 (MMP-2) immunoreactive protein is associated with poor grade and survival in brain neoplasms. J Neurooncol 46:81–90
Kennedy NJ, Davis RJ (2003) Role of JNK in tumor development. Cell Cycle 2:199–201
Ma CY, Ji WT, Chueh FS, Yang JS, Chen PY, Yu CC, Chung JG (2011) Butein inhibits the migration and invasion of SK-HEP-1 human hepatocarcinoma cells through suppressing the ERK, JNK, p38, and uPA signaling multiple pathways. J Agric Food Chem 59:9032–9038
Song H, Moon A (2006) Glial cell-derived neurotrophic factor (GDNF) promotes low-grade Hs683 glioma cell migration through JNK, ERK-1/2 and p38 MAPK signaling pathways. Neurosci Res 56:29–38
Cance WG, Craven RJ, Bergman M, Xu L, Alitalo K, Liu ET (1994) Rak, a novel nuclear tyrosine kinase expressed in epithelial cells. Cell Growth Differ 5:1347–1355
Serfas MS, Tyner AL (2003) Brk, Srm, Frk, and Src42A form a distinct family of intracellular Src-like tyrosine kinases. Oncol Res 13:409–419
Forsyth PA, Wong H, Laing TD, Rewcastle NB, Morris DG, Muzik H, Leco KJ, Johnston RN, Brasher PM, Sutherland G, Edwards DR (1999) Gelatinase-A (MMP-2), gelatinase-B (MMP-9) and membrane type matrix metalloproteinase-1 (MT1-MMP) are involved in different aspects of the pathophysiology of malignant gliomas. Br J Cancer 79:1828–1835
Raithatha SA, Muzik H, Rewcastle NB, Johnston RN, Edwards DR, Forsyth PA (2000) Localization of gelatinase-A and gelatinase-B mRNA and protein in human gliomas. Neuro Oncol 2:145–150
Brell M, Ibanez J, Felpete A, Burguera B, Frontera M, Couce ME (2011) Quantitative analysis of matrix metalloproteinase-2 mRNA expression in central and peripheral regions of gliomas. Brain Tumor Pathol 28:137–144
Weston CR, Davis RJ (2007) The JNK signal transduction pathway. Curr Opin Cell Biol 19:142–149
Vallerie SN, Hotamisligil GS (2010) The role of JNK proteins in metabolism. Sci Transl Med 2:60rv65
Hauck CR, Sieg DJ, Hsia DA, Loftus JC, Gaarde WA, Monia BP, Schlaepfer DD (2001) Inhibition of focal adhesion kinase expression or activity disrupts epidermal growth factor-stimulated signaling promoting the migration of invasive human carcinoma cells. Cancer Res 61:7079–7090
Huynh-Do U, Vindis C, Liu H, Cerretti DP, McGrew JT, Enriquez M, Chen J, Daniel TO (2002) Ephrin-B1 transduces signals to activate integrin-mediated migration, attachment and angiogenesis. J Cell Sci 115:3073–3081
Li Z, Xu X, Bai L, Chen W, Lin Y (2011) Epidermal growth factor receptor-mediated tissue transglutaminase overexpression couples acquired tumor necrosis factor-related apoptosis-inducing ligand resistance and migration through c-FLIP and MMP-9 proteins in lung cancer cells. J Biol Chem 286:21164–21172
Nomura N, Nomura M, Mizuki N, Hamada J (2008) Rac1 mediates phorbol 12-myristate 13-acetate-induced migration of glioblastoma cells via paxillin. Oncol Rep 20:705–711
Mendes KN, Wang GK, Fuller GN, Zhang W (2010) JNK mediates insulin-like growth factor binding protein 2/integrin alpha5-dependent glioma cell migration. Int J Oncol 37:143–153
Fromigue O, Hamidouche Z, Marie PJ (2008) Blockade of the RhoA-JNK-c-Jun-MMP2 cascade by atorvastatin reduces osteosarcoma cell invasion. J Biol Chem 283:30549–30556
Collins CS, Hong J, Sapinoso L, Zhou Y, Liu Z, Micklash K, Schultz PG, Hampton GM (2006) A small interfering RNA screen for modulators of tumor cell motility identifies MAP4K4 as a promigratory kinase. Proc Natl Acad Sci USA 103:3775–3780
Van Slambrouck S, Grijelmo C, De Wever O, Bruyneel E, Emami S, Gespach C, Steelant WF (2007) Activation of the FAK-src molecular scaffolds and p130Cas-JNK signaling cascades by alpha1-integrins during colon cancer cell invasion. Int J Oncol 31:1501–1508
Kwon GT, Cho HJ, Chung WY, Park KK, Moon A, Park JH (2009) Isoliquiritigenin inhibits migration and invasion of prostate cancer cells: possible mediation by decreased JNK/AP-1 signaling. J Nutr Biochem 20:663–676
Ip YT, Davis RJ (1998) Signal transduction by the c-Jun N-terminal kinase (JNK)–from inflammation to development. Curr Opin Cell Biol 10:205–219
Shaulian E (2010) AP-1–The Jun proteins: oncogenes or tumor suppressors in disguise? Cell Signal 22:894–899
Eferl R, Wagner EF (2003) AP-1: a double-edged sword in tumorigenesis. Nat Rev Cancer 3:859–868
Koul D, Shen R, Shishodia S, Takada Y, Bhat KP, Reddy SA, Aggarwal BB, Yung WK (2007) PTEN down regulates AP-1 and targets c-fos in human glioma cells via PI3-kinase/Akt pathway. Mol Cell Biochem 300:77–87
Acknowledgments
We are deeply indebted to Dr. Shiaw-Yih Lin of the University of Texas M.D. Anderson Cancer Center in USA for the gift of the plasmid encoding FRK gene. This study was supported in part by the projects of National Natural Science Foundation of China (No. 81072072, No. 31070933), and the grant from State Key Laboratory of Neuroscience, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (SKLN-2011A01).
Conflict of interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiuping Zhou and Lei Hua contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhou, X., Hua, L., Zhang, W. et al. FRK controls migration and invasion of human glioma cells by regulating JNK/c-Jun signaling. J Neurooncol 110, 9–19 (2012). https://doi.org/10.1007/s11060-012-0933-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-012-0933-1