Abstract
OBJECTIVE To investigate the effect of endostatin and doxycycline on melanoma cellular proliferation and tumor angiogenesis.
METHODS The effects of endostatin and doxycycline were studied in mice transplanted with B16 melanoma cells. The mice were divided into 4 groups that were treated as follows: endostatin treatment (E group), doxycycline treatment (D group), endostatin plus doxycycline trearment (DE group), controls (C group) received no treatment. Following 9 days of treatment the tumor tissue was removed to compare the differences in the tumor necrotic rate and micro-vessel density (MVD) among the different groups. Immunohistochemical staining was conducted to detect the expression of proliferating cell nuclear antigen (PCNA) in the different groups.
RESULTS The MVD of the 3 experimental groups was significantly less than the control group, (F = 10.888, P < 0.05), indicating that doxycycline and endostatin can inhibit tumor angiogenesis by decreasing the tumor blood supply. This effect results in inhibition of tumor cellular proliferation and promotion of tumor cell necrosis. The tumor cell necrotic rate of the 3 experimental groups were all significantly higher than the C group (F = 7.229, P < 0.05) and the difference between the DE and C groups also was statistically significant. PCNA expression in all 3 experimental groups was statistically less than the C group (F = 17.729, P < 0.05).
CONCLUSION The combined use of endostatin and doxycycline in vivo can influence PCNA expression and angiogenesis in melanoma, and significantly inhibit melanoma cellular proliferation.
keywords
Introduction
Melanoma is highly invasive, metastatic and rapidly progressive, with a poor prognosis[1]. Endoststin (ES) is a fragment of type XVIIIcollagen which can specifically inhibit endothelial cellular migration and promote apoptosis. It is one of the most powerful anti-angiogenic factors known. Doxycycline is a tetracycline derivative. Besides its antibiotic effect, it also is a broad-spectrum matrix metalloproteinase(MMP) inhibitor. In order to examine the effect of these two agents on tumor angiogenesis and proliferation, we treated mice carrying transplanted melanoma with endostatin and doxycycline to determine their effect on the tumor micro-vessel density and rate of necrosis. We also performed immunohistochemical staining to detect the expression of PCNA.
Materials and Methods
Experimental animals
Fifty seven, black, inbred, C57/BL6 mice, including 29 males and 28 females, were purchased at 6~7 weeks of age from the Institute of Hematology of Blood Diseases, Chinese Academy of Medical Sciences & Peking Union Medical College.
Agents
Acid hydrochloride-doxycycline tablets (0.1 g/tablet) were purchased from the Tai Hua Medicine Ltd. Co. in Harbin. Endoststin (15 mg/3 ml/syringe) was a gift from the Xian Sheng Mai De Jin Biologic Medicine Ltd. Co. in Shandong.
B16 melanoma tumor tissue
A tumor cell suspension (1 × 106/ml) was made by mincing tumor tissue from transplanted B16 melanoma in mice and adding isotonic sodium chloride. A 0.2 ml suspension was injected into the groin of each mouse.
Endostatin and doxycycline treatment
The mice were divided into 4 groups on the 6th day after tumor implantation. i) Endostatin (E) treated group (n = 14); 0.05 ml endostatin was injected subcutaneouly in the abdominal region of each mouse (far from the tumor tissue) each day. ii) Doxycycline (D) treated group (n = 14); Doxycycline was administated in the drinking water at a concentration of 1 mg/ml[2], and each mouse was given 40 ml of water each day. iii) Doxycycline plus endostatin (DE) treated group; The mice were given doxycycline and endostatin at a dosage similar to the E and D groups. v) Control (C) group (n = 14). Following transplantation of the tumor, the mice were supplied with normal water and food ad libitum.
The length and width of the tumor nodules were measured every day and the tumor volume calculated. Tumor volume (mm3) = (length × width2) / 2. The mice were sacrifed on the 9th day after drug asministration, the tumor tissues separated and 4 μm width paraffin sections prepared.
Immunohistochemical staining
The polyclonal antibodies used in this study were rabbit anti-PCNA (Boster Wuhan, China, dilution 1:50). The bioepiderm goat anti-rabbit antibody and S-P compound were applied, antigen restored with microwave treatment and DAB applied. The sections were counterstained with H&E, dehydrated, cleared and mounted.
Counting methods
Tumor cell necrosis rate. The sections were viewed at 400 ×, and 10 fields were randomly selected. The necrotic cell number was determined by viewing 100 random melanoma cells in every field. The average number of necrotic cells was used to determine the necrotic rate in a section.
MVD
The number of vessels in each field of 5 random fields (× 400) was counted and the average used to determine the MVD.
PCNA positive expression rate
The sections were viewed at 400 × and 10 fields were randomly chosen. The positive cell (with dark-yellow nucleus) number was counted by viewing 100 random melanoma cells in every field. The average number was used to determine the expression of the proteins in a section.
Statistical analysis
SPSS 10.0 for windows statistical software was used to analyze the data. All data were analyzed using ANOVA. P < 0.05 was considered to be statistically significant.
Results
Tumor growth after endostatin and doxycycline treatment
Tumor nodules were appreciable by the 9th day after tumor inplantation in all of the C57/BL6 mice. Tumor growth in all of the 3 treated groups was slower than growth in the C group.
MVD and the tumor cell necrotic rate
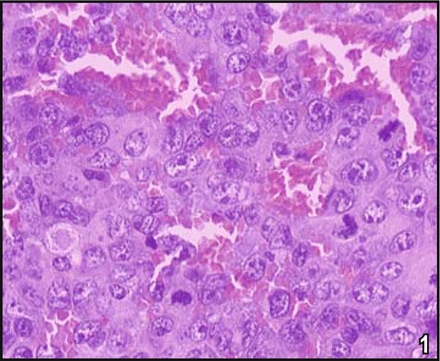
The MVD in the 3 treated groups was statistically lower than in the control group. Furthermore, MVD developed in groups DE, E, D and C from low to high. Group comparision indicated that the differences between DE and D as well as between DE and E were statistically significant (F = 10.888, P < 0.05, Figs.1 and 2).
C group, MVD in the tumor tissue is greater, and the number of necrotic tumor cells less than other groups (S-P method, × 400).
DE group, MVD in the tumor tissue is less, and the number of necrotic tumor cells greater than other groups (S-P method, × 400).
The tumor cell necrosis rates were in the order of DE, D, E, C groups from high to low. The rate in the DE group was statistically higher than in the C group (F = 7.229, P < 0.05). Necrotic cells in regions with a greater blood supply were relatively less than in regions with less blood supply (Table 1).
Necrosis rate, MVD, PCNA positive expression rate in 4 groups.
Determination of the positive expression rate of PCNA by immunohistochemical staining
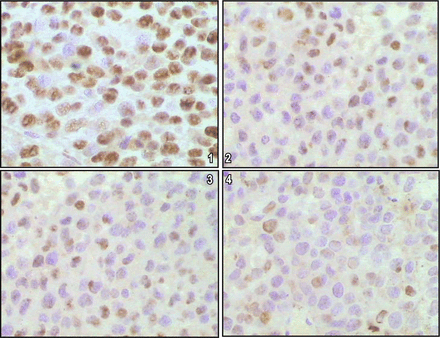
The positive expression of PCNA in the 3 drug-treated groups was lower than in the C group. The rate of expression increased in the order of groups from DE to E to D. The difference between the 3 treated groups and group C was statistically significant (F = 17.729, P < 0.05). The difference between group DE and the D group, as well as between the DE and E groups was statistically significant (Table 1, Fig.3). More tumor cells in regions near blood vessels expressed PCNA compared to regions distant from vessels.
1, C group; 2, D group; 3, E group; 4, DE group (S-P method, × 400).
Discussion
It is well known that tumors stimulate their own growth by enhancing angiogenesis in order to supply nutrients for growth and dissemination. The degree of angiogenesis is a result of a dynamic balance controled by various inhibitory or facilitative factors. Theoretically, inhibitory factors shift the balance in the direction of decreased angiogenesis, influencing the supply of tumor nutrients, inhibiting cellular proliferation and promoting necrosis, thereby constraining tumor growth. At present, anti-angiogenesis research is an area of intense interest in oncology. Many researches have indicated that numerous anti-angiogenic drugs can inhibit tumor growth, dissemination and metastasis[3], and some agents have been tested in clinical trials[4] MMPs often highly expressed in malignant tumors[5] are important regulatory factors that promote the passage of tumor cells through connective tissue, and thus aid in tumor micro-vessel formation.
In our research, MVD development in the 3 treated groups was less compared to the control group, and the anti-angiogenetic effect of combined use of endostatin and doxycycline was better than either endostatin or doxycycline given alone. Endostatin, an angiogenesis inhibitor identified and purified from a murine hemangioendothelioma cell line, inhibits vessel endothelial cell proliferation and migration. In addition it accelerates endothelial apoptosis induced by bFGF[6], decreases expression of VEGF[7], induces formation of local adhesion plaques, influences formation of action tonofibrils, resulting in inhibition of endothelial cell mobility and angiogenesis[8]. In a mouse model, endostatin was shown, to cause tumor ablation without toxicity, and in an athymic mouse model, it inhibited the growth and metastasis of human nasopharyngeal carcinoma[9].
Doxycycline is a tetracycline derivative that interferes with the synthesis of bacterial protein. It also is a broadspectrum MMP inhibitor indicated by recent research[10] to cause tissue disintegration of aneurysms of the aorta and reduce periodontitis and arthritis. It also has been used for cardiovescular disease therapy such as angiosclerosis and vessel restenosis, and to inhibit tumor infiltration, metastasis and angiogenesis. Doxycycline restrains MMPs expression on a transcriptional level, and directly inhibits activity of MMPs by combining with their active sites. It has been shown that doxycycline can inhibit MMP expressiom by MUM2B melanoma cells and tongue carcinoma[11,12].
Decreased expression of MMPs can inhibit both a switch to an angiogenesis phenotype and activation of angiogenesis; inhibit degradation of cell-cell junctions in tumor tissue and restrict tumor cells from entering the blood circulation; and decrease degradation of the basal membrane and extracellular matrix to inhibit new vessel formation by endothelial cells. All of these effects help to decrease the blood supply and restrain melanoma growth. Endostatin and doxycycline inhibit angiogenesis by different mechanisms, so their combined use can efficiently constrain tumor growth.
The tumor cell necrotic rate in the 3 groups treated was higher than the control group, resulting in statistically significant difference between the DE and C groups. The PCNA positive expression rate in the 3 groups treated was less than the control group, again showing a statistically significant difference between them. PCNA is a DNA polymerase accessory protein, which is synthesized less in a cell non-proliferation stage, and more with cellular hyperplasia and transformation. It can serve as an index reflecting cellular proliferation. Our results indicated that doxycycline and endostatin promoted necrosis and restricted proliferation by decreaseing micro-circulation formation. In addition to this mechanism, it has been reported that endostatin and doxycycline constrained tumor growth by the following: inducing apoptosis directly; inhibiting the Wnt pathway[13]; decreasing tumor cell transformation from G1 to S stage[14]; influencing adhesion and migration of vascular smooth muscle cells; and influencing reformation of the fibro-collagen matrix[15].
Many researchers have affirmed the effects and applications of endostatin and doxycycline to restrain tumor growth, but most research on the anti-tumor effect of these agents has been limited in vitro cell lines. In our study, we applied the two agents in an animal model, to observe their anti-angiogenic effect and their influence on tumor cell necrosis and proliferation, thus providing more evidence for their anti-tumor effect. The combined effect of these two agents on anti-tumor growth may be of importance in tumor therapy.
Footnotes
The work was supported by a grant from National Natural Science Foundation of China (No.30370554).
- Received August 17, 2007.
- Accepted December 24, 2007.
- Copyright © 2008 by Tianjin Medical University Cancer Institute & Hospital and Springer