Abstract
OBJECTIVE Various studies examining the relationship between HER-2 over-expression and the response to chemotherapy and clinical outcome in patients with osteosarcoma have yielded inconclusive results. The purpose of the current study was to evaluate the relation of HER-2 status with the response to chemotherapy and clinical outcome in osteosarcoma.
METHODS We conducted a meta-analysis of 6 studies that evaluated the correlation between HER-2 status and histologic response to chemotherapy and 2-year survival. Data were synthesized in summary receiver operating characteristic curves and with summary likelihood ratios (LRs) and relative risk.
RESULTS The quantitative synthesis showed that HER-2 status is not a prognostic factor for the response to chemotherapy. The positive LR was 1.27 (95% confidence interval, 0.91~1.77), and the negative LR was 0.68 (95% confidence interval, 0.38~1.22). There was no significant between-study heterogeneity. HER2-positive status tended to be associated with a worse 2-year survival, but the overall results were not formally statistically significant.
CONCLUSION HER-2 status is not associated with the histologic response to chemotherapy in patients with osteosarcoma, whereas HER-2 positive patients may be associated with decreased survival.
keywords
Introduction
The HER2 (also known as c-erbB2 or neu) proto-oncogene is located at 17q21, and encodes a 185-kDa transmembrane tyrosine kinase glycoprotein with extensive homology to the epidermal growth factor receptor[1-3]. Over-expression of the HER2 gene has been found to correlate with poor prognosis in a variety of human carcinomas, such as breast, ovarian, and lung cancers[4,5]. In some of these tumors, enhanced expression of HER2 has been associated with increased tumorigenicity and metastatic potential[6,7], or with resistance to chemotherapeutic agents[8,9]. Several studies have tried to investigate the clinical significance of HER2 over-expression in osteosarcoma. These studies have yielded conflicting results. In some studies, the increased levels of the HER2 protein at diagnosis resulted in an association with a worse clinical outcome[10-12], whereas other reports either did not show any correlation between HER2 and prognosis[13,14] or indicated a positive prognostic value for increased HER2 expression at diagnosis[15]. Most studies had limited sample size. Thus a quantitative synthesis using rigorous methods would be important to clarify the conflicting results. Accordingly we conducted a meta-analysis of all available studies relating HER2 expression with response to chemotherapy and clinical outcome, as defined by 2-year survival, because all studies had at least 2 years of follow-up.
Materials and Methods
Identification and eligibility of relevant studies
We considered all studies examining the association of HER2 expression with osteosarcoma outcomes. Sources included MEDLINE and EMBASE (last search update 2005). The search strategy was based on combination of “osteosarcoma,” “HER2,” “c-erbB2,” “neu” and “EGFR.” References of retrieved articles were also screened. Investigators were contacted and asked to supply additional data when key information relevant to the meta-analysis was missing.
All studies examining the relation of HER status to the response to chemotherapy and clinical outcome (death) were eligible for our meta-analysis. We accepted the studies measuring HER2 status with the method of immunohistochemistry (IHC) for protein levels. Whenever reports pertained to overlapping patients, we retained only the largest study to avoid duplication of information.
Definition and standardizations
For studies using IHC, we used prespecified rules to standardize, as much as possible, the definition of a positive test for studies that used different cutoff thresholds. We defined HER2 protein positivity as nuclear cell stain in at least 25% of the tumor cells, a definition followed by most studies. When different definitions were used, we accepted the cutoff closest to the 25% level. Whereas in Scott E’classification, we considered as HER2 positive those with 3+ (moderate to strong staining in more than 10% of cells).
We defined “response to chemotherapy” by the percentage of histologic necrosis of tumor cells in speci-mens obtained after chemotherapy. A cutoff of 90% necrosis was used to separate responders from nonresponders. Equivalent cutoffs were used for studies using other classification. Thus, for studies using the Huvos grading system to evaluate histologic necrosis [14], we considered as responders those with a grade 3 or 4 response.
The clinical outcome of interest was mortality. Clinical outcomes were standardized to include 24 month follow-up in all studies to avoid some studies contributing very long-term follow-up data as compared with others.
Data extraction
Two investigators extracted data from eligible studies independently, discussed discrepancies, and reached consensus for all items. We extracted data on characteristics of studies and patients, measurements, and results. In each report we recorded author names, journal and year of publication, country of origin, number of patients analyzed, stage and grade of osteosarcoma, chemotherapy and surgery used, timing of HER2 status measurement (before or after chemotherapy), type of measurement, antibodies used for IHC, definition of positive test, and blinding of measurements to the study outcomes. Data on the main outcomes were entered in 2 × 2 tables showing the histologic response/nonresponse and the occurrence/nonoccurrence of death within 24 months per HER2 status.
Statistical analysis
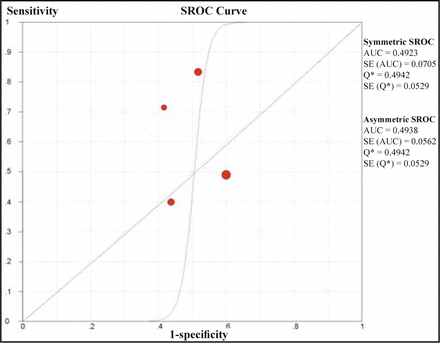
Data on the diagnosis performance of HER2 status for determining histologic response to chemotherapy were evaluated by constructing a summary receiver operating characteristic (SROC) curve and estimating the combined positive and negative likelihood ratio [LR (LR+, LR-)].
For a diagnostic or predictive test, the sensitivity (true-positive rate) and specificity (one minus false-positive rate) are correlated; therefore, it is not correct to estimate these two quantities independently. To bypass this problem, the SROC method may be used. The SROC curve is estimated by the regression D = a + bs, where D is the difference of the logits of the true positive and false-positive rate, and S is the sum of these logits[16]. Both weighted and unweighted regressions were estimated. The SROC curve shows the trade-off between sensitivity and specificity across the included studies.
Likelihood ratios also are metrics that combine both sensitivity and specificity in their calculation. LR+ was defined as the ratio of sensitivity over 1 minus specificity, and LR-was defined as the ratio of 1 minus sensitivity over specificity. When there is absolutely no discriminating ability for a diagnosis or predictive test, both likelihood rations equal 1. Although there is no absolute cutoff level, a good diagnostic test may have LR+ > 5 and LR- < 0.2. Study specific LR values were combined with fixed-effects and random-effects models, and between-study heterogeneity was assessed with the Q statistic[17].
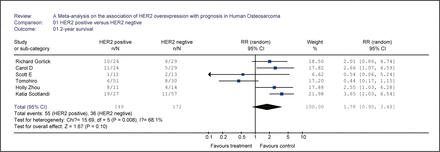
Data on the predictive ability of HER2 over-expression for death were also combined across studies using relative risk for 2-year mortality[17]. Between-study heterogeneity in the relative risk was assessed with the Q statistic[17].
Fixed-effect models assume that differences between the results of the combined studies are due entirely to chance. Random-effect models allow for the possibility that results may differ genuinely between studies. In the presence of between-study heterogeneity, random-effect models provide a wider confidence interval (CI)[18]. We generally present random-effects estimated, unless stated otherwise.
Sensitivity analyses examined the effect of limiting the evaluations studies scoring for membranous staining and studies using the 25% IHC cutoff. We also used appropriate bias diagnostics to examine whether there was evidence that the results differed in small studies compared with large studies[19].
Analyses were conducted with Review Manager 4.2 and Meta-Disc 1.4. For these analyses, P values < 0.05 were considered significant.
Results
Eligible studies
We initially identified 42 reports including the role of HER2 status in patients with osteosarcoma. Of those, 36 reports were excluded due to reviews, cell lines or lack any informative clinical data including 2-year mortality. In all, 6 independent eligible studies, which had data on 2-year survival and/or the response to chemotherapy and enrolled a total of 331 patients, were included in the quantitative synthesis[10-13,15,20]. The mean or median age of patients included in each study ranged from 17 to 29 years across the eligible studies; the populations were young. All analyzed osteosarcoma were treated with combination chemotherapy regimens. Surgery comprised resection, limb salvage, disarticulation, curettage, or amputation procedures.
Six studies used IHC to determine HER2 status [10-13,15,20]; 4 studies used more than 25% cutoff HER2 positivity, whereas different thresholds were used in the remaining reports (Table 1). The cutoff of 90% necrosis to separate responders from nonresponders was used by 3 studies, whereas 2 studies used the Huvos grading system. The incidence of histologic response to chemotherapy ranged from 33% to 58%, 2-year mortality rates ranged between 4% and 48% across the eligible studies. Both the chemotherapy response rates and 2-year death rates differed significantly across studies (P < 0.05 for both). This may be due to differences in the case mix of the population (e.g., grade and stage) and/or the therapies used.
Characteristics of eligible studies.
Data synthesis: response to chemotherapy
HER-2 status had no discriminating ability to identify poor versus good responders to chemotherapy. When all studies were considered, the SROC curve passed very close to the diagonal, suggestive of the total lack of discriminating performance. According to the SROC, a sensitivity of 50% corresponded to a specificity of 48%, and a specificity of 50% corresponded to a sensitivity of 52% in the weighted analysis. Unweighted estimates were similar (Fig.1).
Each study is shown by a circle demonstrating the sensitivity and specificity of HER-2 status for predicating a poor histologic response. The data are summarized with weighted and unweighted SROC.
Separate analyses with studies scoring for membranous staining and studies using the 25% IHC cutoff were similar. In the main analysis and various sensitivity analyses, LR+ remain in the range of 1.27~1.62, and LR-remained in the range of 0.37~0.68, values characteristic of very poor discriminating performance(Table 2). There was no significant between-study heterogeneity for either LR+ or LR-in any of these analyses. There was also no evidence that large studies yielded markedly different results compared with smaller studies, or that early studies differed significantly against later publication.
Likelihood ratios for the association between HER-2 status and no histologic response to chemotherapy.
Data synthesis: survival at 2 years
HER2-positive status tended to be associated with a worse 2-year survival with a 1.78-fold higher risk death at 2 year without significant differences between studies (statistically significant between-study heterogeneity; Fig.2). Conversely the HER2-positive status was strongly associated with a worse 2-year survival and between-study heterogeneity was no longer significant when the analyses were limited to the 25% IHC cutoff (Table 3).
Each study is shown by the name of the lead author, and the relative risk with 95% confidence intervals. Also shown are the summary relative risk (total) and 95% confidence intervals with random effect calculations.
Relative risk for association between HER2 status and mortality in 2 years.
Discussion
This meta-analysis showed that HER-2 positive status in patients with osteosarcoma had no discriminating ability for identifying poor versus good responders to chemotherapy. HER2-positive status showed a modest association with worse 2-year survival in spite of no statistical significance.
The potentially discrepant results concerning survival are challenging. The discrepancy may be due to bias because the correlation with survival was modest and not formally significant for the protein-level data. Alternatively, it might offer a hint on the potential mechanism of action of HER2. HER2 may contribute to tumorigenesis as an early event and determine osteosarcoma features such as tumor grade, type, aggressiveness, and metastatic potential without regulating response to chemotherapy.
Heterogeneity was driven in the searched studies; 3 studies correlated HER2 over-expression with a poor response to chemotherapy and a poor outcome [10-12]. Remarkably, 2 other studies concluded that HER-2 overexpression predicts a better survival and is less frequent in metastatic disease[15]. In the 4 remaining studies, no HER-2 over-expression could be demonstrated[21,22]. These inconsistent findings regarding HER-2 status and its significance in osteosarcoma raise questions about methodological differences. These studies differed considerably in their use of antibody and quality controls, scoring systems, interpretation of positivity of the sample and validation of the IHC result. In breast cancer, HER2 testing and standardization of the method used has been an important issue, because only patients with HER-2 over-expression are eligible for trastuzumab treatment[23].
The quality of the antibody used is also important, since a high rate (up to 40%) of false-positive tests have been reported, due to variable sensitivities[24]. False-positive cases are particularly noted when moderate positivity occurs, and in these cases confirmation of the positive result with other tests was required[25]. On the other hand, different scoring systems to assess HER-2 positivity have been used in osteosarcoma studies. Interpretation of the stained samples may have a high inter-observer variability and a low rate of reproducibility. This particularly occurs when the staining is heterogeneous, weakly-positive, in non-malignant cells, cytoplamic staining or when retraction artifacts occur. Nevertheless, we should acknowledge that the clinically optimal method for detection of HER-2 and the optimal cutoff level are unknown.
Given these measurement issues and the small sample size of most studies, it is probably not surprising that there is a large body of literature showing that HER2 status correlates with outcomes for various malignancies as well as a considerable body of literature of “negative” studies. The documentation or rebuttal of such association should preferably be performed with large-scale evidence on many hundreds of patients because single studies of limited sample size, when seen in isolation, may yield spurious results.
Some other limitations of this meta-analysis should be discussed. First, we tried to identify all relevant data and retrieve additional unpublished information, but the fact that some data were missing was unavoidable. Second, there was some unavoidable variability in definitions of methods, measurements, and outcomes in each study, despite our effort to standardize definitions. Third, the sample size of the meta-analysis is still modest. However, given that osteosarcoma is not very common on a population basis, the sample size of this investigation is one of the largest to date among studies targeting this malignancy. Finally, the estimates that we obtained were unadjusted for other parameters that may be related to osteosarcoma outcomes such as tumor size, histological type, and chemotherapeutic regiments. It is unknown whether a detrimental based on HER2 status on survival may be explained by other tumor determinants such as size and type
The prognostic value of HER-2 in patients with osteosarcoma also should be examined in the context of other proposed molecular markers, including P-glycoprotein, P53 and others. Scotlandi et al.[10] described that immunohistochemical analysis also revealed a positive correlation between HER-2 and P-glycoprotein over-expression in osteosarcoma, and their data demonstrated that both HER-2 and the P-glycoprotein have prognostic value for osteosarcoma, and in fact, event-free survival analyses showed that, among all the clinicopathological parameters considered here, only over-expression at diagnosis of HER-2 or P-glycoprotein, as well as of both molecules, was significantly associated with a higher relapse rate and a worse clinical outcome. They also further demonstrated the value of these markers in predicting osteosarcoma prognosis and response to therapy, and revealed that HER-2 and P-glycoprotein may also act together as adverse prognostic factors in determining a worse prognosis in patients who present inherent increased expression of both molecules at diagnosis. And Ferrari et al.[26] showed that patients with HER2 expression in the primary osteosarcoma were more likely to have multiple metastases and shorter recurrence-free interval compared with patients in whom HER2 expression was not observed, whereas differences in P-glycoprotein, P53, and Bcl-2 expression were not related to differences in metastatic pattern. Postrecurrence survival (PRS) was influenced by P53 expression levels in pulmonary metastases, with patients who had negative staining for P53 having a significantly better PRS rate relative to patients with positive staining for P53.
- Received May 19, 2008.
- Accepted July 4, 2008.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer