Abstract
OBJECTIVE This study was conducted to explore the effect of Rg3 on inhibition of proliferation and induction of apoptosis in bladder cancer cells.
METHODS The EJ bladder cancer cel! line was treated with Rg3 at various concentrations. Cell proliferation was measured by the MTT assay. Morphological changes in the cells were observed by fluorescent staining using Hoechst 33258. The cell cycle and apoptotic rate were analyzed by flow cytometry (FCM) and the expression of caspase-3 in cells was detected by immunocytochemistry. DNA ladder analysis was conducted by agarose gei electrophoresis'.
RESULTS Rg3 inhibited proliferation of EJ cells in a concentration-dependent manner, resulting in an IC50 for Rg3 at 48 h of 125.5 μg/ml. When treated with 150 μg/ml of Rg3 for 24 h and 48 h, the cells showed apoptotic morphological characteristics including condensed chromatin, nuclear fragmentation, apoptotic bodies and bright fluorescent granules as well as a higher caspase-3 expression. The FCM assay indicated that Rg3 altered the cell cycle and induced apoptosis of the EJ cells, when treated for 24 h and 48 h with 75 μg/ml of Rg3 as well as for 48 h with 150 μg/ml. The percentages of cells in the S phase and the G2/M transition were increased, whereas the percentages of cells in the G0-G1, transition were decreased. The apoptotic rates were increased from (1.05±0.17)% in the control group cells to (8.41 ±0.98)%, (18.57±2.20)% and (33.98±1.64)% respectively. Significant changes in the DNA ladders, showed that the effects of Rg3 were displayed in a dose and time dependent manner.
CONCLUSION The results suggest that Ginsenoside Rg3 exerts an inhibitory effect on proliferation of EJ cells by inducing apoptosis.
keywords
Treatment of cancer patients with most chemotherapeutic drugs often induces toxic side effects and drug resistance. At present, natural agents from animal and plant sources are being considered more as potential anti-cancer drugs which are part of Chinese traditional medicine.[1] Ginsenoside Rg3 is a chemical trace component of MW 784.30 Da which was extracted from the red Pinax. After Kitagawa in Japan determined its molecular formula to be C42H720i3 in 1982, research on its anti-tumor action has been conducted in China. [2,3] Many studies have shown that Ginsenoside Rg3 can inhibit cellular proliferation, suppress infiltration and metastasis of cancer cells, as well as enhance immune function of tumor-bearing mice.[5,6] Studies have demonstrated that Rg3 is devoid of side effects on bone marrow, the heart, lung, liver, kidney and nervous system,[6] suggesting that Rg3 is a relatively safe and effective chemotherapeutic agent. The study of its anti-tumor mechanism is of increaseing interest. Bladder cancer is a very common malignant tumor [7] with an incidence rate that ranks it to be the most common of the urogenital system cancers in China. Following surgery, chemotherapeutic treatment is the most important modality of therapy that may enhance survival rate. Our goal has been to search for safe effective chemotherapeutic drugs. In this study we utilized human bladder cancer lines to explore the mechanism of the Rg3 inhibitory effect on cellular proliferation and induction of apoptosis. The results suggest that Rg3 may be useful for bladder cancer therapy.
MATERIALS AND METHODS
Materials
The human bladder transitional EJ cell carcinoma, an invasive cell line, was purchased from the Immunology Department of Medical College in Beijing University, China. Ginsenoside Rg3 was provided by the Dalian Fusheng Pharmacy Corp, at a purity of 99.69%. A storage solution was prepared at a 600 μg/ml concentration in medium, preserved at -70°C. Methylthiazolyldiphenyl-tetrazolium bromid (MTT) and Hoechst 33258 were products of the Sigma Chemical Corp, USA. Dulbecco’s modified Eagle’s medium (DMEM) was obtained from Invitrogen Life Technologies, Carlsbad, USA. Fetal calf serum was a product of Ustars Biotech, Dalian, China. The polyclonal antibody for caspase-3 was obtained from the Santa Cruz Biotechnology, Santa Cruz, CA, USA. The goat antirabbit antibody (IgG/Bio) and SP (Streptavidin/Peroxidase) Histostain™ -Plus Kits were bought from Beijing Zhongshang Biotech, Beijing, China.
Ceil culture
The cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal calf serum, 100 U/ml penicillin and 100 μg/ml strepto-mycin and incubated in 5% CO2 at 37°C.
MTT cell viability assay
To determine cell viability, the MTT assay was performed as previously described.[1] Cells (5×103) were seeded into 96 well plates with 100 μl medium and cultured for 24 h. For the treatment groups, Rg3 was added in 50 p,l at a final concentration of 37.5 μg/ml, 75 μg/ml, 150 μg/ml, 300 μg/ml and 600 μg/ml. For a control, 50 pJ of DMEM complete medium without Rg3 was added. The cells were cultured for 48 h. MTT (20 μl) containing a concentration of 5 mg/ml was added into each well. Then the cells were incubated at 37 °C for 4 h, the medium removed and the wells supplied with 150 pi DMSO, followed by measuring the absorbance with a plate reader at 490 nm. The inhibitory rate of cellular growth was calculated knowing that 490 nm absorbance is directly proportional to the number of living cells. Each point represents the mean of 6 independent samples and the assay was repeated 3 times. Inhibitory rate = (the A490 of control group—the A490 of treatment group)/the A490 of control group×100%. IC50 was calculated with NDST software.
Morphological observation of apoptotic cells
Following incubation of the EJ cells with or without 150 μg/ml Rg3 on coverslips in 6 well plates for 24 h and 48 h, the coverslips were washed with PBS. The cells were fixed with glutaraldehyde for 5 min, stained with 5 μg/ml Hoechst 33258 for 5 min, washed with PBS, and examined under an Olympus multifunction microscope. The cells exhibiting nuclei (intact or fragment) with a bright green fluorescence were scored as apoptotic cells.
Assay of cell cycle distribution
After the EJ cells were incubated with or without Rg3 for 24 h and 48 h in 75 μg/ml of Rg3 and for 48 h in 150 μg/ml, the cells were harvested by trypsinization, washed with PBS, counted and adjusted to 1×106 cells/ml. The cells were fixed in 70% ethanol, treated with 40 μg/ml RNase A and stained with 18 μg/ml propidium iodide (PI). The phase distribution in the cell cycle and the apoptotic rate were analyzed with CellQuest and ModFit software by flow cytometry.
Immunocytochemistry of caspase-3
The EJ cells were incubated with or without 150 μg/ ml Rg3 for 24 and 48 h on coverslips in 6 well plates, fixed with ice-cold 80% acetone for 10 min and washed with PBS. Each coverslip was treated for 10 min with newly diluted 3% H2O2 to destroy internal peroxidases. The samples were incubated for 20 min with normal goat serum to block non-specific antibodies, then the cells were incubated for 90 min with polyclonal primary antibody for caspase-3 antigen (1:50 dilution) at 37°C. The coverslips were incubated in goat anti-rabbit secondary antibodies (IgG/Bio-Rad) for 30 min at 37°C, using SP (Streptavidin/Peroxidase) Histostain™ -Plus Kits and DAB staining. The nuclei were counterstained with hematoxylin.
DNA agarose gel electrophoresis
DNA fragmentation was determined to evaluate apop tosis. After the EJ cells were incubated without or with 75 μg/ml Rg3 for 24 and 48 h and for 48 h in 150 μg/ml Rg3, the cells were harvested from the 6-well culture plates and centrifuged at 2000 g for 10 min. The cell pellets were resuspended in 1 ml of lysis buffer (10 mM Tris-HCl, pH 7.5, 10 mM NaCl, 10 mM EDTA, 100 (μg/ml proteinase K, and 0.5% SDS) and incubated for 1 h at 50 °C. After lysis, samples were extracted with 1 ml of phenol(neutralized with TE buffer, (pH 7.5), followed by extraction with 1ml of chloroform/isoamyl alcohol(24:l). The aqueous supernatants were precipitated with 2.5 volumes of ice-cold ethanol plus 10% volume of 3 M sodium acetate, at -20 °C overnight. After centrifugation at 13,000 g for 10 min, the pellets were air-dried, resuspended with 50 °C of TE buffer, pH 7.5, supplemented with 0.1 (xg/ml RNase A, and electrophoretically separated on a 1.5% agarose gel for 2 h. Photos of the gels were taken by UV transillumination.
Statistical analysis
All data were treated with SPSS 13.0 statistical software. The values were expressed as means ±SD. Student’s tests were used for statistical analysis. P values of <0.05 were considered to be significant.
RESULTS
Effects of Rg3 on cell viability
Incubation of the cells with Rg3 at high concentrations lead to a toxic effects manifested by cell rounding and eventual cell death. Treatment of the cells with Rg3 at a concentration range of 37.5-600 (xg/ml inhibited cellular proliferation. The value of IC50 was found to be 125.5 μg/ml at 48 h. Rg3 inhibited cellular proliferation in a concentration-dependent manner.
Morphological changes of apoptotic cells induced by Rg3
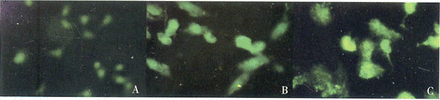
When the EJ cells were treated with 150 μg/ml Rg3 for 24 h and 48 h, the cells displayed typical apoptotic morphology including reduced volume, condensed chromatin, nuclear fragmentation, apoptotic bodies and brighter green fluorescence Fig.1. However, the control cells showed no apoptotic characteristics.
Fluorescence micrographs of cells after treatment with Rg3 (A: EJ control cells; B:EJ-Rg3-24 h cells; C: EJ-Rg3-48 h cells, x 400).
Inhibitory effect of Rg3 on proliferation of cells
Effects of Rg3 on the cell cycle distribution and apoptotic rate
Cell cycle analysis by FCM revealed that Rg3 induced changes in the cell cycle. Treatment of the EJ cells for 24 h and 48 h in 75 (xg/ml of Rg3 as well as for 48 h in 150 p,g/ml, resulted in an increase in the percentage of cells in the S phase and G2-M transition were increased, whereas the percentage of cells in the G0-G1 transition was decreased. The apoptotic rates in the sub-G0/G1 peak (apoptotic peak) region were increased from 1.05±0.17% in the control group cells to (8.41 ± 0.98) %, (18.57± 2.20)% and (33.98±1.64)% in Rg3-EJ cell groups Table 2.
Effects of Rg3 on the cell cycle and apoptotic rate
Effects of Rg3 on expression of caspase-3
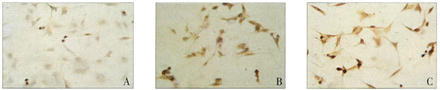
Immunocytochemical analysis was used to detect caspase-3 expression in the EJ cells. The cells in the treatment group showed a higher gray scale and stronger positive signal by DAB visualization compared to the cells in the control group, which showed a weak or negative brown staining in the cytoplasm Fig.2
Immunocytochemical detection of caspase-3.(A: EJ control cells; B:EJ-Rg3-24 h cells; C: EJ-Rg3-48 h cells, × 200)
DNA fragmentation
After the EJ cells were treated for 24 and 48 h in 75 μg/ml of Rg3 and for 48 h in 150 p,g/ml, DNA agarose gel electrophoresis showed a typical ladder of DNA. The response developed in a dose and time dependent manner with Rg3 treatment, as shown Fig.3
Effects of Rg3 on the Induction of apoptosis analyzed by DNA agarose gel electrophoresis. (M: 100 bp DNA Maker; 1: control; 2: 75 μg/ml Rg3-24 h; 3:75 μg/ml Rg3-48 h; 4: 150μg/ml Rg3-48 h).
DISCUSSION
Multidrug resistance is a major problem in cancer chemotherapy.[8] Recently, there have been considerable efforts to search for naturally occurring substances that can inhibit, reverse, or retard multi-stage carcinogenesis.[9] Pinax ginseng has been widely used in traditional herbal medicine for the treatment of various diseases, and many of its pharmacological actions are attributed to the ginsenosides. Certain fractions or purified ingredients of ginseng have been shown to exert anticarcinogenic and antimutagenic activities. Ginsenoside Rg3, one of the constituents of heat-treated ginseng, has been shown to significantly inhibit tumor growth, invasion and metastasis of various cells including rat hepatoma cells, B16 melanoma cells, human small cell lung carcinomas, human pancreatic adenocarcinoma cells, colon cancer cells, intestinal cancers, prostate cancer cells, human ovarian carcinoma cells, human leukemia cells, breast cancers, gastric cancers and so on.[10]
A decrease in cell apoptosis is one of the characteristic changes in tumors. Ginsenoside Rg3 inhibits proliferation, invasion as well as metastasis of tumor cells, which might relate to with its induction of apoptosis, but the molecular mechanism remains unclear. Induction of apoptosis is an important research strategy for the development of of anti-tumor drugs. Malignant growth of cancer cells is the result of abnormal proliferation or a defect in the regulatory mechanism for cell death. An imbalance between cell proliferation and apoptosis would result in tumorigenesis.
Apoptosis, an evolutionarily conserved form of cell suicide, requires specialized biochemical reactions. The central component of these reactions is a prote-olytic system involving a family of proteases called caspases. These enzymes participate in a cascade that is triggered in response to proapoptotic signals, and culminates in the cleavage of a set of proteins, resulting in disassembly of the cell. Caspase-3 is an important apoptotic inducer, which plays a central role in cell apoptosis. At the molecular level, increased expression/activity of caspases-3 will result in cell death.[11-13]. Our studies indicated that caspase-3 had a stronger expression in cells treated with Rg3. In contrast, caspase-3 expression was weak or negative in control cells.
Apoptosis is a specific mode of cell death recognized by the characteristics of morphological, bio-chemical, and molecular changes.[14] The changes in cell morphology, such as condensation of chromatin, nuclear fragmentation, DNA ladders and apoptotic bodies, are the most reliable markers of apoptosis.[15] These classic apoptotic morphologic changes were observed in Hoechst33258 stained cells treated with Ginsenoside Rg3. In addition, our study showed that cells treated with Rg3 accumulated in the S phase and G2/M transition of the cell cycle. The alterations in the cell cycle and induction of apoptosis are in agreement with the findings of Kung et al.[16] They concluded that cell death was evoked, not by a biochemical lesion, but by the dissociation of normally integrated cell cycle events. Our results demonstrated that cell proliferation was suppressed with a decrease in the growth rate, G0-G1, transition reduction, S phase and G2-M transition delay, and induction in apoptosis. When the EJ cells were treated with Rg3, the cells were arrested at the S phase and G2-M transition, and subsequently cell growth was inhibited through a caspase3-mediated apoptotic mechanism. Our data suggest that Ginsenoside Rg3 may be useful for chemotherapy of tumors, including bladder cancer. Ginsenoside Rg3 has an excellent potential as an anti-cancer drug.
- Received May 9, 2006.
- Accepted August 23, 2006.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer