Abstract
OBJECTIVE The antioxidative system in human hepatocellular carcinoma was investigated.
METHODS The activities of cytosolic catalase (CAT), superoxide dismutase, glutathione peroxidase (GSH-Px), glutathione S-tranferase and levels of reduced glutathione, total protein thiols and malondialdehyde were assayed in 10 cases of hepatocellular carcinoma and adjacent normal liver.
RESULTS Hepatoma tissues showed higher activities of CAT, GSH-Px and lower content of total antioxidative capacity compared to adjacent normal liver tissue (P<0.05).
CONCLUSION These findings suggest that the antioxidative defense-related enzymes and antioxidants are largely regulated in hepatoma cells. However, the mechanism which is not clear requires further investigation.
keywords
Liver cancer is one of the most frequent malignant tumors, which has the third mortality rate of all malignancies. It has been reported that the incidence of hepatocellular carcinoma maybe related to an imbalance in metabolism of trace elements.[1,2] A number of elements (Fe, Cu, Zn, Se, Mn, etc.) are components of active centers in redox enzymes, which are important for human health. Certainly, an imbalance of the elements would change the activities of the enzymes. Blood tests indicate that the selenium concentration, activities of glutathione peroxidase (Se-GSH-Px) and superoxide dismutase (Cu, Zn-SOD) are significantly higher in the serum and whole blood of liver cancer patients.[2,3] Significant differences of metalloproteins between human hepatoma and their adjacent tissues in both types and quantities have been observed in our earlier work.[4] Until now, few investigations in this area have been conducted, some of which have shown an inverse relationship between redox enzymes and antioxidants. The interrelationships still remain confused.
Reactive oxygen species (ROS), such as superoxide anions (O2·-), hydrogen peroxide (H2O2), and hydroxyl radicals (·OH), are a series of highly reactive substances. They can damage intracellular components such as membranes (peroxidizing lipids), proteins (depressing enzyme activity), and DNA(inducing cell mutations).[5] Metabolism of carcinogens can produce ROS, so the relationship between free radicals and cancer has been of great interest. In this work, in order to investigate the relationship between the antioxidative system and carcinoma, hepatoma and adjacent liver tissues of same individual have been studied by analyzing some redox enzymes and related species, which are closely relevant to ROS.
MATERIALS AND METHODS
Materials and instruments
Mannitol 3-(N-morpholino) propane sulfonic acid (M-OPS), and ethylene glycol bis (2-amino-ethyl-ether-tetraacetic acid (EGTA) were purchased from the Beijing Jingke Chemical Co. (China). Phenylmethyl sulfonyl fluoride (PMSF) was obtained from Tianxian-gren Bioengineering Co. (China). All the above reagents were ultra pure. Sucrose and ethanol were from the Beijing Chemical Plant, while phosphonic acid was purchased from the Beijing Hongxing Chemical Plant. Reagent kits for enzymes and for measuring biological parameters were provided by the Nanjing Jiancheng Bioengineering Institute. A Beckman L7 (U.S.A.) ultra centrifuge was used to prepare cytosolic extracts.
Enzyme activities and biological parameter measurement
Cytosolic extracts of hepatoma and adjacent tissue were prepared by ultra centrifugation as follows. Human liver tissue 1~2 g was cleaned of fat, divided into pieces and suspended in 10 volumes of MOPS buffer (containing 0.25 M mannitol, 0.075 M sucrose, 0.5 mM EGTA, 2 mM MOPS, and 1 mM PMSF). The tissue was homogenized using a glass homogenizer and the resulting solutions centrifuged at 10,000 g for 2 h. The supernatants which comprised the liver cytosolic extracts were stored at -20 °C. All the processes were performed at 4°C.
Measurements of the following were conducted using the corresponding kits: superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), glutathione S-transferase (GST), reduced glutathione(GSH), total sulfhydryl groups (Thiol), malonaldehyde (MDA), total antioxidative capacity (T-AOC). For total protein quantification, the coomassie brilliant blue G250 colorimetric assay was used (Bradford, 1976).
Statistical analysis
The analytical data were subjected to a statistical comparison using the paired t-test.
RESULTS
Ten pairs of hepatoma and adjacent normal liver tissues of eight patients with liver cancer were collected and analyzed to reduce the individual differences. The results of measuring various enzyme activities and antioxidant levels are summarized in Table 1.
Activities of enzymes or concentrations of related compounds in hepatoma and adjacent normal tissues (Mean±SD)
Activities of antioxidative enzymes
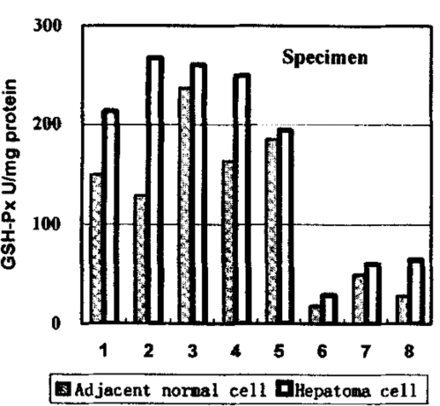
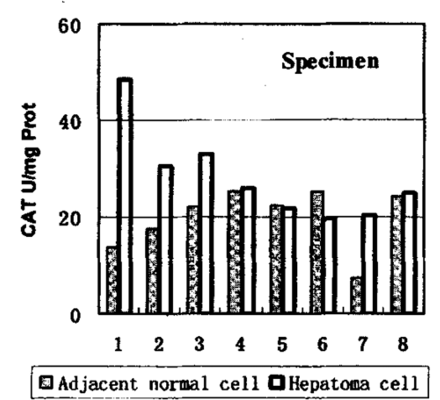
As shown in Table 1, the CAT and GSH-Px activities show significant differences. Comparisons of 8 individual tissue and tumor samples for GSH-Px and CAT activities are shown in Figs.1 and 2, respectively. In spite of the great individual differences among the samples, the trend between hepatoma and adjacent cells are quite identical. The enzymes in hepatoma tissues were more active than those in adjacent tissues. But the GST and SOD failed to give similar results. The GST was slightly depressed in hepatomas, however, the SOD activities did not show a similar trend and differences were more or less.
Activity of GSH-Px in hepatomas and adjacent normal tissues.
Activity of CAT in hepatomas and adjacent normal tissues.
Levels of T-AOC and Thiols
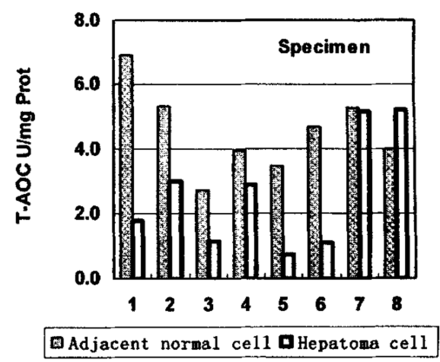
From Table 1 it can be seen that the levels of GSH, total thiols are higher in hepatomas, however that of MDA was slightly higher in adjacent tissues, while T-AOC was significantly higher in adjacent tissues. A paird comparison of T-AOC between hepatoma and adjacent normal tissues is shown in Fig.3. Most samples exhibited significant differences.
T-AOC in hepatoma and adjacent normal tissues (Eight pairs were compared, tissues from same patient chose one pair of samples).
DISCUSSION
The results of enzyme activities resemble that of Lee et al., [6] who reported that the activities of CAT, GSH-Px, and SOD in hepatoma cells were 4.3, 2.9 and 2.8 fold higher than those in normal cells, while the activity of GST in normal cells was 3.2 fold higher than hepatoma cells. Yokomizo et al.[7] also discovered lower activity of GST in human epidermoid cancer cell lines. However, in research using the H22 primary hepatic cancer injected into mouse liver, Chen et al.[8] observed decreased activity of SOD. In another investigation of liver cancer induced by DENA and 2-ac-etamidofluorene, Li et al.[9] indicated that the activity of SOD was depressed at an early stage, and then dramatically increased in a later promotion phase. These observations implied that in the process of carcinogenesis, the antioxidative system had an activating response, which lagged after carcinoma developed.
The primary roles of these enzymes in organisms are to quench endogenous free radicals. Since our samples are from terminal carcinomas, all the observed phenomenon should be the result of an extended period, which indicate that free radicals were increased during carcinomatous development. Since each measurement is paired with tissue from the same patient, the most reasonable resource of excess free radicals in hepatomas should be a consequence of carcinoma development. Excess free radicals in vivo induce the production of antioxidative enzymes in organisms, so that the enzyme level returns to normal or even more active, which quenches excess ROSs. So the level of free radicals which is represented by MDA didn't show a significant difference. The depression of GST concentration could result in lower detoxification ability of human liver. It is reported that excess hepatic iron might be a carcinogen, while an iron deficit can decrease the risk of liver cancer. There are many more transferrin receptors on the surface of tumor cells (5~10 times), and about 100-1,000 fold higher inside the tumor cells.[10] Much greater significant difference in CAT has been observed in this study, which suggested that hydrogen peroxide could play an important role in the process of liver cell carcinoma.
T-AOC includes the total capability of the cellular antioxidative system, not only the antioxidative enzymes, but also involves some important small molecules, such as vitamin E and C, carotene, and GSH, etc. In biological membranes, a chain reaction is continual once the peroxidization process of lipid is initiated by free radicals. Neither SOD, CAT nor GSH-Px can break the reaction. However, Vitamin E, because of its solubility, mainly exists in hydrophobic areas of the cell, so it could terminate the chain reactions via competition with fatty acid by combining with LOO · produced by a lipid peroxidization process.[11] Vitamin C protects extracellular lipid from peroxidation.
Eventhough there was enhanced enzyme activity and no significant difference in the GSH level, the large decrease of the T-AOC level in hepatomas indicates that those antioxidative small molecules should be heavily consumed. These small molecular antioxidants play a more important role than GSH when excess ROSs are produced, as GSH can be regenerated in vivo, whereas no mechanism is available for vitamin regeneration. In fact, the activities of enzymes related to GSH synthesis are greatly up-regulated in carcinoma cells.[6,12] In that study, the activities of γ-glutamyl-cysteine synthetase, γ-glutamyltranspeptidase, glutathione synthetase, glutathione reductase in hepatoma cells were 5.9, 9.5, 8.land 4.9 fold higher than those of normal cells, respectively. Thus, the production of ROSs greatly decreases the antioxidant content, depresses the capability of cell antioxidation, and finally makes the cell vulnerable. The different mechanisms in adjustment of endogenous and exogenous antioxidative reagents suggests that more attention should be given to the use of antioxidants in anti-tumor treatment.
Cancer proliferation will seriously interfere with normal functions of liver tissue. In our research, some antioxidative enzymes represented significant differences suggesting that an imbalance of the antioxidative system has a strong impact on whole process of carcinogenesis. A clear mechanism of the antioxidative defense system and its role require further investigation.
Footnotes
This work was supported by the National Natural Science Foundation of China (Major Project No.10490180) and Chinese Academy of Sciences (No. KJCX-N01).
- Received October 31, 2005.
- Accepted December 14, 2005.
- Copyright © 2006 by Tianjin Medical University Cancer Institute & Hospital and Springer