Abstract
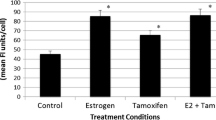
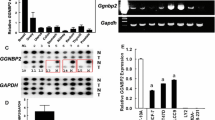
Early growth response-1 (Egr-1) is an immediate early gene involved in relevant biological events including the proliferation of diverse types of cell tumors. In a microarray analysis performed in breast cancer cells, 17β-estradiol (E2) and the estrogen receptor antagonist 4-hydroxitamoxifen (OHT) up-regulated Egr-1 through the G protein-coupled receptor named GPR30/GPER. Hence, in this study, we aimed to provide evidence regarding the ability of E2, OHT and the selective GPER ligand G-1 to regulate Egr-1 expression and function through the GPER/EGFR/ERK transduction pathway in both Ishikawa (endometrial) and SkBr3 (breast) cancer cells. Interestingly, we demonstrate that Egr-1 is involved in the transcription of genes regulating cell proliferation like CTGF and cyclin D1 and required for the proliferative effects induced by E2, OHT, and G-1 in both Ishikawa and SkBr3 cells. In addition, we show that GPER mediates the expression of Egr-1 also in carcinoma-associated fibroblasts (CAFs). Our data suggest that Egr-1 may represent an important mediator of the biological effects induced by E2 and OHT through GPER/EGFR/ERK signaling in breast and endometrial cancer cells. The results obtained in CAFs provide further evidence regarding the potential role exerted by the GPER-dependent Egr-1 up-regulation in tumor development and progression. Therefore, Egr-1 may be included among the bio-markers of estrogen and antiestrogen actions and may be considered as a further therapeutic target in both breast and endometrial tumors.










Similar content being viewed by others
References
Morgan JI, Curran T (1995) Immediate-early genes: ten years on. Trends Neurosci 18:66–67
Carbone M, Levis AS (1990) Oncogenes, antioncogenes and the regulation of cell growth. Trends Endocrinol Metab 1:248–253
Sukhatme VP (1990) Early transcriptional events in cell growth: the Egr family. J Am Soc Nephrol 1:859–866
Gashler A, Sukhatme VP (1995) Early growth response protein 1 (Egr-1): prototype of a zinc-finger family of transcription factors. Prog Nucleic Acid Res Mol Biol 50:191–224
Christy B, Nathans D (1989) DNA binding site of the growth factor-inducible protein Zif268. Proc Natl Acad Sci USA 86:8737–8741
O’Donovan KJ, Tourtellotte WG, Millbrandt J, Baraban JM (1999) The EGR family of transcription regulatory factors: progress at the interface of molecular and systems neuroscience. Trends Neurosci 22:167–173
Gitenay D, Baron VT (2009) Is EGR1 a potential target for prostate cancer therapy? Futur Oncol 5:993–1003
Shao H, Kono DH, Chen LY, Rubin EM, Kaye J (1997) Induction of the early growth response (Egr) family of transcription factors during thymic selection. J Exp Med 185:731–744
Yan SF, Lu J, Zou YS, Soh-Won J, Cohen DM, Buttrick PM, Cooper DR, Steinberg SF, Mackman N, Pinsky DJ, Stern DM (1999) Hypoxia-associated induction of early growth response-1 gene expression. J Biol Chem 274:15030–15040
Mora-Garcia P, Sakamoto KM (2000) Granulocyte colony-stimulating factor induces Egr-1 up-regulation through interaction of serum response element-binding proteins. J Biol Chem 275:22418–22426
Hodge C, Liao J, Stofega M, Guan K, Carter-Su C, Schwartz J (1998) Growth hormone stimulates phosphorylation and activation of Elk-1 and expression of c-fos, Egr-1, and JunB through activation of extracellular signal-regulated kinases 1 and 2. J Biol Chem 273:31327–31336
Rolli M, Kotlyarov A, Sakamoto KM, Gaestel M, Neininger A (1999) Stress-induced stimulation of early growth response gene-1 by p38/stress-activated protein kinase 2 is mediated by a cAMP-responsive promoter element in a MAPKAP kinase 2-independent manner. J Biol Chem 274:19559–19564
Sukhatme VP, Cao X, Chang LC, Tsai-Morris C-W, Stamenkovich D, Ferreira PCP, Cohen DR, Edwards SA, Shows TB, Curran T, Le Beau MM, Adamson ED (1988) A zinc finger-encoding gene coregulated with c-fos during growth and differentiation, and after cellular depolarization. Cell 53:37–43
Huang RP, Fan Y, de Belle I, Niemeyer C, Gottardis MM, Mercola D, Adamson ED (1997) Decreased Egr-1 expression in human, mouse, and rat mammary cells and tissues correlates with tumor formation. Int J Cancer 72:102–109
Nair P, Muthukkumar S, Sells SF, Han SS, Sukhatme VP, Rangnekar VM (1997) Early growth response-1-dependent apoptosis is mediated by p53. J Biol Chem 272:20131–20138
Cicatiello L, Sica V, Bresciani F, Weisz A (1993) Identification of a specific pattern of “immediate-early” gene activation induced by estrogen during mitogenic stimulation of rat uterine cells. Receptor 3:17–30
Naciff JM, Overmann GJ, Torontali SM, Carr GJ, Khambatta ZS, Tiesman JP, Richardson BD, Daston GP (2007) Uterine temporal response to acute exposure to 17α-ethinyl estradiol in the immature rat. Toxicol Sci 97:467–490
Pratt MAC, Satkunaratnam A, Novosad DM (1998) Estrogen activates raf-1 kinase and induces expression of Egr-1 in MCF-7 breast cancer cells. Mol Cell Biochem 189:119–125
Chen CC, Lee WR, Safe S (2004) Egr-1 is activated by 17beta-estradiol in MCF-7 cells by mitogen-activated protein kinase-dependent phosphorylation of ELK-1. J Cell Biochem 93:1063–1074
Pandey DP, Lappano R, Albanito L, Madeo A, Maggiolini M, Picard D (2009) Estrogenic GPR30 signalling induces proliferation and migration of breast cancer cells through CTGF. EMBO J 28:523–532
Maggiolini M, Vivacqua A, Fasanella G, Recchia AG, Sisci D, Pezzi V, Montanaro D, Musti AM, Picard D, Andò S (2004) The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17β-estradiol and phytoestrogens in breast cancer cells. J Biol Chem 279:27008–27016
Revankar CM, Cimino DF, Sklar LA, Arterburn JB, Prossnitz ER (2005) A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 307:1625–1630
Thomas P, Pang Y, Filardo EJ, Dong J (2005) Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells. Endocrinology 146:624–632
Vivacqua A, Bonofiglio D, Albanito L, Madeo A, Rago V, Carpino A, Musti AM, Picard D, Andò S, Maggiolini M (2006) 17β-Estradiol, genistein and 4-hydroxytamoxifen induce the proliferation of thyroid cancer cells through the G protein-coupled receptor GPR30. Mol Pharmacol 70:1414–1423
Vivacqua A, Bonofiglio D, Recchia AG, Musti AM, Picard D, Ando` S, Maggiolini M (2006) The G protein-coupled receptor GPR30 mediates the proliferative effects induced by 17β-estradiol and hydroxytamoxifen in endometrial cancer cells. Mol Endocrinol 20:631–646
Albanito L, Madeo A, Lappano R, Vivacqua A, Rago V, Carpino A, Oprea TI, Prossnitz ER, Musti AM, Ando S, Maggiolini M (2007) G protein-coupled receptor 30 (GPR30) mediates gene expression changes and growth response to 17β-estradiol and selective GPR30 ligand G-1 in ovarian cancer cells. Cancer Res 67:1859–1866
Albanito L, Lappano R, Madeo A, Chimento A, Prossnitz ER, Cappello AR, Dolce V, Abonante S, Pezzi V, Maggiolini M (2008) G-protein-coupled receptor 30 and estrogen receptor-α are involved in the proliferative effects induced by atrazine in ovarian cancer cells. Environ Health Perspect 116:1648–1655
Albanito L, Sisci D, Aquila S, Brunelli E, Vivacqua A, Madeo A, Lappano R, Pandey DP, Picard D, Mauro L, Maggiolini M, Maggiolini M (2008) EGF induces GPR30 expression in estrogen receptor negative breast cancer cells. Endocrinology 149:3799–3808
Vivacqua A, Lappano R, De Marco P, Sisci D, Aquila S, De Amicis F, Fuqua SA, Andò S, Maggiolini M (2009) G protein-coupled receptor 30 expression is up-regulated by EGF and TGF alpha in estrogen receptor alpha-positive cancer cells. Mol Endocrinol 23:1815–1826
Filardo EJ, Quinn JA, Bland KI, Frackelton AR Jr (2000) Estrogen induced activation of Erk-1 and Erk-2 requires the G protein coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol Endocrinol 14:1649–1660
Cui L, Zhang Q, Mao Z, Chen J, Wang X, Qu J, Zhang J, Jin D (2011) CTGF is overexpressed in papillary thyroid carcinoma and promotes the growth of papillary thyroid cancer cells. Tumour Biol 32:721–728
Fu M, Wang C, Li Z, Sakamaki T, Pestell RG (2004) Minireview: Cyclin D1: normal and abnormal functions. Endocrinology 145:5439–5447
Xiao D, Chinnappan D, Pestell R, Albanese C, Weber HC (2005) Bombesin regulates Cyclin D1 expression through the early growth response protein Egr-1 in prostate cancer cells. Cancer Res 65:9934–9942
Deng YZ, Chen PP, Wang Y, Yin D, Koeffler HP, Li B, Tong XJ, Xie D (2007) Connective tissue growth factor is overexpressed in esophageal squamous cell carcinoma and promotes tumorigenicity through beta-catenin-T-cell factor/Lef signaling. J Biol Chem 282:36571–36581
Liu LY, Han YC, Wu SH, Lv ZH (2008) Expression of connective tissue growth factor in tumor tissues is an independent predictor of poor prognosis in patients with gastric cancer. World J Gastroenterol 14:2110–2114
Mullis TC, Tang X, Chong KT (2008) Expression of connective tissue growth factor (CTGF/CCN2) in head and neck squamous cell carcinoma. J Clin Pathol 61:606–610
Kang Y, Siegel PM, Shu W, Drobnjak M, Kakonen SM, Cordon-Cardo C, Guise TA, Massagué J (2003) A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3:537–549
Han HJ, Russo J, Kohwi Y, Kohwi-Shigematsu T (2008) SATB1 reprogrammes gene expression to promote breast tumour growth and metastasis. Nature 452:187–193
Guillemot L, Levy A, Raymondjean M, Rothhut B (2001) Angiotensin II-induced transcriptional activation of the cyclin D1 gene is mediated by Egr-1 in CHOAT(1A) cells. J Biol Chem 276:39394–39403
De Sousa LP, Brasil BS, Silva BM, Freitas MH, Nogueira SV, Ferreira PC, Kroon EG, Bonjardim CA (2005) Plasminogen/plasmin regulates c-fos and egr-1 expression via the MEK/ERK pathway. Biochem Biophys Res Commun 329:237–245
Dupont WD, Page DL (1991) Menopausal estrogen replacement therapy and breast cancer. Arch Intern Med 151:67–72
Clavel-Chapelon F, Hill C (2000) Hormone replacement therapy in menopause and risk of breast cancer. Presse Med 29:1688–1693
Fournier A, Hill C, Clavel-Chapelon F (2003) Hormone replacement therapy in menopause and risk of breast cancer. Bull Cancer 90:821–831
Early Breast Cancer Trialist’ Collaborative Group (2005) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-years survival: an overview of the randomized trials. Lancet 365:1687–1700
Dowsett M, Houghton J, Iden C, Salter J, Farndon J, A’ Hern R, Sainsbury R, Baum M (2006) Benefit from adjuvant tamoxifen therapy in primary breast cancer patients according oestrogen receptor, progesterone receptor, EGF receptor and HER2 status. Ann Oncol 17:818–826
Herynk MH, Fuqua SA (2007) Estrogen receptors in resistance to hormone therapy. Adv Exp Med Biol 608:130–143
Linke SP, Bremer TM, Herold CD, Sauter G, Diamond C (2006) A multimarker model to predict outcome in tamoxifen-treated breast cancer patients. Clin Cancer Res 12:1175–1183
Ignatov A, Ignatov T, Roessner A, Costa SD, Kalinski T (2010) Role of GPR30 in the mechanisms of tamoxifen resistance in breast cancer MCF-7 cells. Breast Cancer Res Treat 123:87–96
Filardo EJ, Graeber CT, Quinn JA, Resnick MB, Giri D, DeLellis RA, Steinhoff MM, Sabo E (2006) Distribution of GPR30, a seven membrane-spanning estrogen receptor, in primary breast cancer and its association with clinicopathologic determinants of tumor progression. Clin Cancer Res 12:6359–6366
Smith HO, Leslie KK, Singh M, Qualls CR, Revankar CM, Joste NE, Prossnitz ER (2007) GPR30: a novel indicator of poor survival for endometrial carcinoma. Am J Obstet Gynecol 196:386 e381–389
Madeo A, Maggiolini M (2010) Nuclear alternate estrogen receptor GPR30 mediates 17beta-estradiol-induced gene expression and migration in breast cancer-associated fibroblasts. Cancer Res 70:6036–6046
Morelli C, Garofalo C, Sisci D, del Rincon S, Cascio S, Tu X, Vecchione A, Sauter ER, Miller WH Jr, Surmacz E (2004) Nuclear insulin receptor substrate 1 interacts with estrogen receptor α at ERE promoters. Oncogene 23:7517–7526
Musgrove EA (2006) Cyclins: roles in mitogenic signaling and oncogenic transformation. Growth Factors 24:13–19
Chu CY, Chang CC, Prakash E, Kuo ML (2008) Connective tissue growth factor (CTGF) and cancer progression. J Biomed Sci 15:675–685
Kalluri R, Zeisberg M (2006) Fibroblasts in cancer. Nat Rev Cancer 6:392–401
Polyak K (2007) Breast cancer: origins and evolution. J Clin Invest 117:3155–3163
Filardo EJ, Quinn JA, Sabo E (2008) Association of the membrane estrogen receptor, GPR30, with breast tumor metastasis and transactivation of the epidermal growth factor receptor. Steroids 73:870–873
Prossnitz ER, Maggiolini M (2009) Mechanisms of estrogen signaling and gene expression via GPR30. Mol Cell Endocrinol 308:32–38
Maggiolini M, Picard D (2010) The unfolding stories of GPR30, a new membrane bound estrogen receptor. J Endocrinol 204:105–114
Scott JA, McGuire WL (1991) New molecular markers of prognosis in breast cancer. Raven Press, New York
Ponzone R, Biglia N, Jacomuzzi ME, Mariani L, Dominguez A, Sismondi P (2006) Antihormones in prevention and treatment of breast cancer. Ann N Y Acad Sci 1089:143–158
Howell A, DeFriend D, Robertson J, Blamey R, Walton P (1995) Response to a specific antioestrogen (ICI 182780) in tamoxifen-resistant breast cancer. Lancet 345:29–30
Ishii Y, Waxman S, Germain D (2008) Tamoxifen stimulates the growth of cyclin D1-overexpressing breast cancer cells by promoting the activation of signal transducer and activator of transcription 3. Cancer Res 68:852–860
Massarweh S, Osborne CK, Creighton CJ, Qin L, Tsimelzon A, Huang S, Weiss H, Rimawi M, Schiff R (2008) Tamoxifen resistance in breast tumors is driven by growth factor receptor signaling with repression of classic estrogen receptor genomic function. Cancer Res 68:826–833
Herynk MH, Fuqua SAW (2004) Estrogen receptor mutations in human disease. Endocr Rev 25:869–898
Giordano C, Cui Y, Barone I, Andò S, Mancini MA, Berno V, Fuqua SA (2009) Growth factor-induced resistance to tamoxifen is associated with a mutation of estrogen receptor alpha and its phosphorylation at serine 305. Breast Cancer Res Treat 119:71–85
Bhowmick NA, Neilson EG, Moses HL (2004) Stromal fibroblasts in cancer initiation and progression. Nature 432:332–337
Acknowledgments
This study was supported by Associazione Italiana per la Ricerca sul Cancro (AIRC, project no. 8925/2009 and Calabria project, 2011), Fondazione Cassa di Risparmio di Calabria e Lucania and Ministero dell’Università e Ricerca Scientifica e Tecnologica (MIUR, Cofin project prot. 2008PK2WCW/2008).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vivacqua, A., Romeo, E., De Marco, P. et al. GPER mediates the Egr-1 expression induced by 17β-estradiol and 4-hydroxitamoxifen in breast and endometrial cancer cells. Breast Cancer Res Treat 133, 1025–1035 (2012). https://doi.org/10.1007/s10549-011-1901-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1901-8