keywords
Introduction
Tumor lysis syndrome (TLS) is a potentially lethal emergency caused by lysed tumor cells, and it frequently occurs in tumors of hematologic origin. Up until now, there has been only one known report published overseas about TLS resulting from post-transplant lymphoproliferative disorder (PTLD)[1]. In 2008, a PTLD patient who had previously undergone renal transplantation was treated in the renal transplantation center of our hospital, and TLS occurred during treatment for PTLD. At the time, no further literature regarding the treatment of post-transplant renal inadequacy complicating TLS was available. So, continuous veno-venous hemofiltration (CVVH) was used to effectively control the progression of TLS. In this article, the experiences gained from our management of this complex case and our analysis, supported by relevant literature, are described.
Case Report
A 36 year-old, male with a history of renal transplantation (RT), performed 12 years previously, was admitted to our hospital in January 2008 for evaluation and treatment for newly discovered an abdominal mass. In 1998, the patient underwent RT owing to uremia, and was subsequently treated with a regimen of cyclosporine A/azathioprine/prednisone for anti-rejection by oral administration. Laboratory studies conducted 7 years ago indicated that the patient’s creatinine levels were fluctuant and ranged from 180 to 200 μmol/L. The abdominal mass was found 2 months before hospitalization, and it caused a discontinuous, vague pain, perceived after having a meal. A CT scan indicated a space-occupying lesion in the mesentery of the abdomen. The biopsy of the mass through paracentesis was analyzed using immunohistochemical studies which showed CD20 (+), CD79α (+), CD3 (-), ALK (-), CD30 (-), EMA (-), CK (-), bcl-2 (-), and 50% Ki-67 reactivity. Pathologic examination showed a non-Hodgkin lymphoma (diffuse large B-cell type), and the final diagnoses were as follows: i) lymphoproliferative disorder after RT, ii) post-transplant chronic rejection and RT-related renal inadequacy. After the treatment including antirejection drugs, rituximab, and regional radiotherapy, the growth of the abdominal mass slowed significantly. However, the patient developed pain and swelling of his right inner malleolus during treatment. Blood chemical analysis indicated that the blood uric acid (BUA) value had increased (713.7 μmol/L), and that serum electrolytes were normal. After administration of allopurinol and sodium bicarbonate, the BUA decreased, and the pain and swelling disappeared. In May 2008, the abdominal mass was no longer detectable.
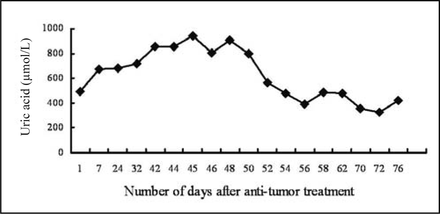
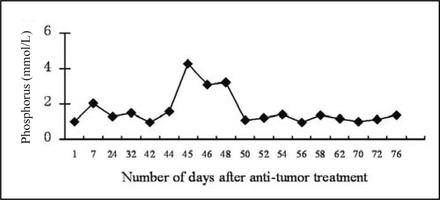
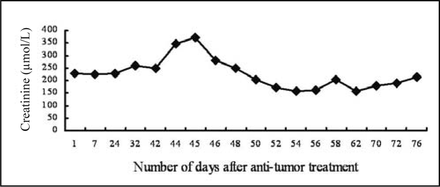
Three months later, the abdominal mass reappeared with progressive growth. The patient was also found positive for fecal occult blood and developed a bowel obstruction. A CT scan showed that there was an enlarged lymph node behind the uncinate process of the head of the pancreas, which indicated recurrent PTLD. Blood biochemical analysis showed the following: BUN 12.98 mmol/L, serum creatinine (SCre) 227 μmol/L, BUA 490 μmol/L, LDH 4020 U/L, K 4.1 mmol/L, TCO2 24.90 mmol/L, Ca 2.22 mmol/L and P 1.00 mmol/L. Hydration therapy was given while cyclophosphamide was administered through intravenous drip. The subsequent blood chemistries on the 7th day after surgery showed that the BUA was 672 μmol/L and P was 2.05 mmol/L. In consideration of the potential for developing TLS, intravenous sodium bicarbonate was administered, with allopurinol at a dose of l0.3g/d. Interferon-α, rituximab, CHOP regimen and regional radiotherapy were also administered in combination. The BUA and P levels increased progressively (Figs. 1, 2), and the patient’s renal function deteriorated (Fig. 3). Forty-five days after the patient’s hospital admission, a recheck of the blood chemistries showed the following: BUN 23.74 mmol/L, SCre 371 μmol/L, BUA 941 μmol/L and P 4.3 mmol/L. CVVH treatment was then performed. Vascular access was established by a femoral vein 2-way catheter, and an AQUARIUS hemofilter and Fresenius Ultraflux AV600S blood filter (polysulfone membrane, with an area of 1.4 m2) were used. Low molecular weight heparin calcium anticoagulation was utilized, with an initial dose of 3,000 iu, and a bolus was given when needed. A pharmaceutical formulation of modified displacement fluid (each bag included: a 3500 ml of 0.9% sodium chloride injection, 500 ml of 5% glucose, 30 ml of 10% calcium gluconate, 3.2ml of 25% magnesium sulfate, 12 ml of 10% potassium chloride and 250 ml of 5% sodium bicarbonate) was used. For the treatment, microregulation of the compounding was done based on the electrolyte content of the patient’s blood. Blood flow was 200 ml/min, the flow rate of displacement liquid 2 L/h, and the volume of displacement was 12-16 L each time. Indicators such as renal function and serum electrolytes were checked before and after CVVH. SPSS13.0 was used for statistical analysis, and the matched t test was used to compare the BUA, P, BUN and SCre values before and after CVVH.
Trend-graph of BUA variance.
Trend-graph of P-value variance in the blood.
Trend-graph of SCre variance.
No complications occurred during CVVH, and BUA, P, BUN and SCre levels decreased significantly following treatment (P < 0.05, especially BUA, BUN and SCre, P < 0.001) (Table 1). After CVVH was initiated, the patient’s BUA and P levels started to trend downward (Figs. 1, 2), while the patient’s renal function improved and stabilized (Fig. 3). Throughout the course of disease, no other TLS-related complications occurred. Two months after treatment, the patient died of perforation of the bowel secondary to abdominal metastasis.
Values of BUA, P and SCre (mean ± SD) before and after CVVH.
Discussion
Tumor lysis syndrome (TLS) was first reported by Crittenden and Ackerman in 1977[2], and denoted a series of metabolic disturbances and homologous syndromes resulting from massive tumor cytolysis and oncolysis which caused the release of the intracellular metabolites into the blood and exceeded the body’s ability for self-clearance. The clinical features of TLS include hyperuricacidemia, hyperkalemia, hyperphosphatemia, hypocalcemia, acidosis and renal failure (RF), among others[3]. TLS is common in neoplastic hematologic disorders, such as acute lymphoblastic leukemia, Burkitt lymphoma and non-Hodgkin lymphoma[4], as well as in solid tumors, such as small cell lung cancer, colon and breast cancer[5]. The pathologic diagnosis of the case in this study was non-Hodgkin lymphoma, which can readily lead to TLS.
The risk factors for TLS are as follows: large tumor load, rapid tumor cell proliferation, hypersensitivity to chemotherapy, rise of LDH, osseous metastasis, and impairment of renal function[6-8]. With the development of anticancer drugs, the incidence rate of TLS has increased. With the exception of chemotherapeutic agents, treatments such as rituximab chemotherapy, radiotherapy, interferon-a and endocrine therapy, all may induce TLS[9]. In our case, the patient developed chronic rejection resulting in post-transplant renal insufficiency, and developed a large tumor load and increased LDH following tumor recurrence. Thus, this case had a high risk for TLS.
For guiding the diagnosis and treatment of TLS, Hande and Garrow[10] classified TLS as laboratory TLS (LTLS) and clinical TLS (CTLS) in 1993. In 2004, Cairo and Bishop[11] improved the Hande-Garrow classification method in order to further meet the clinical requirements. It included the following: i) LTLS was defined as 2 or more of the following symptoms occurring within 7 days before the chemotherapy: blood uric acid of ≥ 476 μmol/L or a 25% higher than it before; potassium of ≥ 6.0 mmol/L or a 25% higher than it before; phosphorous of ≥ 1.45 mmol/L (adult) or 25% higher than it before; blood calcium of ≤ 1.75 mmol/L or a 25% lower than before; ii) CTLS was defined as LTLS plus any one or more of the following: SCre value equivalent to or 1.5 times higher than the normal SCre level; cardiac arrhythmia/sudden death; epileptic episodes. They believed that only conservative treatment was needed in treating LTLS, while specific treatments such as blood purification would be the first choice for CLTS[10,11]. In this case, hyperuricacidemia, hyperphosphatemia and deterioration of renal function occurred, which was in accordance with the diagnostic criteria of CTLS. Since the patient developed post-transplant renal insufficiency, the oncolysis resulted in a release of large amount of potassium, phosphorus, as well as purines and nucleic acids. The latter were transformed into uric acid in the body, and then a large quantity of uric acids was crystallized in the renal tubule, bringing about a uric acid nephropathy. The sedimentation of calcium and phosphorus in the renal tubule resulted in a stromal inflammatory reaction, which could serve to aggravate the RF[9]. Therefore, active removal of tumor-related metabolites may interrupt the progression of TLS.
CVVH is a method of blood purification frequently seen in clinical, and usually used as a salvage procedure for patients in critical condition, such as acute renal failure, septicemia, and severe pancreatitis. Characteristically, it promotes hemodynamic stability and continuity because of the slow removal of the internal metabolites, in comparison to the intermittent nature of hemodialysis. Thus, it can be well tolerated by patients and is suitable for treating TLS. Previous studies have confirmed the therapeutic value of CVVH on TLS. Suzanne et al.[12] carried out a study of 5 pediatric patients with leukemia and Burkitt lymphoma, complicating high-risk TLS. CVVH was performed in the patients before chemotherapy, and BUA levels decreased significantly in all of the pediatric patients after CVVH. Acute RF did not develop in 4 of the 5 patients, although the acute RF that did occur in 1 patient could be attributed to an interruption of CVVH owing to a blockage of the hemofilter. This patient’s renal function returned to normal after CVVH. Choi et al.[13] retrospectively analyzed the data of 11 adult leukemia and Burkitt lymphoma patients with a poor outcome predicted after prognostic evaluation (IPI > 3). The appropriate chemotherapeutic regimen was given in combination with concurrent CVVH. The results showed that after this treatment, BUA and P levels clearly decreased, and that TLS-related fatal complications did not develop in these patients. After 1 year follow up, the survival rate of the patients reached 82%. This conclusion suggests that chemotherapy combined with CVVH is a safe and effective method for treating tumors with a high risk of developing TLS. Regarding this case, complicated TLS occurred after RT. After initiating CVVH, BUA and P levels significantly decreased, suggesting that CVVH could effectively eliminate the metabolites of oncolysis and could interrupt the progression of TLS. This is in conformity with the previous report in the literature[12,13]. Further, CVVH can act as a substitute for renal functions and correct electrolyte disturbances, thus helping the patient to survive high-risk period during TLS.
Nakamura et al.[14] also found that the level of the intra-corporeal cell factors in the patient with TLS (α-TNF, IL-6, IL-8 and IL-10) was apparently higher than that in the healthy population, suggesting t]hat cell factors may participate in the occurrence and progression of TLS. After CVVH, all of the cell factors were markedly decreased. It can be seen that the role of CVVH in absorbing and removing internal cytokines and in lessening systemic inflammatory reactions has been sufficiently delineated[15,16].
On the whole, CVVH is a satisfactory method for treating TLS. After RT, patients may face the high-risk of developing complicated TLS, or sometimes may develop CTLS. Therefore, antitumor treatment should be conducted concurrently with CVVH in order to effectuate tumor cell death and, simultaneously, to quickly remove tumor cell metabolites. As a result, the chance of inflammatory reactions and secondary complications such as RF may be effectively reduced. During CVVH, monitoring indices, including serum electrolytes, hemagglutination and other laboratory values, is necessary, so as to adjust the treatment in sufficient time to avoid excessive anticoagulation. In addition, there is an increasing loss of amino acids and minor elements during prolonged treatment, so timely supplementation of these elements is needed.
- Received August 20, 2009.
- Accepted October 9, 2009.
- Copyright © 2009 by Tianjin Medical University Cancer Institute & Hospital and Springer