Abstract
OBJECTIVE The aim of this investigation was to assess the association between single nucleotide polymorphisms (SNPs) in HLA-DRB1 and the risk of developing cervical cancer. Our study focused on the second exon of the HLA-DRB1 alleles, which have most of the SNP sites on HLA-DRB1.
METHODS We examined 30 cervical cancer patients and 66 control patients using the sequence-based typing polymerase chain reaction technique (PCR-SBT) to type 55 single nucleotide polymorphisms (SNPs) and haplotypes in the second exon of HLA-DRB1. The Chi-square test and the Bonferroni correction method were utilized for the statistical analysis of the data. An association between the alleles and cervical cancer was examined by the linkage disequilibrium test and the odds ratio (OR).
RESULTS Compared with the control group, among the 55 SNPs we studied in the second exon of HLA-DRB1, 4 showed an evident association with cervical cancer. Rs17880292 (P = 0.033, OR = 0.322) and rs1059586 (P = 0.029, OR = 2.657) had positive significance for the risk of developing cervical cancer, while rs17879702 (P = 0.016, OR = 0.222) and rs17882525 (P = 0.025, OR = 0.128) were negative. The difference in frequency of the 5582A-5592A-5667T haplotype between cervical cancer patients and the controls was significant (P = 0.043, OR = 2.735).
CONCLUSION The rs17880292 G/A genotype and the rs1059586 A/A genotype could be linked to an increased risk of cervical cancer. Rs17879702 and rs17882525 might be haplotype-tag SNPs (htSNPs) or belong to some other haplotypes, which might exert a protective effect against cervical cancer in combination. The 582A-5592A-5667T haplotype was shown to be a marker for susceptibility to cervical carcinogenesis.
keywords
Introduction
Cervical cancer is the second most common cancer in women worldwide, and about 9.8% or 371,200 new cancer cases are diagnosed every year[1], with more than 80% of these occurring in developing countries[2]. It is a preventable and curable disease if identified at an early stage. It is widely accepted that specific human papillomavirus (HPV) types, particularly high-risk HPV such as HPV16 and 18, are the central etiologic agents of cervical carcinogenesis[3]. More than 90% of cervical cancer is accompanied by HPV infection, but the HPV infection in the majority of women, can resolve spontaneously, and only a small proportion of women have infectious foci that ultimately develop into cervical cancer; those that do typically evolve after quite a long period of latency. Thus, both environmental and host factors play decisive roles in persistent HPV infections in which further development and malignant conversion of cervical epithelium occurs[4].
The human leukocyte antigen (HLA) system is involved in presenting foreign antigens to immune cells. This family of molecules is divided into 2 major classes. The Class I molecules which includes A, B and C are expressed in most nucleated cells and are essential to targeting infected cells for killing by cytotoxic T-cells. Certain HLA alleles have been shown to be associated with an increased or decreased risk for cervical cancer among different ethnic populations. It has been shown that protective alleles are more readily revealed and consistently observed in epidemiological studies. The role that SNPs in functional gene regions play in cervical carcinogenesis is largely unknown, although many previous studies have focused on HLA-DR alleles. In this study we have chosen the second exon of HLA-DRB1 which contains the most SNP alleles on the HLA-DRB1 coding region.
Materials and Methods
Patient samples
Patients with cervical cancer admitted and treated in Tianjin Central Hospital for Ob / Gyn from March 2006 to September 2006 were enrolled in the study. The diagnosis of cervical cancer in the patients was given in accordance with the WHO pathologic diagnostic guide.
The control group consisted of women with benign tumors who visited the same hospital for physical examination over the same time period. None of these women had a history of a previous oophorectomy, and none of the controls had a history of cancer or genetic disease. The study was approved by the Ethics Committee of Tianjin Cancer Institute, and an informed consent was obtained from all of the subjects recruited. Five ml of venous blood was taken from each subject using vacuum tubes containing EDTA and, the samples were then stored at 4°C.
DNA preparation and sequencing
Genomic DNA was extracted from blood using the TI-ANamp Genomic DNA Kit (Tiangen Biotech, Beijing, China) and the instructions from the company were followed carefully. After extraction, the genomic DNA was stored at -80°C. The sequences of PCR primers used in the assays for the second exon of HLA-DRB1 are shown in Table 1.
Forward and reverse primers of HLA-DRB1 for PCR and for sequencing.
The reaction conditions were as follows: 250 ng of DNA, 200 μmol/L of deoxynucleotide triphosphate, 3.75 mmol/L MgCl2, 0.4 μmol/L of each primer, and 1.25 units of TaqDNA polymerase. PCR reactions were denatured for 5 min at 96°C, and amplified 35 cycles at 96°C for 30 s, at 60°C for 30 s, and at 72°C for 30 s, followed by final extension at 72°C for 10 min. Each PCR experiment contained a negative control comprising all of the reagents except for DNA to rule out contamination.
HLA-DRB1 typing of genomic DNA was performed through polymerase chain reaction sequencing typing (PCR-SBT) method using BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, USA). Primers were provided by BGI Life Tech Co. Ltd. PCR products were depurated before the fluorescent dideoxy sequencing method. Sequencing reaction conditions were as follows: at 96°C for 2 min, followed by 25 cycles of 10 s at 95°C, 5 s at 50°C and 4 min at 60°C.
Statistical analysis
Hardy-Weinberg proportions were tested for each polymorphism. Linkage disequilibrium (D’r2) was detected between 2 SNPs according to the methods described in the literature[5]. Statistical analysis was performed using the SPSS11.0 software package (SPSS Company, Chicago, Illinois, USA). For each polymorphism, the genotype-specific odds ratio (OR) and 95% confidence intervals (CI) were computed using the Chi-square test. Fisher’s exact test was used to assess the global effect of this SNP. A value of P < 0.05 was considered as statistically significant.
Results
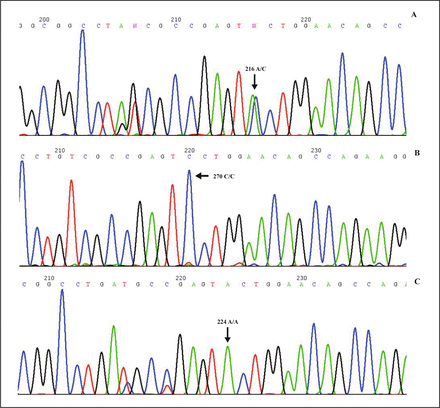
DNA sequencing was performed to identify the genotype of the SNP in the second exon of HLA-DRB1 and to determine whether the genotype of the SNP was heterozygous or homozygous. Fig. 1 shows some of the results from genotyping.
A, Heterozygous G/A homozygous genotype with G/A peak at rs1059586 genotype of HLA-DRB1. B, Homozygous C/C genotype with C peak at rs1059586 genotype of HLA-DRB1. C, Heterozygous A/A genotype with A peak at rs1059586 genotype of HLA-DRB1.
The results of the genetic analyses are shown in Table 2. In total, 55 nsSNPs in the second exon of HLA-DRB1 were tested in SNP database. In our study, 21 SNPs were not detected; 9 SNPs had 3 or 4 alleles; and other 25 SNPs had 2 alleles. As shown in Table 2, the frequency of 4 SNPs among 25 SNPs had significant differences in the case-control group (P < 0.05): rs17880292 (P = 0.033, OR = 0.322), rs1059586 (P = 0.029, OR = 2.657), rs17879702 (P = 0.016, OR = 0.222) and rs17882525 (P = 0.025, OR = 0.128). The distributions of the 4 genotypes were compatible with those expected from the Hardy-Weinberg equilibrium.
Genotype distributions of SNPs at the second exon of HLA-DRB1 in the study subjects.
Compared with the G/G (57Asp/Asp) genotype in rs17880292, the G/A57 (Asp→Ser) genotype significantly altered the risk of cervical cancer (OR = 0.322, 95%CI = 0.110-0.943), similar to the A/C (60Tyr/Ser) genotype and to the A/A (60Ser/Ser) genotype in rs1059586 (OR = 2.657, 95%CI = 1.089-6.482). We can conclude that the 2 SNPs are susceptibility loci for cervical cancer.
In addition, the risk of developing cervical cancer for the T/C (85Val/Ala) genotype of rs17882525 was lower than that for the T/T (85Val/Val) genotype and for the genotype C/C (85Ala/Ala). The risk for the C/T (16His/Tyr) genotype of rs17879702 was lower than that for the homozygous T/T (16Tyr/Tyr) genotype and for the homozygous C/C (16His/His) genotype.
The data in Tables 3 and 4 suggest that the rs17879702-rs17882525 genotype and the 582 A-5592A-5667T haplotype (P = 0.043, OR = 2.735) might play a role in the earliest stage of cervical carcinogenesis. There should be 23(8) combinations in DNA256-cDNA266-cDNA341, but only 5 haplotypes were detected because of the linkage disequilibrium.
Results of the linkage disequilibrium analysis on rs17880292-rs1059586 and rs17879702-rs17882525.
DNA256-cDNA266-cDNA341 haplotypes correlation analysis between case-control groups.
Discussion
Most studies regarding the relationship between HLA-DRB alleles and cervical cancer have focused on the genotype, such as DRB1*0301, DRB1*1301, DRB1*1101. In our early study, we found a significant tendency toward a positive association between DRB1*1101 and cervical cancer, which was consistent with that in earlier reports[6,7]. It is reported that DRB1*11 is associated with an increased risk of cervical cancer. DRB1*1101-DQB1*0301 among Senegalese[8] and Swedish women[9], DRB1*0301-DQB1*02 in HPV16-infected women[6] have been found to be associated with an increased risk of cervical cancer. DRB1*070101-DQB1*0201[10], DRB1*150101-DQB1*0602[11], and DQB1*0601[12,13] have a similar effect. It was reported previously that DRB1*13 alleles was associated with a decreased risk of cervical cancer in Spanish[14], French[15], Senegalese[8], German[16], and Mexican women[17]. In addition, a decreased frequency of DRB1*1301 has also been observed among the Costa Rican population with cervical cancer[18].
In this study, we examined the coding region in HLA-DRB1. In comparison to the case-control group, 4 SNPs had significant differences in distribution. Rs17880292 (P = 0.033, OR = 0.322) and rs1059586 (P = 0.029, OR = 2.657) showed positive significance for the risk of cervical tumor genesis, while rs17879702 (P = 0.016, OR = 0.222) and rs17882525 (P = 0.025, OR = 0.128) were negative.
SNPs may fall within the coding sequences of genes, the non-coding regions of genes, or in the intergenic regions between genes. SNPs that are in protein-coding regions could lead to changes of peptide type[19]. A great amount of the variability in the immune response in different individuals is caused by the high degree of genetic variability in immune-related genes, such as the HLA genes[20]. As HLA antigen molecules in the sequence of groove-based sequences is encoded by each allele, polymorphisms of HLA lead to significant differences in the combination of antigens and conformations of the groove, and present the antigen to T cells, which induce different susceptibilities determined by different HLA genotypes to cervical cancer. Recently HLA has also been considered to be an important factor or auxiliary factor in the pathogenesis of the tumor [21].
Haplotype is a combination of alleles at multiple loci that are transmitted together on the same chromosome. Most of the chromosomal regions only have a few common haplotypes, each with a frequency of at least 5%, which represents the majority of polymorphisms among groups of people.
A chromosome region contains many SNP sites, but only a few of the SNPs provide most of the patterns of genetic polymorphism. So haplotype tag sites are extremely useful for correlation analysis[22-24]. In this study, 5 types of cDNA256-cDNA266-cDNA341 haplotypes were examined, and the distributions in the cervical cancer group were different from those in the case-control group. The combination of haplotype cDNA256A-cDNA266A-cDNA341T had a higher risk of morbidity in CC patients (OR = 2.735). As a DNA tag, the main genetic parameters of SNPs are polymorphism information content and heterozygosity. PIC value can be improved as several SNP adjacent sites combined[25]. The haplotype cDNA256A-cDNA266A-cDNA341T is more meaningful than a single cDNA256A or cDNA266A in predicting cervical cancer. Statistical analysis has suggested that the 5582 A-5592A-5667T haplotype is associated with cervical cancer, and it could be used to assess susceptibility for high risk people.
In conclusion, the G/A genotype in rs17880292, the A/A (60Ser/Ser) genotype in rs1059586 and the 582 A-5592A-5667T haplotype are potential susceptibility factors for developing cervical cancer. Further studies should be focused on gene function in HLA-DRB1, the possible pathways, and the molecular mechanisms involved in the pathogenesis of cervical cancer.
Conflict of interest statement
No potential conflicts of interest were disclosed.
- Received September 30, 2009.
- Accepted January 29, 2010.
- Copyright © 2010 by Tianjin Medical University Cancer Institute & Hospital and Springer