Abstract
OBJECTIVE To investigate whether small hairpin RNA (shRNA) targeting Bcl-2 mRNA could inhibit the growth of lymphoma transplanted subcutaneously in nude mice.
METHODS Recombinant Bcl-2 shRNA expression vector with green fluorescence protein (GFP) gene was constructed and preserved in our lab. We evaluated the antitumor effect of the Bcl-2 shRNA in vivo which was the model of nude mice bearing Raji cells xenografts. Human Raji cells were injected subcutaneously into nude mice to establish lymphoma models. When the diameters of tumor were above 0.5 cm after Raji cells injection, the mice bearing tumor were randomly divided into four groups: saline control group, negative shRNA group, plasmid vector group, Bcl-2 shRNA group. The polyethylenimine (PEI) was used to transfect shRNA into tumor. The mixed PEI and shRNA was injected into tumors. The growth and size of tumor were observed. Tissue was stained by H&E for its pathological morphology. The expression of Bcl-2 mRNA in the tumor mass was detected by reverse transcription polymerase chain reaction (RT-PCR).
RESULTS A significant difference in median tumor weight was observed in mice treated with Bcl-2 shRNA, compared with those in the groups of negative shRNA or plasmid vector or saline solution (P< 0.05). Pathological evaluation was completed in all excised tumors from nude mice bearing Raji cells xenografts. The tumor tissue of the mice treated with Bcl-2 shRNA showed apoptosis, serious necrosis of the cells and inflammatory cells infiltration. There was no change in the morphology of cells among negative shRNA, plasmid vector and saline solution group. In the group of the Bcl-2 shRNA, the expression levels of Bcl-2 mRNA of the tumor tissue were effectively inhibited (P < 0.05).
CONCLUSION The shRNA targeting at the Bcl-2 mRNA could inhibit the growth of human lymphoma transplanted subcutaneously in nude mice.
keywords
Introduction
Double-stranded, RNA-dependent, posttranscriptional gene silencing, also known as RNA interference (RNAi), is a phenomenon that double-stranded small interfering RNA (siRNA) complexes can target specific genes. A 20-nucleotide acid siRNA is long enough to induce RNAi[1,2]. Expression of Bcl-2 protein has been observed in the majority of human cancer specimens and cell lines. The Bcl-2 protein regulates the mitochondria-mediated apoptosis pathway, and various cell death stimuli, including chemotherapeutic agents, activate caspases by this pathway, thereby promote apoptosis. A high level of expression of Bcl-2 protein is associated with resistance to radiation and chemotherapeutic agents in a number of types of tumor, therefore a drug to reduce the levels of this protein would be expected to promote apoptosis and be considered a promising chemotherapeutic agent[3,4]. At present, siRNA can be synthesized via chemical synthesis and in vitro transcription. Shortcomings of these two methods are the cost and the stability problem. Stable gene repression can also be achieved in mammalian cells by using vectors to express small hairpin RNA with a U6 or H1 promoter under the direction of RNA polymerase III[5-7]. In our lab, we have identified a siRNA targeting against Bcl-2 mRNA using T7 in vitro transcription could effectively down-regulate Bcl-2 protein. A U6 promoter based vector was used to express Bcl-2 shRNA structure[8-10]. The cationic polymer polyethylenime (PEI) easily outclasses other lipid- or polymer-based vectors[11]. We thus chose to use PEI vectorization to transfect shRNA into tumor. In this study, we further investigated the inhibitory effects of Bcl-2 shRNA on tumor growth of human lymphoma transplanted subcutaneously in nude mice, which might be helpful in developing new anti-tumor drugs. Our data show that the shRNA targeting at Bcl-2 mRNA could suppress the growth of transplanted human lymphoma in nude mice.
Materials and Methods
Reagents
In our lab, an effective siRNA sequence targeting against Bcl-2 mRNA was identified by synthesis of transcription in vitro. Bcl-2 shRNA expression plasmid and a negative control hairpin RNA which encodes a hairpin structure were constructed[9]. RPMI-1640 and newborn calf serum was purchased from Gibco BRL.
Cell culture
The Raji cell line was purchased from the Shanghai Cell Bank. The cells were cultured in RPMI medium added with 10% heat-inactivated fetal calf serum at 37°C, 5% CO2 and then kept in a humidified incubator.
Animals
BALB/C nude (nu/nu) mice (female, 4~6 weeks of age) were purchased from First Military Medical University and weighing18~22 g at the start of the experiments. The mice were housed in filter-topped cages in which a sterile condition was maintained. All animal experiments were conducted in accordance with the principles and procedures described in Guidelines for care and use of experimental animals.
In vivo tumor-inhibitory experiments
Twenty-five nude mice were injected subcutaneously with Raji (1 × 107) cells. Treatment was initiated when the tumor size reached 0.5~1 cm in diameter. Twenty tumor-bearing nude mice were divided into 4 groups randomly: Bcl-2shRNA group, negative shRNA group, the plasmide Pgenesil-1group, saline control group, respectively. DNA was compounded with PEI 22 kDa in a 5N/P charge ratio (N = the positive charges carried by protonated amines of PEI, P = the negative charges carried by DNA phosphates). All solutions were prepared in 5% glucose. Bcl-2shRNA was administered locally into tumor body once a day. The animals were observed for the diameters and the growth of their tumor. After thirty days, the animals were sacrificed and their tumors were excised. Tumor masses were dissected and each of their weight was measured. The inhibition rates of growth of xenografted tumors were calculated according to the formula: inhibition rate (%) = 1− mean weight (treatment group)/mean weight(saline group) × 100%.
Histological study
Tumor-bearing animals were sacrificed and autopsied on the indicated day. Tumors were removed and fixed in 10% phosphate-buffered formalin and embedded in paraffin. Sections of 4 μm were stained with H&E for microscopic evaluation and examined by a pathologist.
Assay of Bcl-2 mRNA by RT-PCR
Total RNA was isolated using TRIzol isolation system (Invitrogen Corporation, USA). One microgram of total RNA was reversely transcribed into first-strand cDNA by using cDNA synthesis kit (Promega, CA). PCR reaction was carried out with a final volume of 50 μl for each reaction containing specific primers targeting at human Bcl-2 mRNA (forward: 5′-CGA CGA CTT CTC CCG CCG CTA CCG C-3′; reverse: 5′-CCG CAT GCT GGG GCC GTA CAG TTC C-3′) and human glyceraldehyde-3-phosphate dehydrogenase (GAPDH, forward: 5′-GTC AGT GGT GGA CCT GAC CT-3′, reverse: 5′-TGA GGA GGG GAG ATT CAG TG-3′). GAPDH was used as an internal control. At 95°C for 3 min, the experimental reaction consists of 30 cycles of 95°C for 30 s, 60°C for 60 s, 72°C for 60 s. The PCR products were detected by gel electrophoresis. The relative expression of Bcl-2 was quantitatively analyzed using gene expression analysis software.
Statistical analysis
Data were expressed as mean ± SD and analyzed using ANOVA. And a level of P < 0.05 was considered significant.
Results
Therapeutic efficacy of the Bcl-2 shRNA
We evaluated the antitumor effect of the Bcl-2 shRNA in vivo which was the model of nude mice bearing Raji cells xenografts. Seven days after tumor cell injection, 4 groups were randomly selected to receive different treatments including saline, negative shRNA, plasmid vector, Bcl-2 shRNA. After 30 days, the animals were sacrificed, and their tumors were excised. Median tumor weight was (2.611 ± 0.023) g for saline solution-treated mice, (2.592 ± 0.090) g for the negative shRNA-treated mice, and a decreased number of (2.001 ± 0.633) g for the Bcl-2 shRNA-treated mice (Table 1). A significant difference in tumor weight was observed between the group of mice treated with Bcl-2 shRNA, and the groups of negative shRNA, plasmid vector and saline solution group (P < 0.05). The inhibitory rate of Bcl-2 shRNA on tumor growth was 23.36%. Bcl-2 shRNA significantly suppressed tumor growth.
Median tumor weight and inhibitory rate in excised tumors (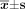 , n = 5).
, n = 5).
Pathological evaluation
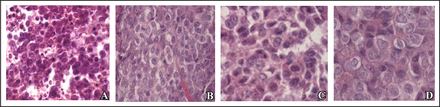
The effect of treatment with Bcl-2 shRNA was evaluated based on the tumor histology. Pathological evaluation was completed in all excised tumors. As shown in Fig. 1, tumor tissue of the mice treated with Bcl-2 shRNA showed apoptosis, serious necrosis and inflammatory cells infiltration. There was no change in the morphology of cells among groups of negative shRNA, plasmid vector and saline solution
Effects of treatment with Bcl-2 shRNA on the tumor histology of Raji cells transplanted into nude mice (H&E stain, × 400). A, Bcl-2 shRNA; B, Plasmid vector; C, Negative shRNA; D, Saline solution.
Inhibition of Bcl-2 shRNA on Bcl-2 mRNA in the tumor tissue
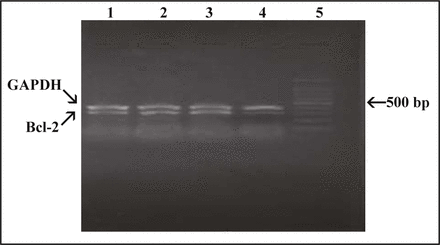
The Bcl-2 and GAPDH genes are 318 and 400 bp, respectively. RT-PCR data shown include at least 3 independent experiments. As shown in Fig. 2, in the group of the tumor tissue treated with Bcl-2 shRNA vector, mRNA expression ratio (Bcl-2/GAPDH) was 0.742 ± 0.006; and in the groups of tumor treated with the negative shRNA, plasmid vector and saline solution, mRNA expression ratio (Bcl-2/GAPDH) was 0.970 ± 0.01, 0.972 ± 0.008, 0.996 ± 0.002, respectivily. Compared with negative shRNA, plasmid vector and saline solution groups, Bcl-2 mRNA expression from Bcl-2 shRNA group was significantly reduced (P < 0.05), whereas the shRNA control and pgenesil-1 plasmid vector had no effect.
Expression of Bcl-2 mRNA in excised tumors by RT-PC. 1, Saline solution; 2, Negative shRNA; 3, Plasmid vector; 4, Bcl-2 shRNA; 5, 100bp DNA ladder Mark.
Discussion
Bcl-2 protein is a prominent member of the Bcl-2 family of proteins that regulate the induction of apoptosis by a wide variety of stimuli. Increased tumor expression of Bcl-2 is considered the main factor among the major determinants of resistance to chemotherapeutic drugs and radiotherapy[3,4]. Blocking the expression and function of the Bcl-2 may represent a relevant therapeutic strategy, and Bcl-2 antisense oligonucleotides used in several preclinical and clinical studies have been successful. The currently ongoing clinical studies in phase I~II using antisense Bcl-2, known as G3139, aim at the treatment of patients with non-Hodgkin’s lymphoma, melanoma, and prostate cancer[12-14].
Our previous studies showed that reduced Bcl-2 mRNA by antisense oligonucleotides can inhibit tumor formation and progression directly, and the growth of human lung carcinoma transplanted subcutaneously in nude mice[15]. Raji (Human Burkitt’s lymphoma cell line) has been previously shown to produce large amounts of Bcl-2[16].
In this study, we have also demonstrated the antitumor effect of Bcl-2 shRNA in treatment. After 30 days, Bcl-2 shRNA group presented the inhibition of tumor growth at the mean of 23.36%, compared with that in the group of control shRNA-treated mice. The control shRNA was ineffective. The results in our study have demonstrated that the Bcl-2 shRNA inhibits the growth of tumor in sequence-dependent way. Other previous studies have shown that downregulation of Bcl-2 expression inhibited the growth of tumors in mice[3,4]. In this study, we have also observed the apoptosis, serious necrosis and inflammatory cells infiltration in tumor tissue with Bcl-2 shRNA treated. Finally, as expected, in the tumor tissue of those mice treated with Bcl-2 shRNA vector, Bcl-2 mRNA expression has showed a greater decrease compared with that in the tumor tissue of the control shRNA. Our experimental evidence, as previously reported[3,4,8], also implies that the selective blockade of Bcl-2 could trigger the apoptosis and suppress the transduction of survival signals, thus leading to the inhibition of the cancer cell growth. Our data indicate that down-regulation of the Bcl-2 mRNA by Bcl-2 shRNA inhibit the growth of tumors in mice, and that Bcl-2 shRNA may be useful for the treatment of tumor with overexpression of Bcl-2.
Footnotes
This work was supported by a grant from Natural Science Program Foundation of Guangdong Province, China (No. 04010446).
- Revision received August 2, 2008.
- Accepted January 7, 2009.
- Copyright © 2009 by Tianjin Medical University Cancer Institute & Hospital and Springer