Abstract
OBJECTIVE To explore the inhibition of ACHN cells via shRNA expression vector mediated cyclinE1 gene silencing.
METHODS The shRNA targeting at cyclinE1 gene was designed and synthesized. By ligation, the fragment was inserted into pGenesil-1-U6 to construct the recombinant plasmid pGenesil-1-U6-cyclinE1. The identified recombinant plasmid was introduced into ACHN cells with lipofectamine 2000. The inhibition of cyclinE1 mRNA and protein expression were analyzed by RT-PCR and western-blotting. MTT method was used for observing cell proliferation and drawing growth curve. The cell cycle and ratios of apoptotic cell were assessed by flow cytometric detection. The ability of invasion and speed of cell migration were detected by transwell chamber invasive models and cell scratch method.
RESULTS The inhibition of expression of cyclinE1 in ACHN cells mediated by recombinant vector (0.0933 ± 0.05) was significantly lower than that in the group of transfected with empty vector (0.8827 ± 0.04) and the control group (0.9021 ± 0.03) (P < 0.05). Flow cytometry showed that recombinant cells were blocked in the G1 phase and the apoptotic ratio was increased significantly (11.15 ± 4.00)% (P < 0.05). The curves of cell growth indicated that the proliferation of cell transfected with recombinant plasmid was inhibited significantly compared with that in control group (P < 0.05). The results of transwell and cell scratch suggested that the abilities of invasion and migration of the cells transfected with recombinant plasmid were decreased conspicuously (P < 0.05).
CONCLUSION The expression of cyclinE1 could be inhibited successfully by RNA interference induced by shRNA expression vector. This consequently inhibits the cell growth and induces apoptosis. Our study provided a preliminary result in searching of RNA interference (RNAi) therapy for renal cell carcinoma.
keywords
Introduction
Renal cell carcinoma (RCC), also known as renal adenocarcinoma, accounts for 3% of all adult tumors. RCC is the most common kidney tumor in adult and also is the most fatal one in uropoietic system. Studies have suggested that cyclinE1 in many tumors, including renal cell carcinoma, displayed over-expression which was closely related to the prognosis. Then, cyclinE1 could become a new target to tumor gene therapy. RNA interference (RNAi) is a kind of gene silencing technology which can block specific gene expression with small fragment of RNA in vivo. This technology has been widely used in treatment of the virus, cancer and the research of other diseases. The purpose of our study, through the shRNA expressing vector-mediated cyclinE1 gene silencing technology, is to observe the changes of proliferation and invasion of ACHN cells after interference. At the same time, it can provide a theoretical basis for genetic therapy of kidney cancer.
Materials and Methods
Materials
Human ACHN cell lines were purchased from Typical Species Preservation Center of Wuhan University and pGenesil-1-U6 plasmid came from GeneSil Biotech Co. (Wuhan, China). Trizol and Lipofectamine 2000 were products of Invitrogen Co. (USA). Fetal bovine serum and MEM medium were provided by Gibco Co (USA). RT-PCR and related products of purification kits were provided by Promega Co. The polyclonal antibody of rabbit anti-human cyclinE1 and HRP labeled goat anti-rabbit IgG was the products of Santa Cruz Co. MTT and Propidium Iodide (PI) were Sigma’s products. Primers of cyclinE1 and 3-phosphorylation glycerol dehydrogenase (GAPDH) were synthesized by Invitrogen Co. Restriction endonuclease and T4 ligase were purchased from Jingme Biotech Co. Reconstruction of the basement membrane material (Matrigel plastic products) was from BD Co. and transwell chamber was product of Costar Co.
Design and synthesis of shRNA
Homo cyclinE1 gene sequence was from GenBank (accession No. NM-001238). Interference sites were designed to target at 454-472 bp site of cyclinE1 using the intersection of design results from Ambion Co. and Dharmacon Co., the target sequence as the following: ACGTGCAAGCCTCGGATTA. A BLAST search was carried out to eliminate the possibility of expressed sequence tag (EST) of homology with other coding sequence.
The Construction and identification of recombinant pGenesil-1-cyclinE1
The terminal of shRNA template was designed with Bam HI, Hind III at the restricted site (Table 1). After annealed, the two single-stranded DNA template (Genesil synthesis) transformed into double-strands DNA templates which were connected with pGenesil-1-U6 empty vector. Plasmid was amplified, purified, digested by enzyme and ranked to ensure the accurate recombinant plasmid (Songon Co.).
Design and expected product of human cyclinE1 gene-specific shRNA template.
Cell culture, transfection and establishment of stable cell lines
The ACHN cells were cultivated in MEM medium supplemented with 10% fetal bovine serum and maintained at 37°C, 5% CO2 in the incubator with constant temperature. Twenty four hours before transfection, the ACHN cells were inoculated to 24-well plates (1 × 105 cells/well). The ACHN cells were transfected with Lipofectimine 2000. The cells were divided into 3 groups, namely the interference group (shRNA group), empty vector group and blank control group (non-transfected group). After 48 h, cells were inoculated into 96-well plate with limiting dilution assay. Then the successfully transfected single cell was selected (carrying EGFP gene) and amplified continuously. After 20 generations, fluorescence has not been lost (Fig. 1).
Cells observed with optical microscope (A) and fluoroscope (B) respectively after the passage 20 times (× 400).
Semi-quantitative RT-PCR
Cells were collected from 6-well plate and then total RNA was extracted. The primers for cyclin E1 were 5’-CTG GAT GTT GAC TGC CTT GA-3’ (upstream) and 5’-TCT TTG GTG GAG AAG GAT GGG GTG G-3’ (downstream) and those for GAPDH were 5’-CCA CCC ATG GCA AAT TCC ATG GCA-3′ (upstream) and 5′-TCT AGA CGG CGA GTC AGG TCC ACG-3′ (downstream). The mixture of RNA and primers were loaded in the PCR amplifier with predenaturate setting at 95°C for 5 min, 94°C for 45 s, annealing at 50°C for 1 min and extension at 72°C for 1 min. The procedures were carried out 30 cycles, followed by a final extension at 72°C for 10 min. The PCR product then underwent 1.5% of agarose gel electrophoresis. The result was scanned using UV spectrophotometer and quantitatively analyzed with LabWorks 4.5 analysis software.
Western blotting analysis of the expression of cyclin E1
An equal number of cells were collected and were cracked by cell lysis. The content of protein in the supernatant liquid was detected with BCA method. Equal weight of proteins was used for SDS-PAGE electrophoresis and transferred to PVDF membrane (Bio-Rad Semi-to-film, 24 V, 24 min) which was incubated for 2 h with rabbit anti-cyclin E1 (1:500), and then incubated for another 2 h with di-antibody (1:2000). Finally, after colored and fixed, the film was analyzed with Image Analysis System.
Flow cytometry for cell cycle and apoptosis index
The logarithmic phase cells were fixed with 75% alcohol overnight and washed 3 times with PBS, then mixed with RNase A (10mg/L) and kept at 37°C for 30 min. The mixture was stained by PI (50 mg/l) for 30 min. The amount of cells was adjusted to 1 × 106/ml followed by flow cytometry analysis which used 200-mesh nylon nets for the procedure.
MTT used for observing cell proliferation and drawing growth curve
Cell suspension was inoculated into 96-well plates (1 × 103/well) and kept for 10 days, daily sampling was taken and incubated with 20 μl of MTT for 4 h. After centrifugation, the precipitate was added with 150 μl of DMSO and measured absorbance (A) of wells with enzyme-labeled instrument at the 490-band. The growth curve was drawed, which showed MTT absorbance values.
Transwell used for cell invasion assays
The end membrane of transwell chamber was coated by diluted matrigel and dried at 4°C. Basement membrane was hydrated with serum-free medium including BSA (10 g/L) at 37°C for 30 min. Cell suspension (2 × 105/ml, 200 μl) containing 10% of FBS was added into the upper chamber and 500 μl of medium containing 20% of FBS was added into the lower one. Then, after placed in incubator at 37°C, 5% CO2 for 36 h and 48 h separately, the chambers of two groups were washed with PBS and fixed with Xylene for 10 min, and then stained with trypan blue followed by a final step of rinsing. Cells that migrated through the membrane to the lower surface were counted with light microscope to get the mean of the cells.
Cell scratch test used for observing cell migration
Three groups of cells in logarithmic phase were cultured in 24-well plates and kept until 50% of confluence of cells was present. Then straw was used to scratch and then the width of scratch was measured respectively with micrometer after 24 h, 72 h and 96 h. The formular of migration rate of cells = (1−scratch width after incubation / scratch width before incubation)×100% was used.
Statistical analysis
Statistical analysis was performed using SPSS11.0 statistical software. Significance was determined with 95% confidence.
Results
Construction of recombinant plasmid PGenesil-1-U6-cyclinE1
Recombinant vectors were digested by enzyme. The sequencing results proved that shRNA template at cyclinE1 gene (Genbank accession No. nm_001238) had been inserted into vector successfully.
Analysis of inhibiting the expression of cyclinE1 mRNA
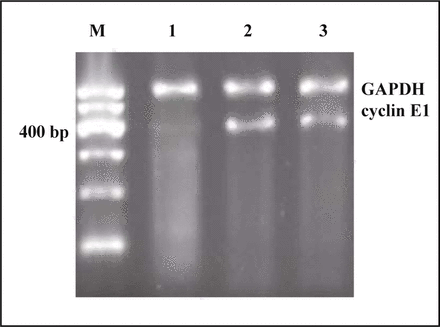
Fig. 2 visualizes the results of RT-PCR in the 4 groups. Three bands of cyclinE1 (400 bp) and one band of GAPDH (600 bp) were found, but the intensity of the bands of cyclinE1 in the interference group was significantly weaker than that in the other two groups. That is to say, the expression of cyclinE1 mRNA in ACHN cells transfected with recombinant plasmid was significantly reduced (P < 0.05). The analysis of UV gel image demonstrated that the inhibition ratio of cyclinE1 expression was 91% which was higher than that in the group without transfection (Table 2, Fig. 2).
Dates analysis of RT-PCR, flow cytometry, transwell (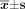 ).
).
Detection of cyclinE1 mRNA by RT-PCR assay at different cells. Lane M, Marker; 1, recombinant group; 2, empty group; 3, non-transfection.
Western blotting analysis of the expression of cyclin E1
The molecular weigh of cyclinE1 protein ranges between (49~51) × 103 (49~51 KD). Fig. 3 shows that the expression of cyclinE1 protein in group transfected with pGenesil-1-U6-cyclinE1 of recombinant plasmid was significantly reduced compared with those in the groups of empty vector and non-transfection.
Western-blotting analysis of expression of cyclinE1. Lane 1, non-transfection; 2, empty group; 3, recombinant group.
The effect of RNA interference on cell cycle and apoptosis
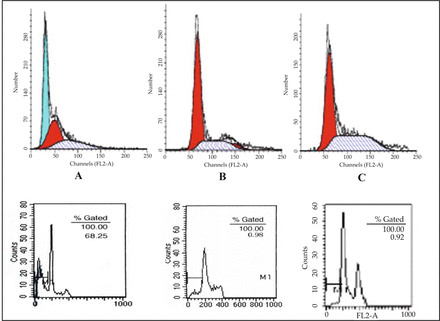
Flow cytometry analysis indicated that the cells in stage S accounted for (8.2 ± 2.6)% in the group of transfected with recombinant plasmid, (27.6 ± 3.3)% and (31.2 ± 2.5)% in the negative and blank control groups, respectively. In the interference group, about (80.8 ± 5.5)% of the cells exhibited cell growth arrest at stage G1 and the proportion of apoptotic cells was increased up to (11.15 ± 4.0)% significantly. The apoptotic ratios of other two groups were (0.98 ± 0.14)% and (0.83 ± 0.15)%, respectively (P < 0.05). The inhibition of cyclinE1 expression can cause cell growth arrest at G1 phase and promote apoptosis (Fig. 4, Table 2).
Detection of the ratios of apoptosis and G1 phase cell. A, cells transfected recombinant vector; B, cells transfected empty vector; C, cells non-transfection, cells passed 20 passages.
The inhibition of cell growth by RNAi
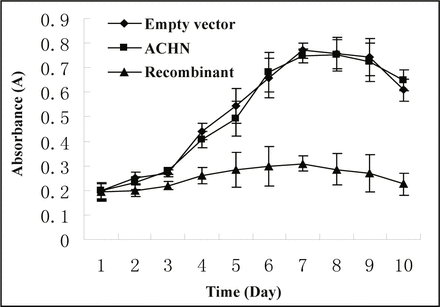
The colorimetric assay was used to get the absorbance (A) of MTT samples in 10 consecutive days. As the absorbance is proportional to the ratio of cell survival, value A was made as ordinate directly and days as abscissa. From the curve of cell growth (Fig. 5), we found that radian of growth curve of the cells in interference group was much lower than those in other two groups and had almost no logarithmic growth phase (P < 0.05). The number of survival cells and growth velocity were relatively lowered in the group of transfected with recombinant plasmid (P< 0.05, Table 3).
Growth curves of cells in different groups.
Data results of MTT ( ).
).
Effect of cyclinE1 inhibition on cell invasion
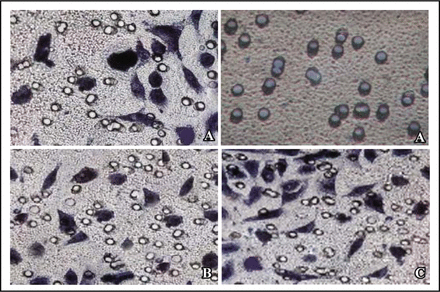
After 36 h and 48 h, the results showed that the count of transmembrane cell was significantly reduced in cyclinE1 gene silencing groups (P < 0.05). In comparison, empty vector and non-transfected groups had an increasing crossing. We concluded that cyclinE1 gene silencing down-regulated the invasion and migration of ACHN cell (Fig. 6, Table 2).
Matrigel invasion assay after trypan blue staining (× 400). A, cells transfeced recombinant vector; B, cells transfected empty vector; C, cells non-transfection.
Mobility of cells
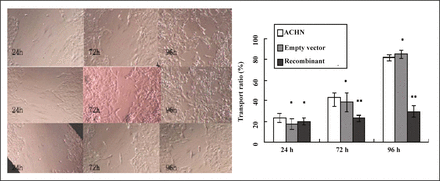
The results of erasion trace in 3 groups showed no significant difference 24 h after transfection and while, at 72 h and 96 h after transfection, the mobility of the cells in the group with recombinant vector was significantly smaller than those in other 2 groups (P < 0.05, Table 4, Fig. 7).
Mobility of cells in scratch test ( ).
).
The migration of different cells observed by scratch assay in 24 h,72 h,96 h: (×400). * P > 0.05: ** P < 0.05 vs. ACHN
Discussion
The regulation of cell cycle is a strict, orderly and complex network activity. Any imbalance of link in cyclin can lead to tumor formation. Cell cycle proteins always play an important role in the regulation of cyclin. CyclinE1 works as a cell cycle key regulating factor to promote cell proliferation in the G1/S phase. CyclinE1 overexpression can shorten G1 phase and cause excessive proliferation of centrosome so that disrupt mitosis and form the instable chromosome. Numerous studies confirmed that cyclinE1 over-expression was closely related to the majority of tumor formation, evolution and even prognosis. So it has become the new target of tumor gene therapy[1-8]. RNAi is an RNA-dependent post-transcriptional gene silencing technology. Currently shRNA molecules have been recognized as the major dsRNA to mediate RNAi in mammalian cells[9]. These molecules usually contain 21~23 of base pairs with 2 or 3 dangling nucleotides at the 3’ end, and when introduced into cells, they can specifically inhibit mRNA expression of its homologous target gene. As a new technology for specific inhibition of target gene expression, it has been widely used in the analysis of gene function, anti-virus and researches of cancer therapy [10,11].
Overexpression of cyclinE1 in renal cell carcinoma is closely related to tumor characteristics such as invasion, metastasis and higher grade of malignancy[12-14]. Previous studies on RNAi for cyclinE1 in renal cell carcinoma were not sufficient. The researchers in the previous studies only used siRNA derived from chemical synthesis, vitro transcription and long fragments of dsRNA degradated by RNase III. Additionally, these studies about inhibitory effect were instantaneous[15-18]. Some researchers also used recombinant vector to study inhibition of target gene expression, however, due to non-screening gene, were unable to establish a stable cell line and only were limited to detect instantaneous effect. To overcome these insufficiencies, in our study, we used pGenesil-1-u6 from Gensil Co., then designed and successfully constructed the human cyclinE1 shRNA expression vector, pGenesil-1-cyclinE1. With U6 promoter driving, recombinant vectors entered into cells, and theoretically, they were able to produce cyclinE1 specific shRNA ending with five U like hairpin. The vector carried enhanced green fluorescent protein gene (EGFP) and neo gene as screening gene.
We can observe the cells transfected successfully through the former one and killed the tumor cells transfected unsuccessfully through using G418. Limiting dilution assay helped us to select ACHN cells transfected, and then we amplified and established stable cells line. In observation, firstly, we found that transfected cells had similar shape and size, and the giant cell, irregular-shaped cells and the cella with pathological caryocinesia were decreased significantly. Secondly, passage of cells with recombinant vector needed longer interval time than those of cells in group of empty vector and in blank control group. Thirdly, through 20 times of passages and recovery after cryopreservation, the fluorescence did not significantly weaken or lose in transfected cells.
Results of RT-PCR showed that, compared with non-transfection, the inhibition rate of the expression of cyclinE1 mRNA could reach up to 91% in the group of ACHN cells with transfected recombinant vector, which was close to the inhibitory effects of using siRNA in liver cancer cell lines, and was also significantly higher than those of using other methods[15-17]. The further research by western-blotting detected that the expression of cyclinE1 protein was significantly decreased. That is, the vector-mediated RNAi to cyclinE1 is specific. FCM results showed that, after cyclinE1 gene silenced, the cells of growth arrest in phase G1 accounted for 80.80% which was significantly higher than those in other 2 groups (empty-vector: 63.58%; non-transfection: 65.36%). On the contrary, the ratio of cells in phase S was decreased to 8.21%. At the same time, the proportion of apoptotic cells reached to 11.15% which was far higher than those in other two groups.
The above results showed that cyclinE1 gene of ACHN cells can be silenced by shRNA expression vector and cyclinE1 gene silencing can indeed block cell growth at phase G1, and then finally induced apoptosis of cells successfully. The curve of cell growth made from MTT further confirmed that inhibiting the expression of cyclinE1 resulted in a slow growth and proliferation, and even without the logarithmic growth phases. In order to inhibit the cell proliferation, the scratch assay was conducted using the cells of the 3 groups, which were cultured with low concentration of serum medium and processed with 10 μg/ml Mitomycin (MMC) for 1 h. As we know, the toxic effects of MMC not only inhibit cell proliferation but also, to some extent, affect the migration ability of cells. Therefore, it explains the reason why there was no significant difference in cell migration in the 3 groups after 24 h. At the same time, we used transwell assay to make up for one shortcomings of scratch assay and to further confirm the conclusion. Similarly, after observing the changes of invasion and interfered migration of ACHN cells, we found that down-regulation to cyclinE1 could significantly decrease the ability of ACHN cells in migration and invasion. It means that cyclinE1 gene plays a very important role in the development of renal cancer.
Our studies have explored the feasibility of introducing RNAi mediated by vector into ACHN cells at cyclinE1 gene and achieved preliminary success. Furthermore, we have established ACHN cell lines transfected stably with pGenesil-1-u6-cyclinE1. Compared with RNAi triggered by siRNA of vitro synthesis, the efficacy of RNAi mediated with vector is more stable and relatively higher. After several times of passages and recovery from cryopreservation, we found by observing the fluorescence that the shRNA to target gene was expressed continuously. Moreover, our research indirectly confirmed that cyclinE1 might play a positive role in regulation of malignant proliferation and invasive growth of RCC and provided a basis for genetic therapy using RNAi targeting to the key gene in caner cells.
Footnotes
This work was supported by a grant from Major State Basic Research Development Program, China (No.2002CB513107).
- Revision received September 11, 2008.
- Accepted January 5, 2009.
- Copyright © 2009 by Tianjin Medical University Cancer Institute & Hospital and Springer